Key research themes
1. How can pH measurement accuracy and calibration be optimized across highly alkaline to natural aqueous systems?
Accurate pH measurement is fundamental across chemical, environmental, and industrial applications but faces challenges due to non-idealities in solution chemistry, wide pH ranges, and instrument limitations. Optimizing pH meter calibration methodologies, especially for extreme pH values (e.g., high alkalinity in cementitious systems) and variable ionic strengths in natural waters, is essential to ensure traceability, reduce systematic errors, and reconcile discrepancies between measurement approaches and theoretical models.
2. What role does ion-specificity and solution chemistry play in the precision and interpretation of pH measurements near interfaces and in non-standard solvent systems?
pH measurement and interpretation are influenced by complex physicochemical interactions including ion pairing, ionic dispersion forces, solvent effects, and interfacial phenomena. These effects lead to ion-specific deviations in glass electrode responses and differences in acidity scales between solvents, complicating conventional pH measurement and necessitating advanced approaches to unify pH scales and account for co-ion impacts. Understanding these effects is critical for accurate acidity assessment in biological, environmental, and mixed solvent media.
3. How can machine learning and advanced analytical methods enhance pH value estimation accuracy using low-cost, accessible tools like pH paper under varying environmental conditions?
Conventional pH measurement by pH electrodes provides high accuracy but is costly and requires careful calibration and maintenance. Alternatively, colorimetric pH strips are affordable and easy to use but typically suffer from low precision and subjective interpretation, especially under varying lighting conditions. Integrating machine learning algorithms with RGB color data extracted from pH paper images enables precise, automated pH quantification, improving accuracy to nearly that of potentiometric methods and enabling portable, cost-effective pH sensing for field and household applications.


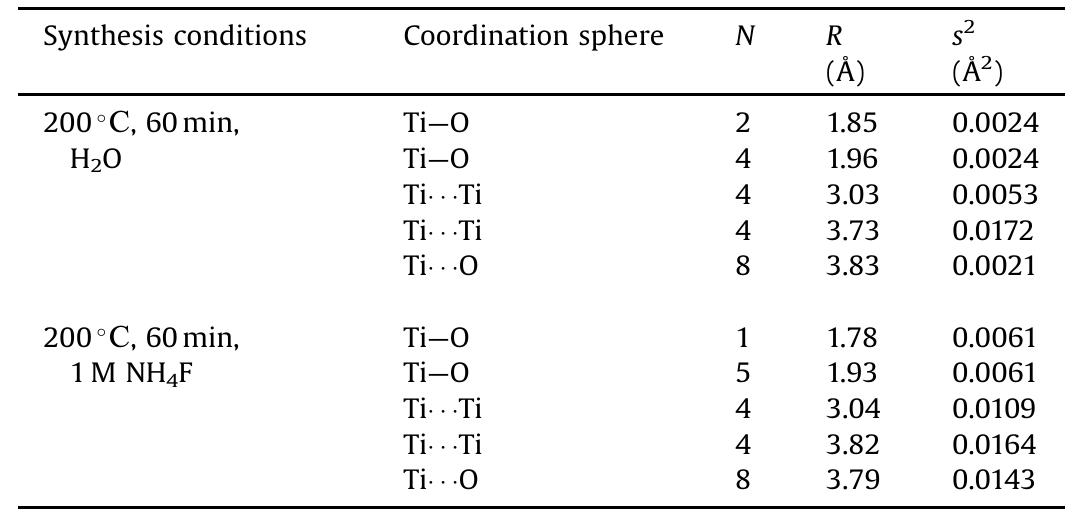
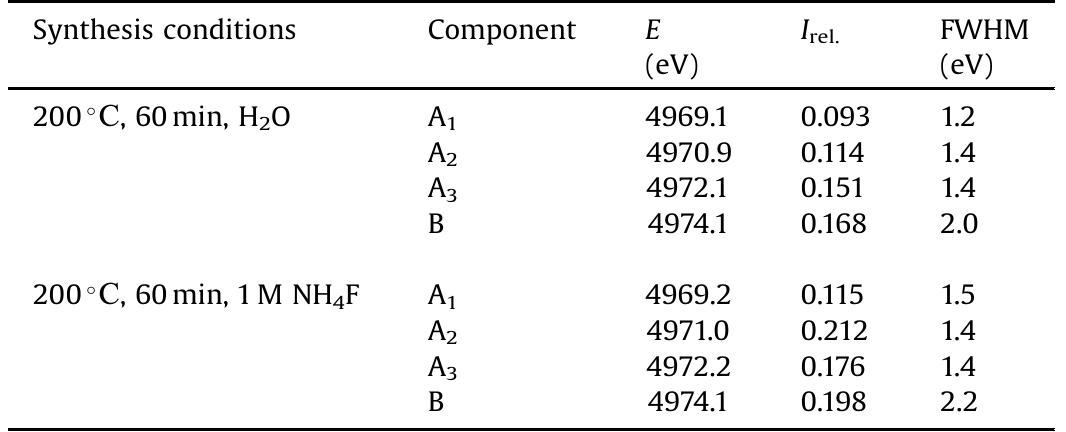
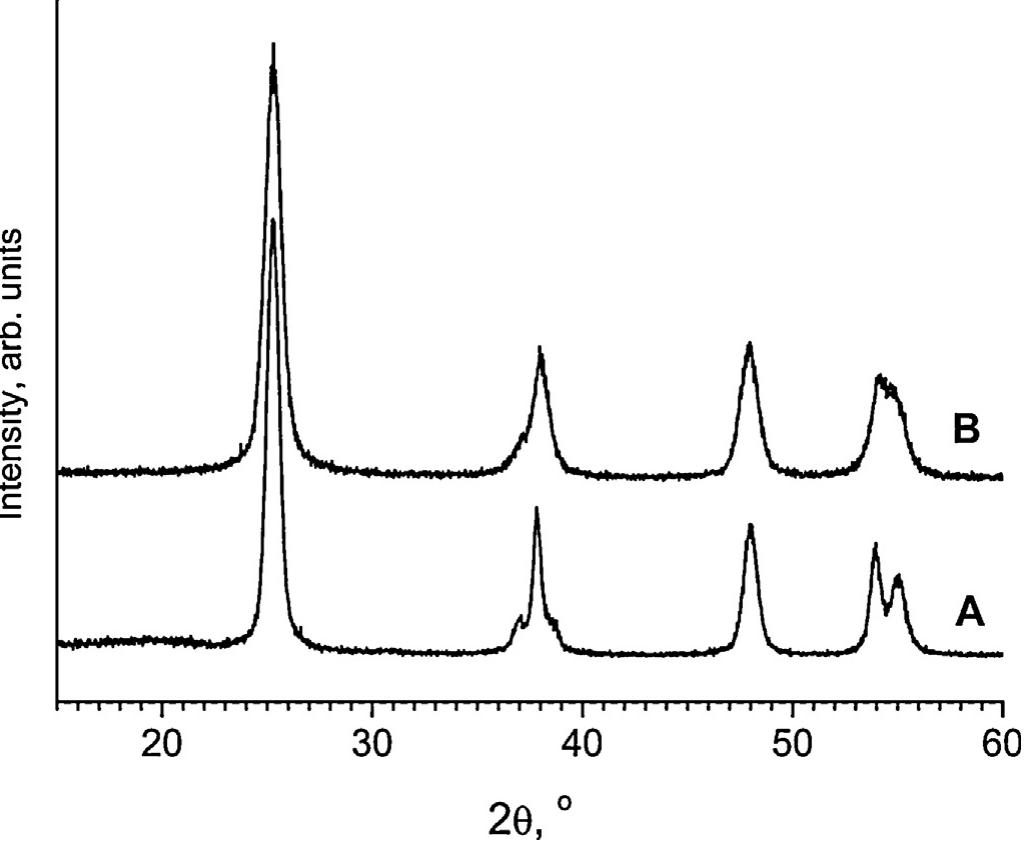
![Main characteristics of the titanium dioxide samples obtained by microwave-assisted hydrothermal treatment of titanium dioxide xerogels with the addition of ammoniun luoride. * Phase composition of Evonik Aeroxide™ TiOz P25, as estimated using the method of Spurr and Myers [40], was 74 wt.% anatase and 26 wt.% rutile. The content of the amorphous phase was 5% at most. Particle size, as estimated using the Scherrer equation, was 31 nm for anatase and 58 nm for rutile.](https://figures.academia-assets.com/100855763/table_001.jpg)

![Fig. 8. Ionic current values versus the temperature for the TiO2 sample, prepared from TiO2-xH20 at 200°C for 60 min, in the presence of 1M NH,F. SS a SEES EA ENS LER! NI OREN Se Pee NE ME e | Tare Set a eee et ade High photocatalytic activity of titania samples synthesized in fluorine-rich media may be due to either a high specific surface area of the samples or the presence of fluorine on the TiO, surface [62]. In order to clarify the possible role of fluorine in the photocatalytic performance of titania, we have calcined the most active fluorinated titania sample at 200°C for 2h to eliminate fluorine. Such a treatment preserved particle size and did not change the specific surface area, while the fluorine content decreased from 2.3 at.% to approximately 0.5at.%. The photo- catalytic activity of the calcined sample remained practically](https://figures.academia-assets.com/100855763/figure_007.jpg)
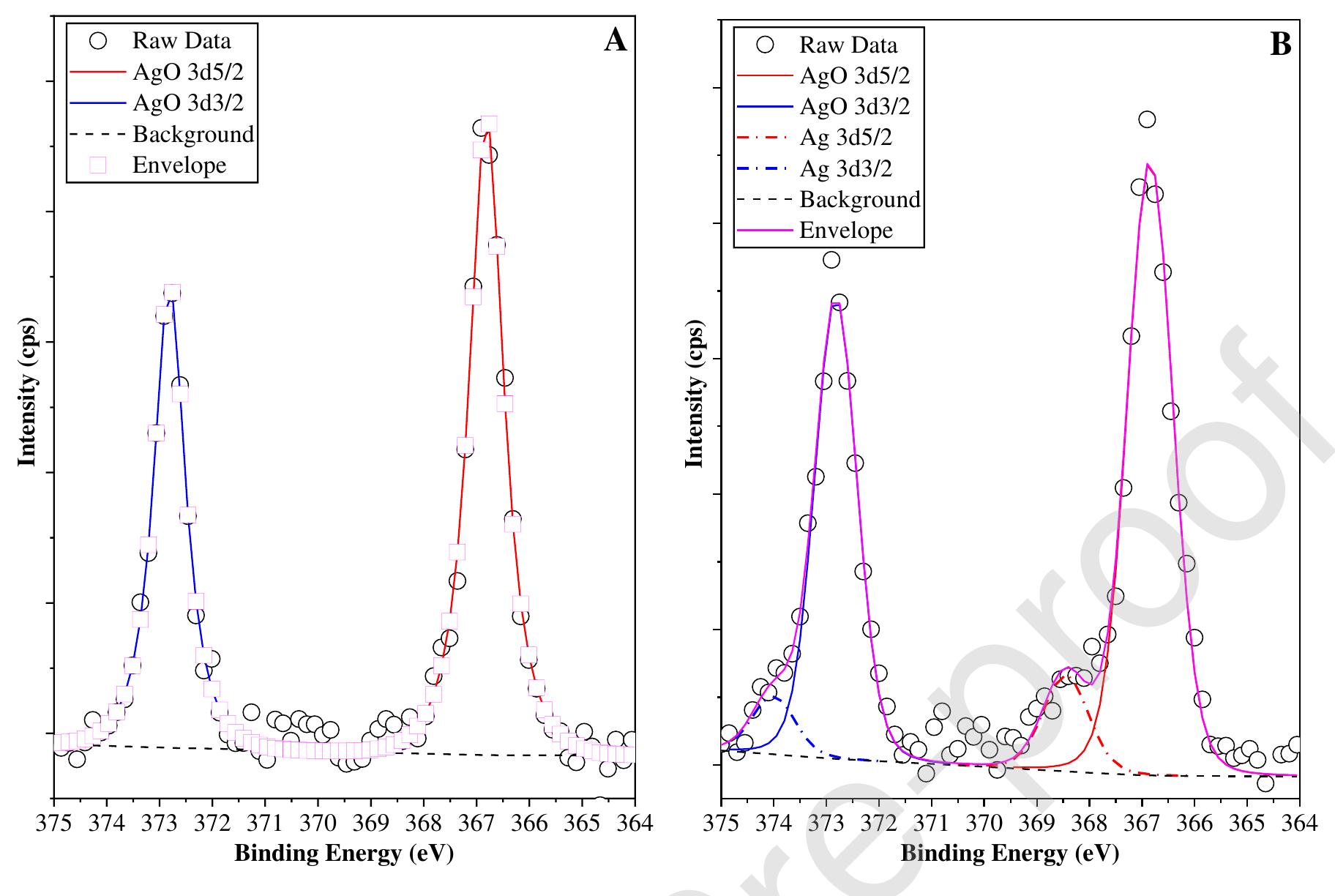
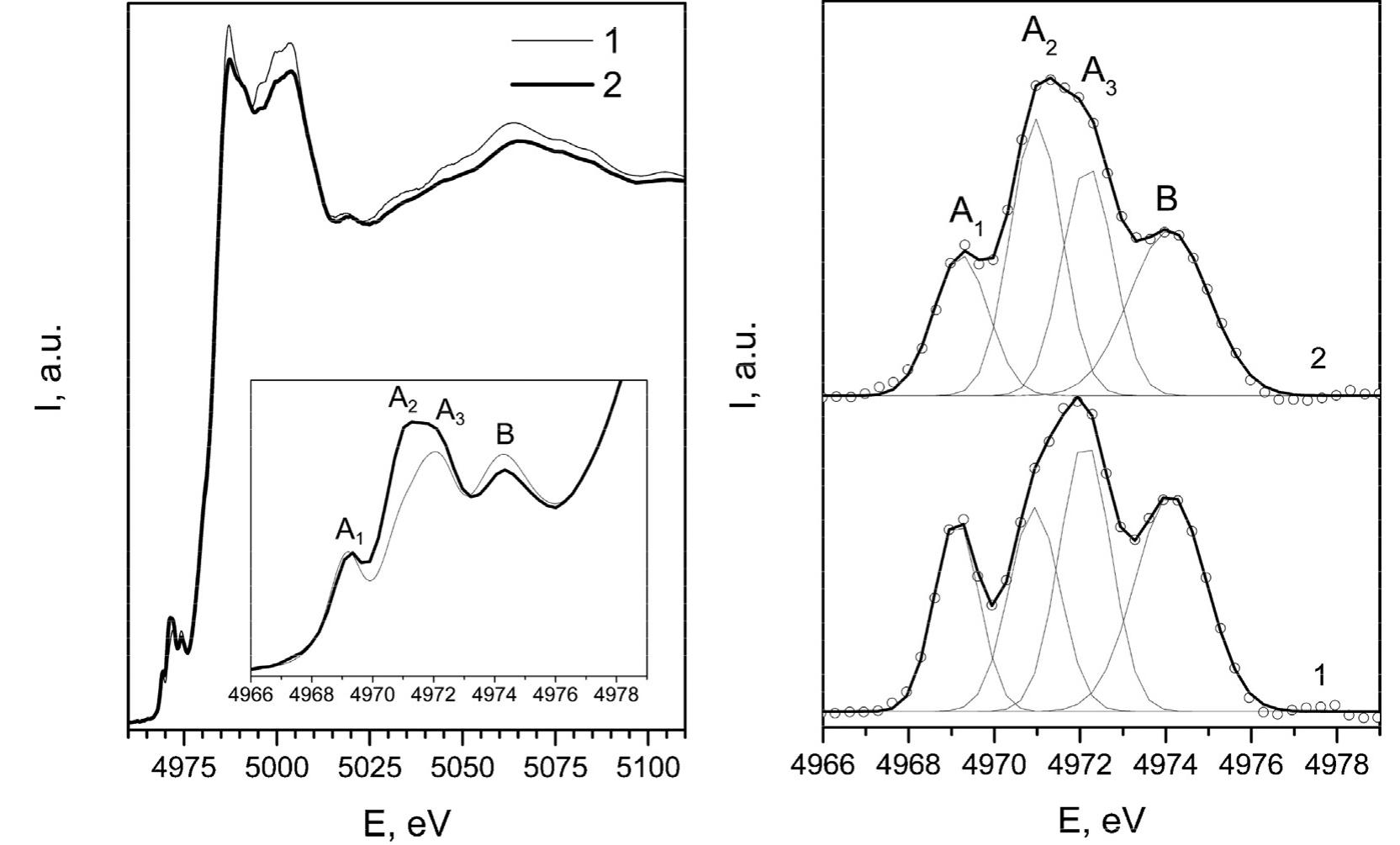
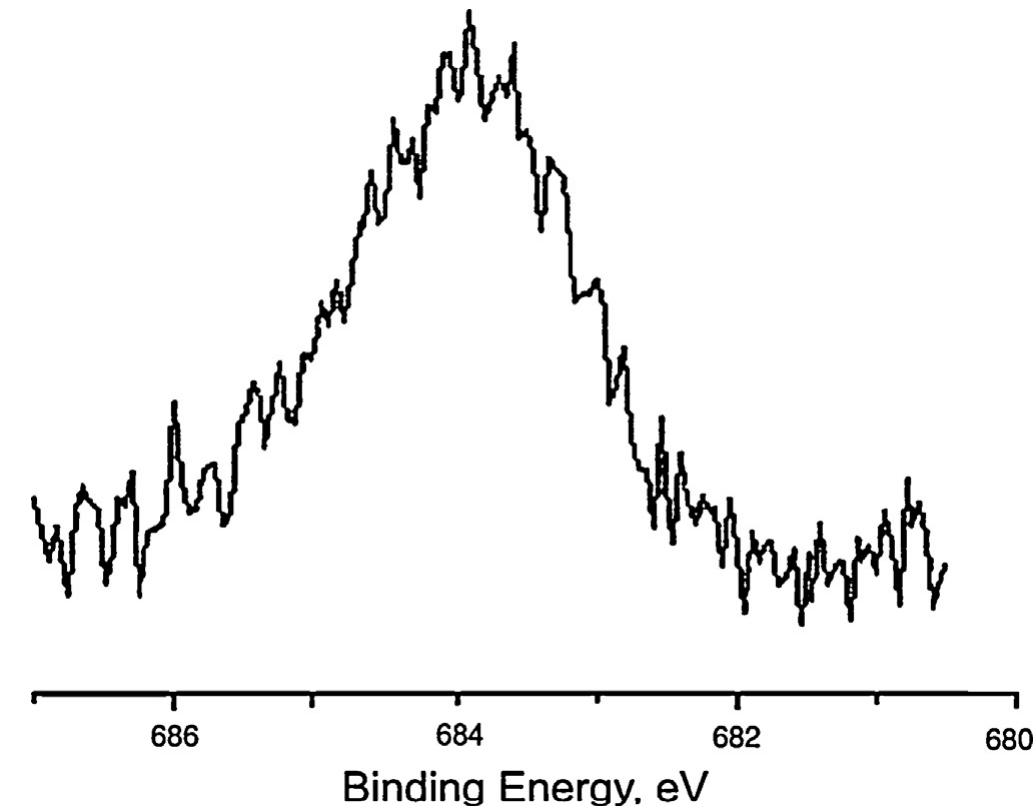
![Fig. 6. XPS survey of the TiO2 spectrum for a TiO2 sample synthesized from TiO2-xH20 at 200°C for 60 min in the presence of 1M NH4F. Fluorine localization on the surface of TiO2 was confirmed by optical diffuse reflectance spectroscopy and thermal analysis data. The absorption spectra of titanium dioxide synthesized under different conditions do not differ from each other, practically. There is no shift of the absorption band edge in comparison to the titanium dioxide prepared by hydrothermal-microwave treatment of the titanium dioxide xerogel in distilled water. The bandgap for all of the samples was 3.2 eV, which is in excellent accordance with the generally accepted value of 3.2 eV [61]. This can be explained by the same band structure of the samples, and therefore the](https://figures.academia-assets.com/100855763/figure_006.jpg)
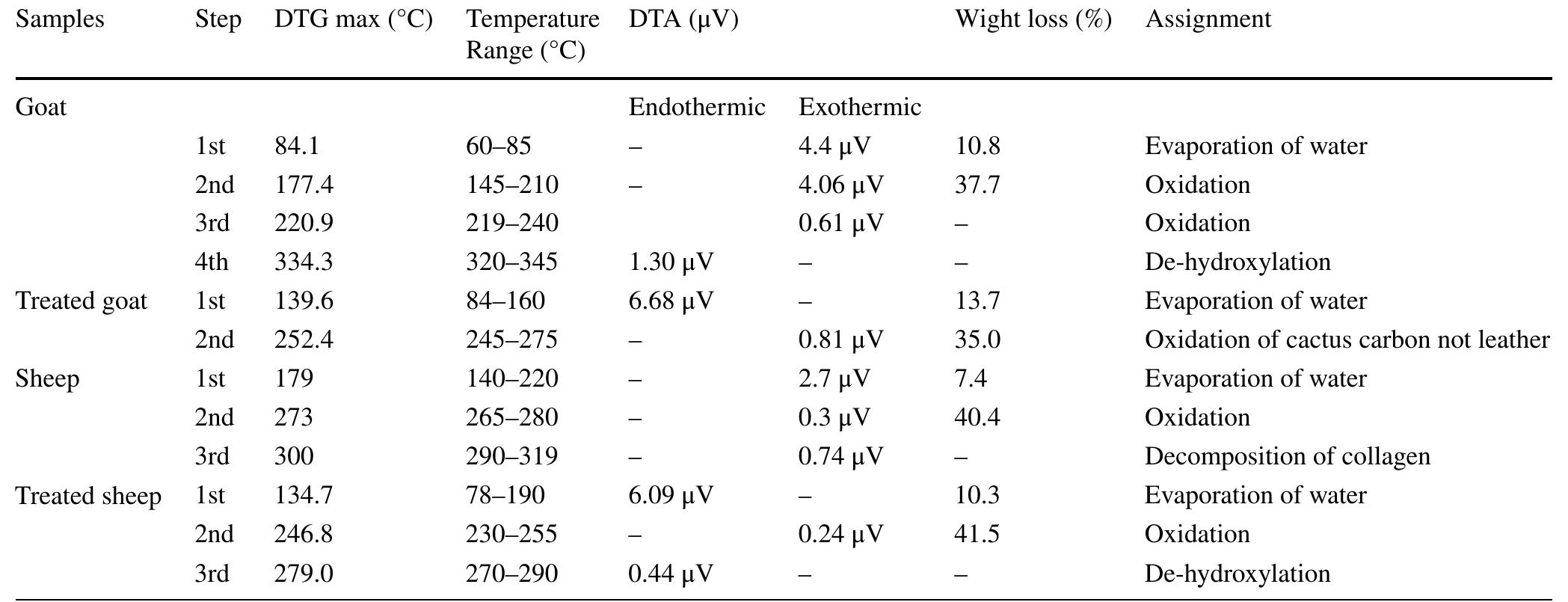
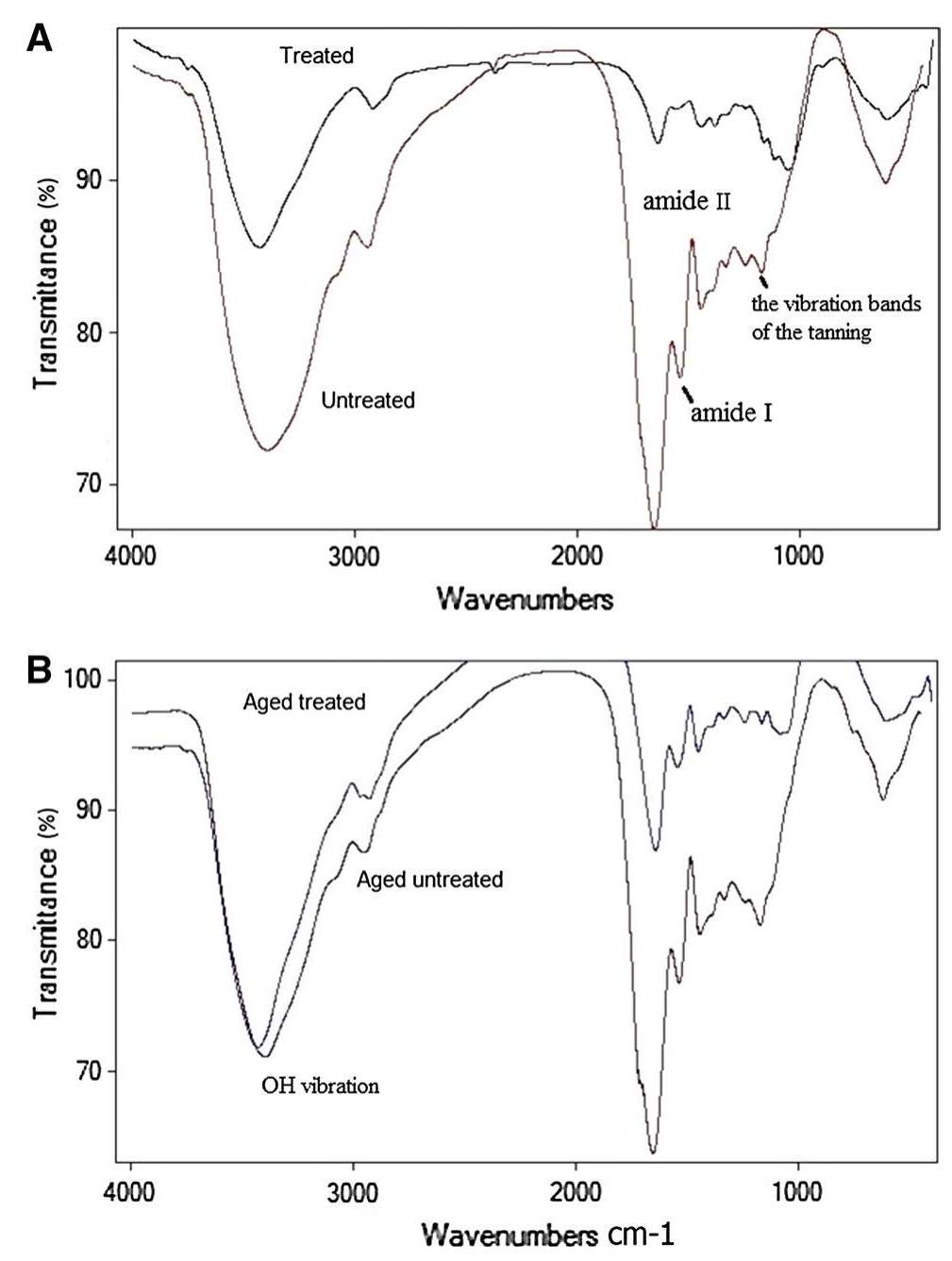

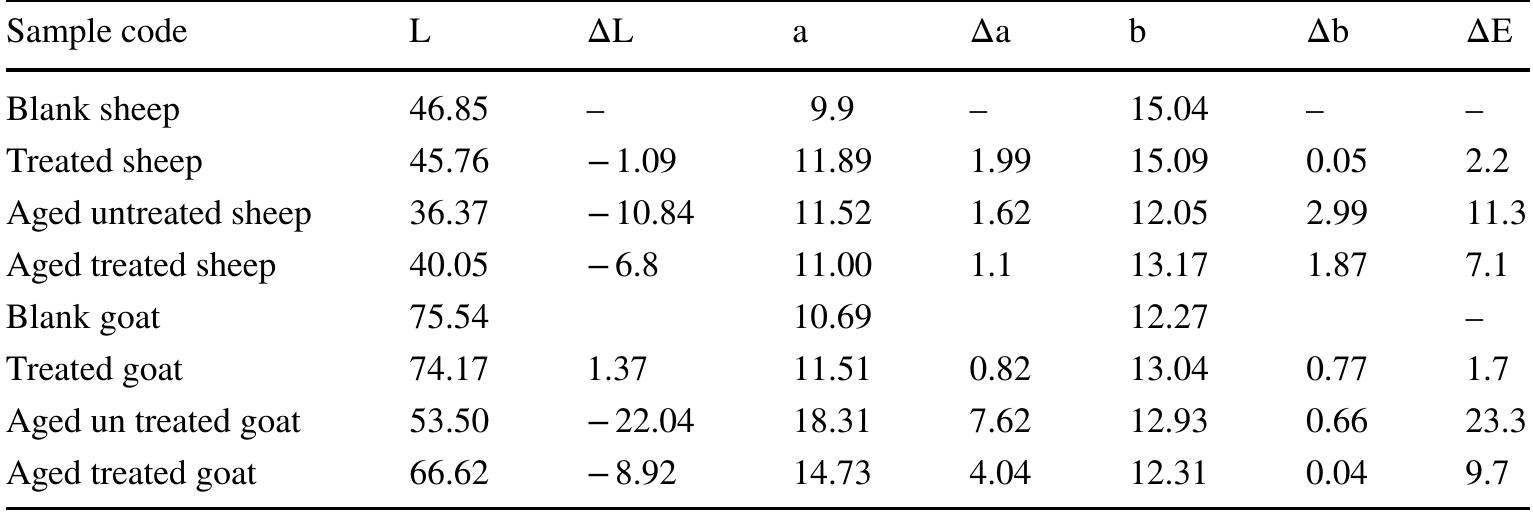


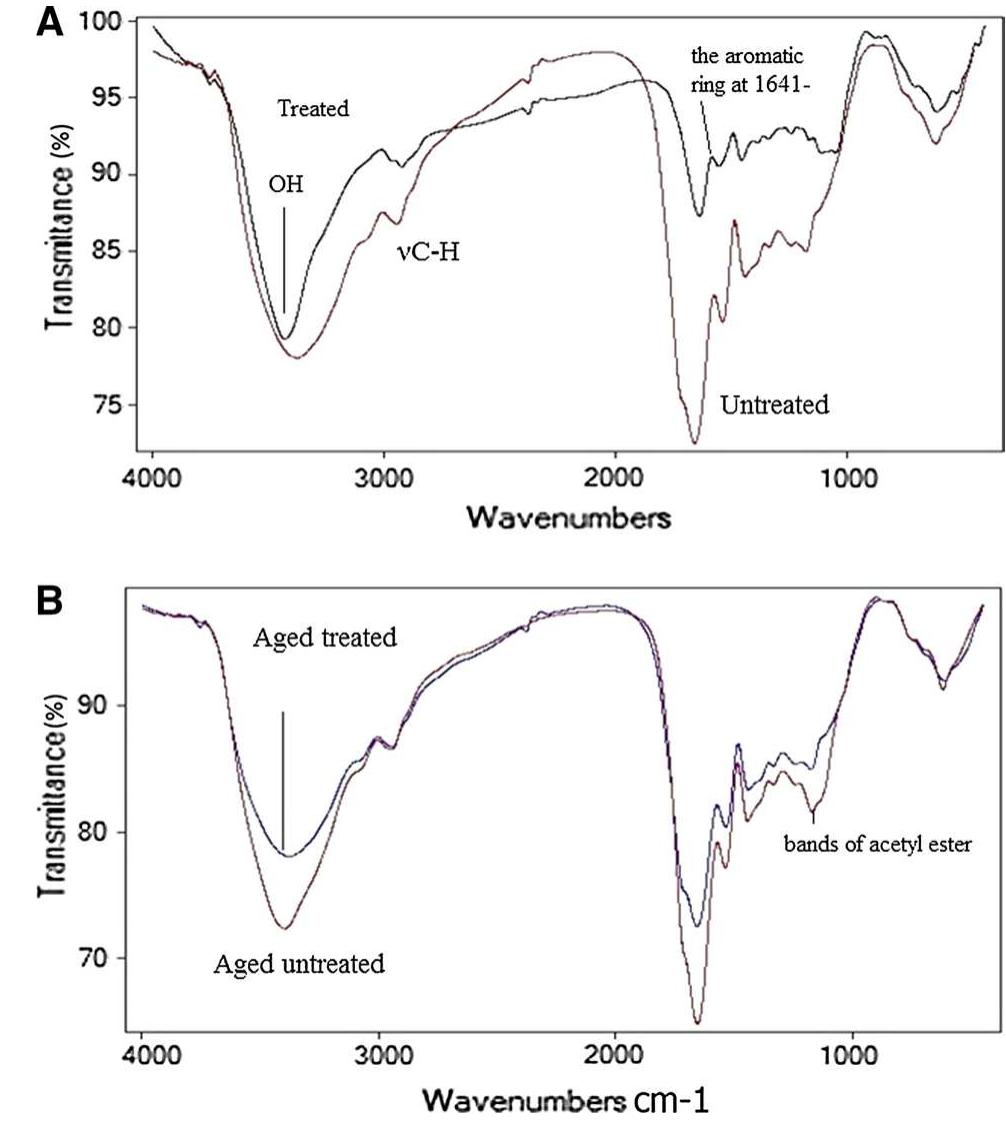
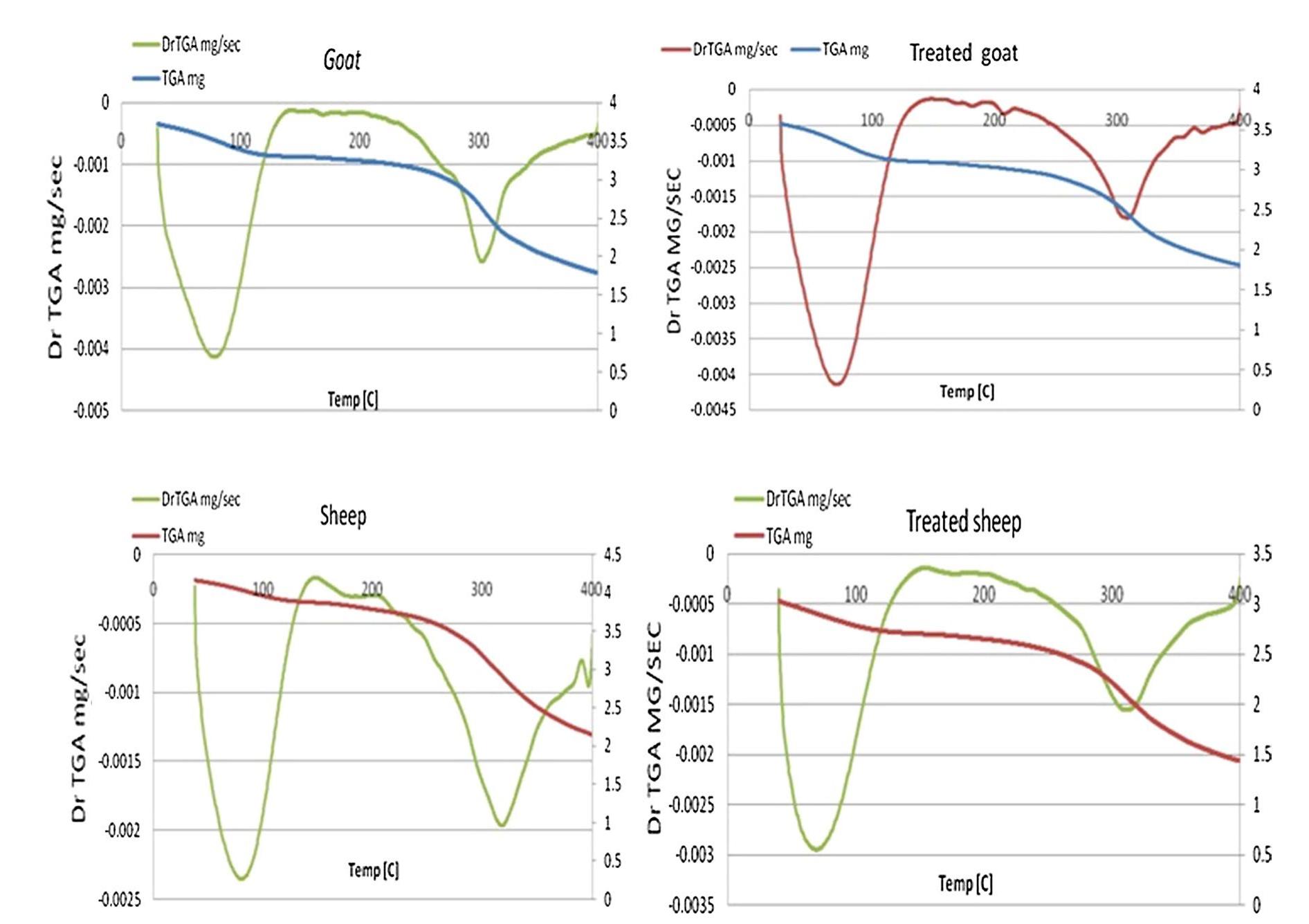
![3392 cm’, confirming the loss of hydration and hydrogen bond between amides, N—H vibration, C=O stretching and amide III at 1560, 1630, 1242 cm™!, respectively. They lost their intensities resulting in changes in protein structure, the ratio of amide I (1650 cm!) to amide II (1560 cm™') decreased. Accordingly, the B sheet to a-helix ratio increased which illustrates a dramatic decrease in protein folds [35, 36].](https://figures.academia-assets.com/95593951/figure_002.jpg)
![aging. Recently, it has been discovered that aloe vera gel contains polyphenols [46, 47]. 4 Conclusion The unique properties of aloe vera gel have contributed to reducing leather oxidation processes during accelerated aging, leading to a slight reduction of the mechanical prop- erties as compared to untreated samples. As well, the gel supported the protein structure by increasing the intensi- ties of vibrations for amides I, IJ and III, enhancing the strength of alpha helix structure. The polyphenols o gel played a significant role in reducing leather color change during thermal aging inasmuch its high resistance to dation. Oxidation resistance is one of the first defense against degradation of organic matter therefore aloe Oxi- ines vera gel protect leather during aging systematic. On the mor- phological level, the used concentration penetrated the](https://figures.academia-assets.com/95593951/figure_006.jpg)

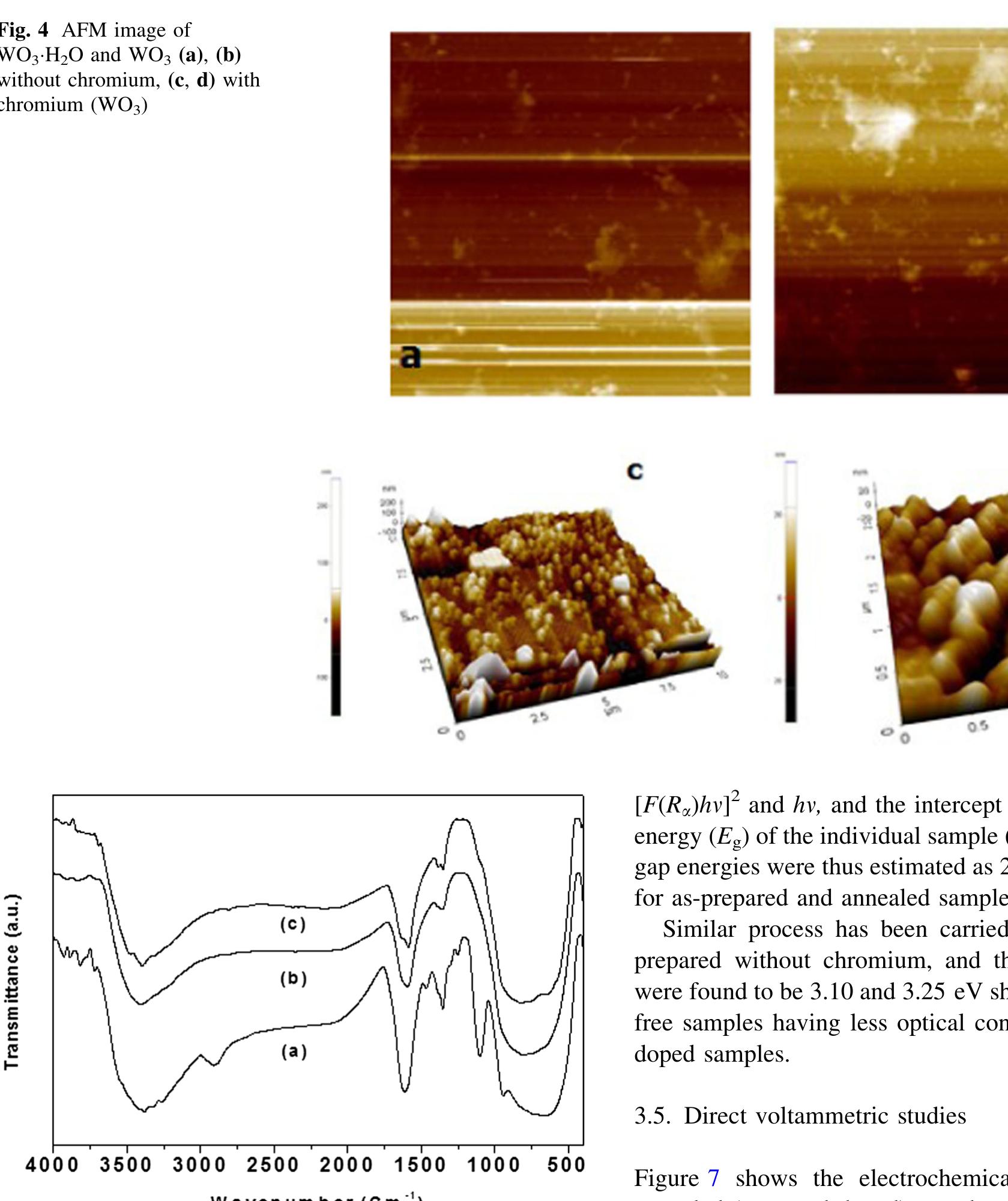
![The trailblazer solution was prepared by dissolving 2.49 g of tungstic acid (H2WO,) in 10 ml of sodium hydroxide (NaOH) with one molar ratio concentration. It results in yellow colored hydrated sodium tungstate solution due to According to reaction (1), high concentrations of hydrated sodium tungstate would shift the reaction to the right ensuring the formation of WO3_,, although many intermediate steps and thus compounds and phases may exist. It is thus the formation of WO; should have dependence on the acid medium. To convert WO, to neutral WO3, excess divalent oxygen must be removed by microwave irradiation method [14].](https://figures.academia-assets.com/94645252/figure_001.jpg)
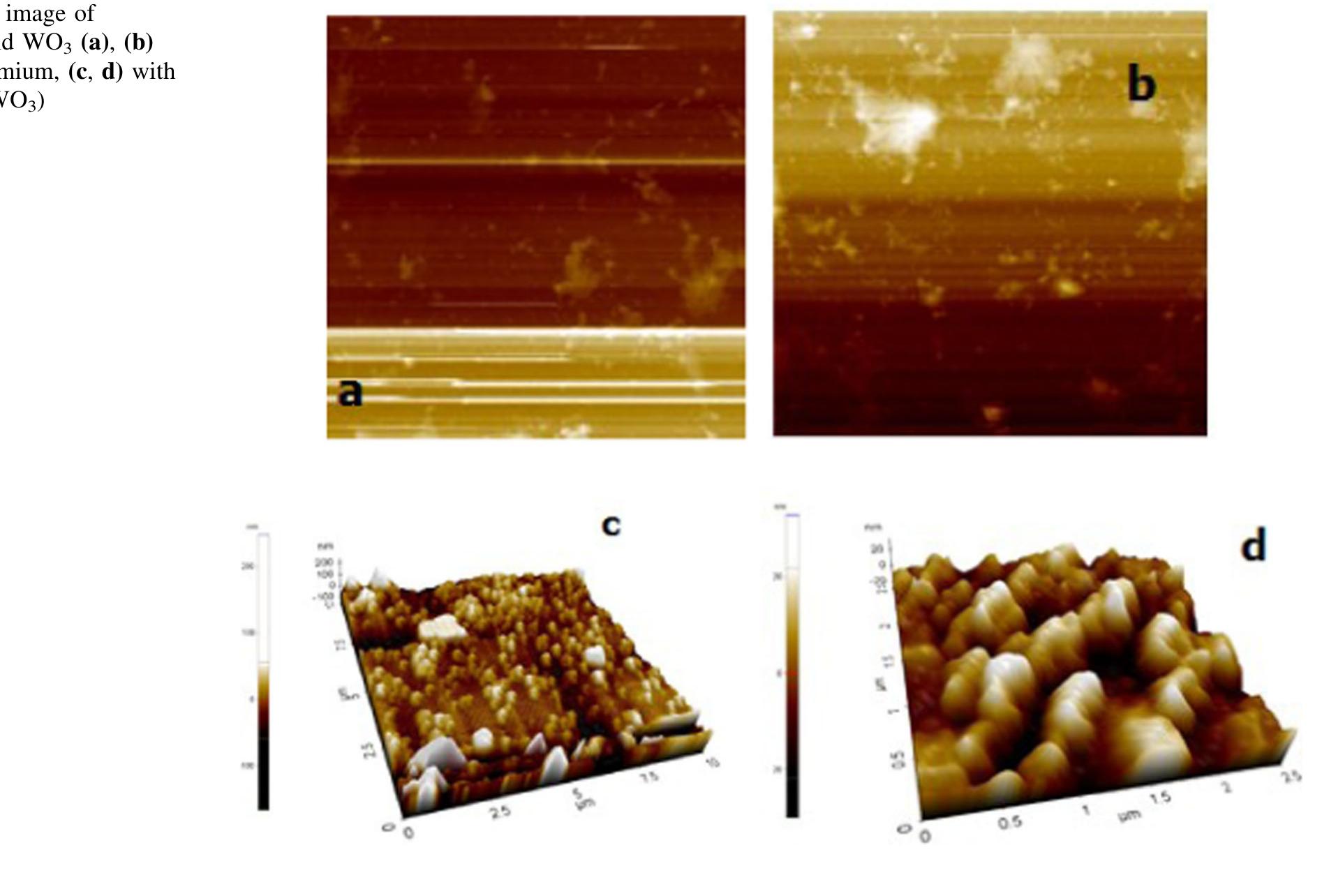
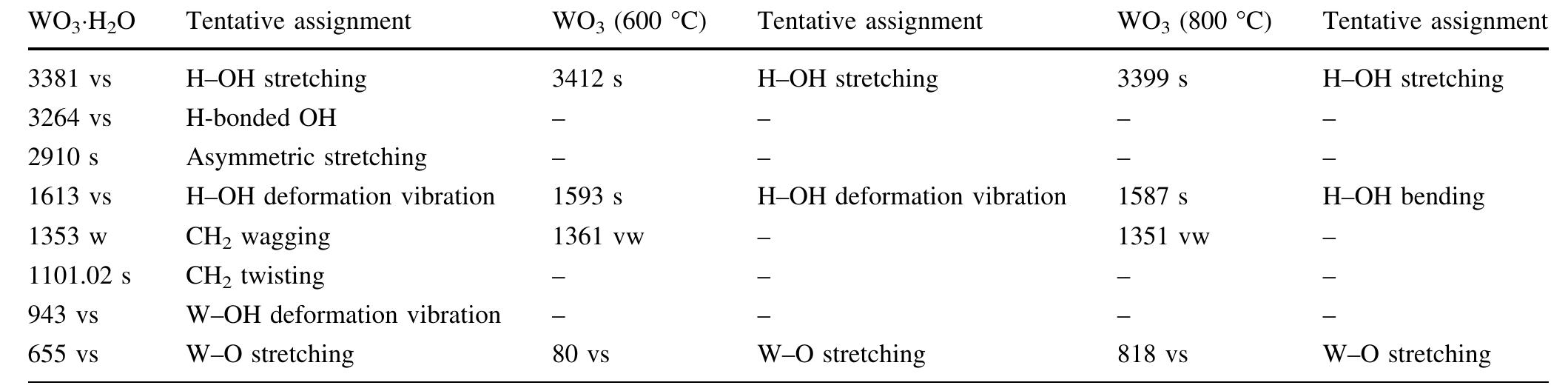
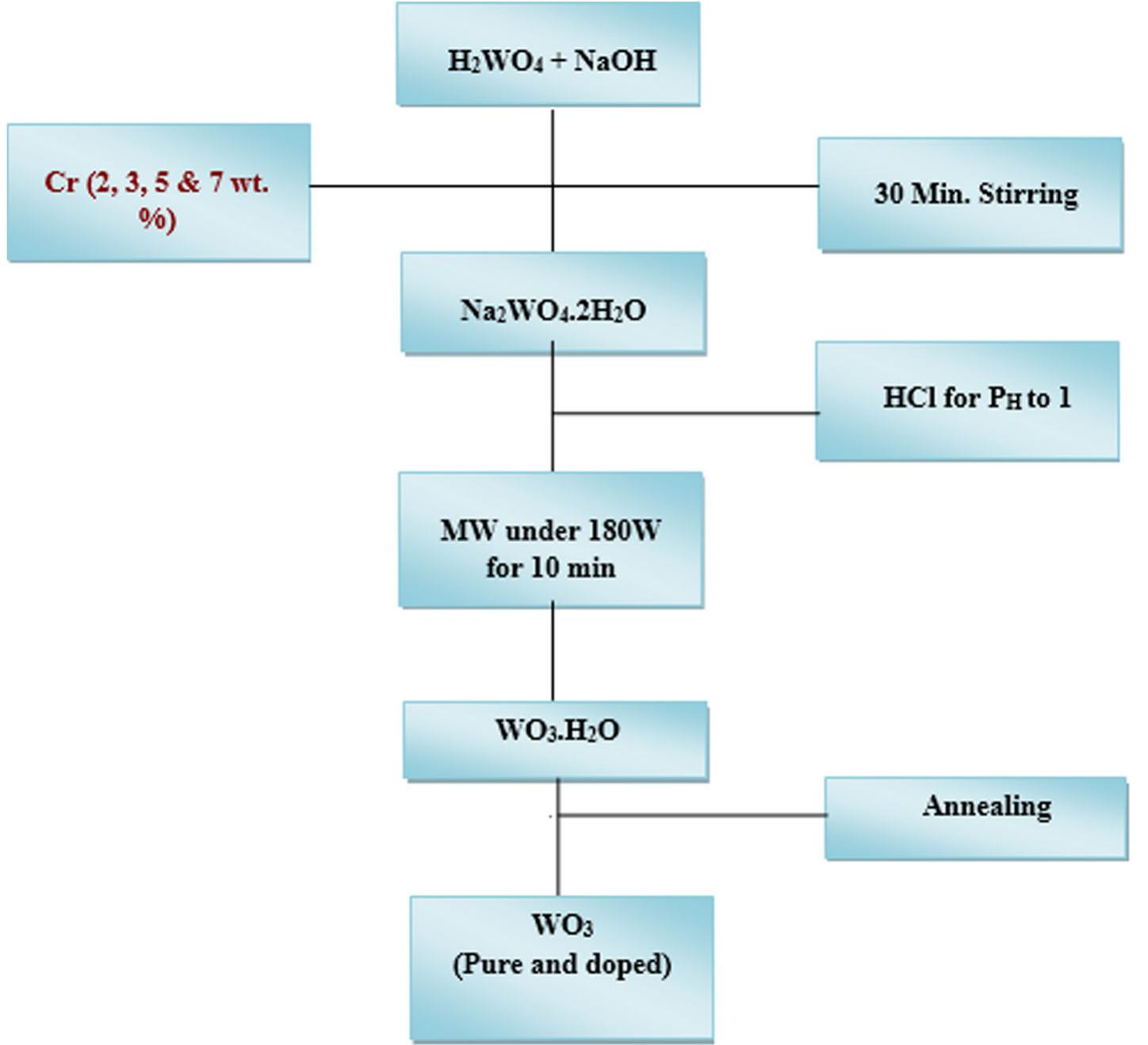
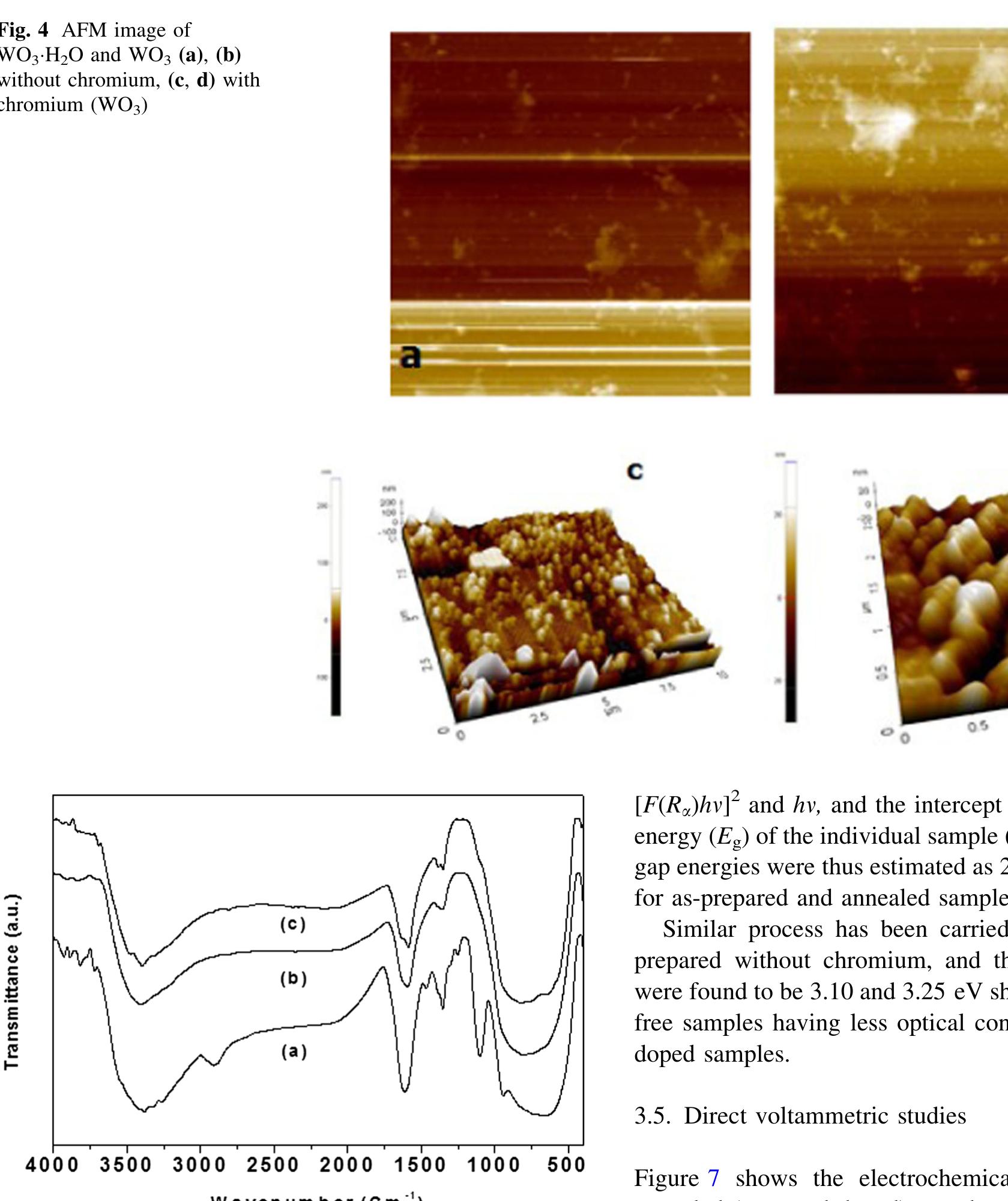
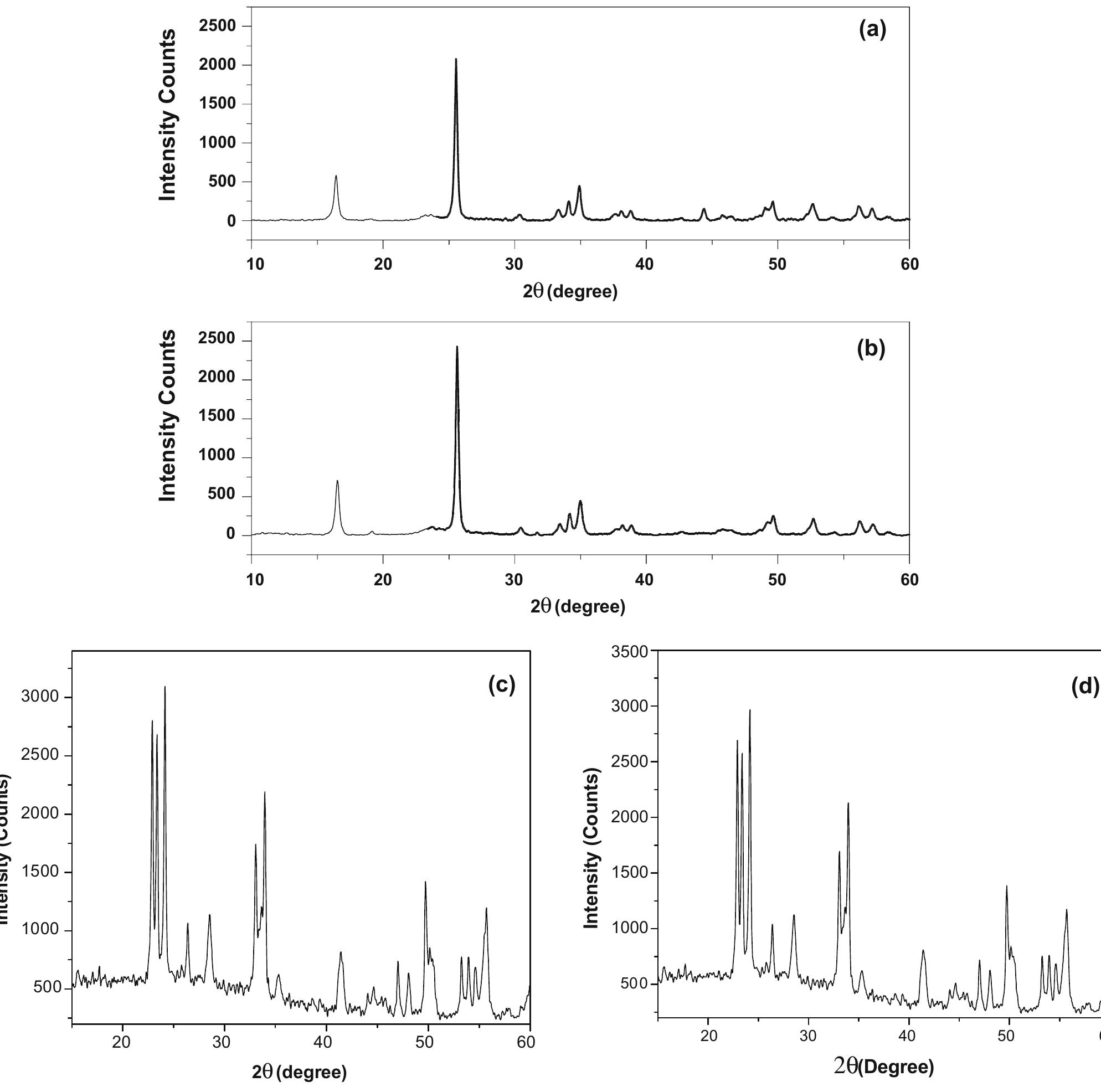
![The trailblazer solution was prepared by dissolving 2.49 g of tungstic acid (H2WO,) in 10 ml of sodium hydroxide (NaOH) with one molar ratio concentration. It results in yellow colored hydrated sodium tungstate solution due to According to reaction (1), high concentrations of hydrated sodium tungstate would shift the reaction to the right ensuring the formation of WO3_,, although many intermediate steps and thus compounds and phases may exist. It is thus the formation of WO; should have dependence on the acid medium. To convert WO, to neutral WO3, excess divalent oxygen must be removed by microwave irradiation method [14].](https://figures.academia-assets.com/89808539/figure_001.jpg)
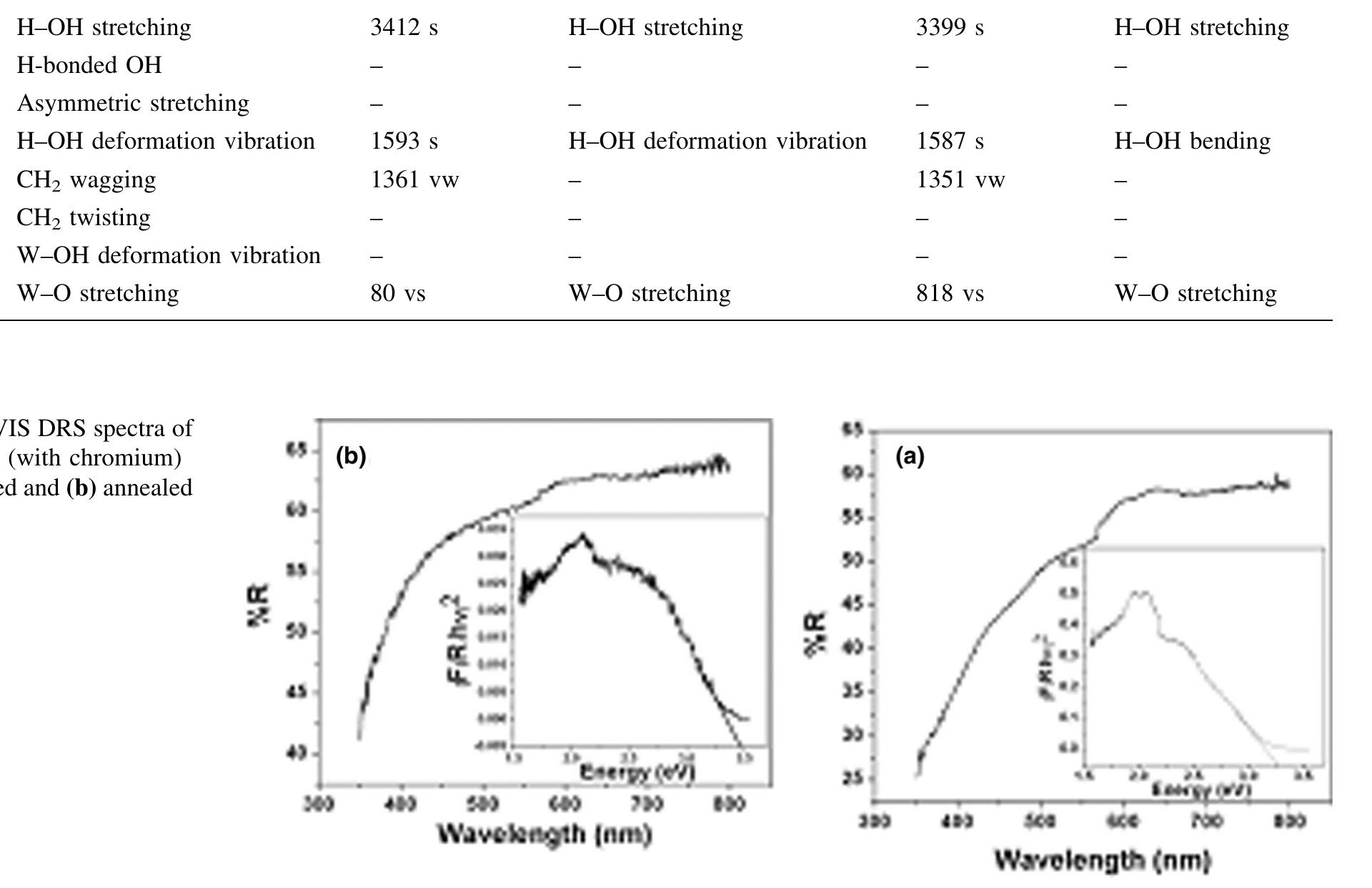
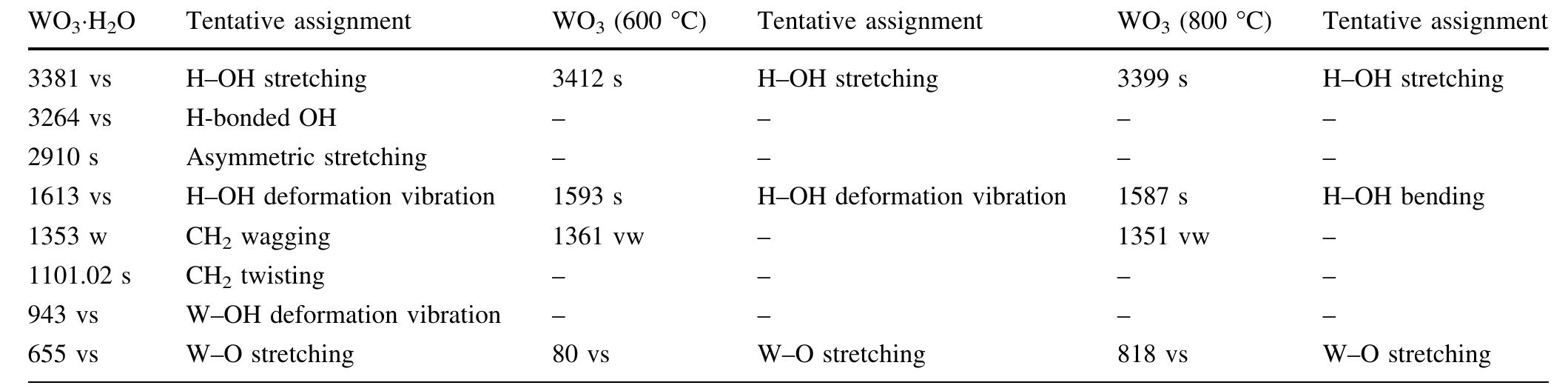

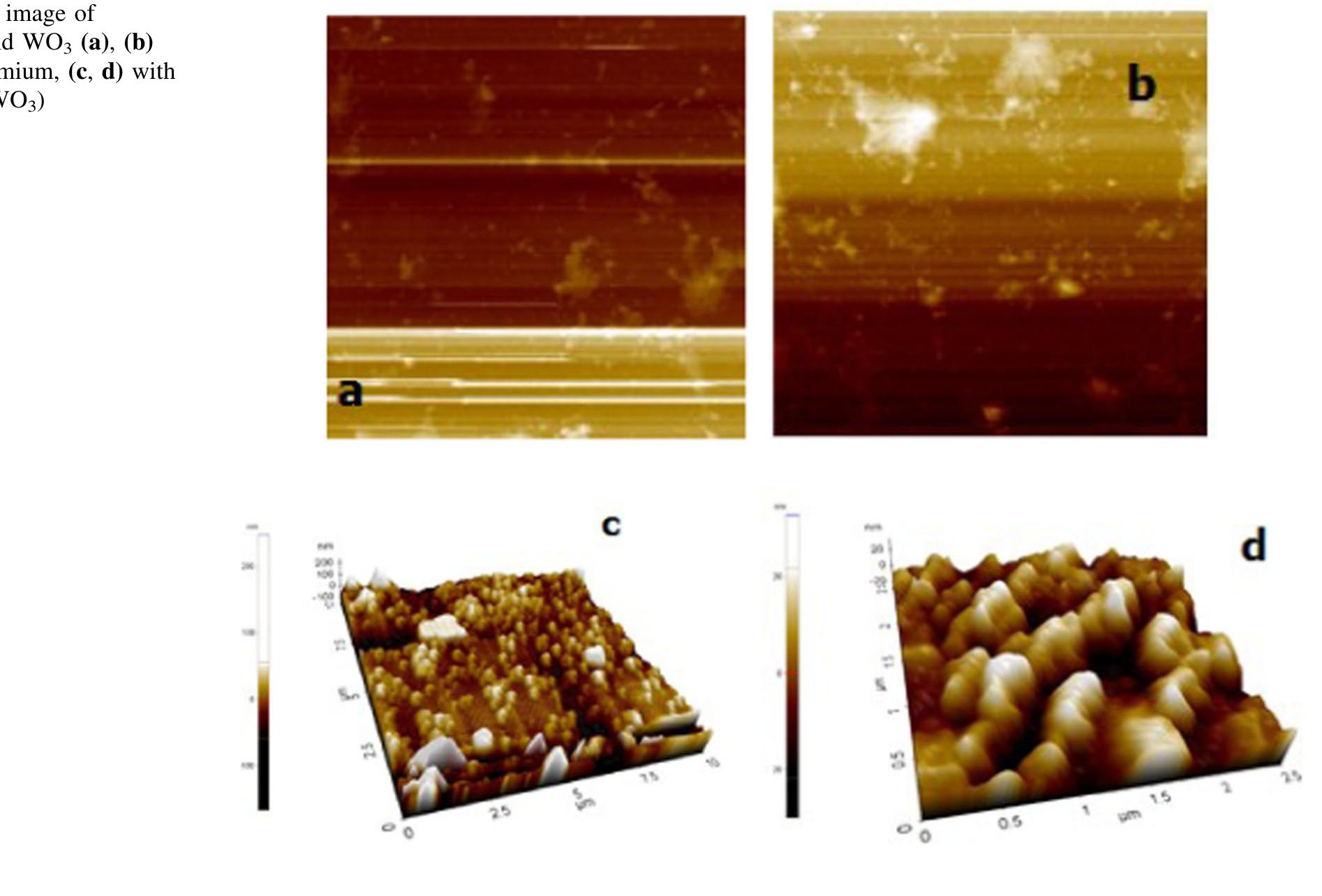
![The electrochromic performance of WO; is closely related to its crystalline, and by increasing the porosity and precisely controlling the crystal size, crystalline WO3 films with both good electrochromic stability and fast response can be achieved [3].](https://figures.academia-assets.com/88213827/figure_001.jpg)
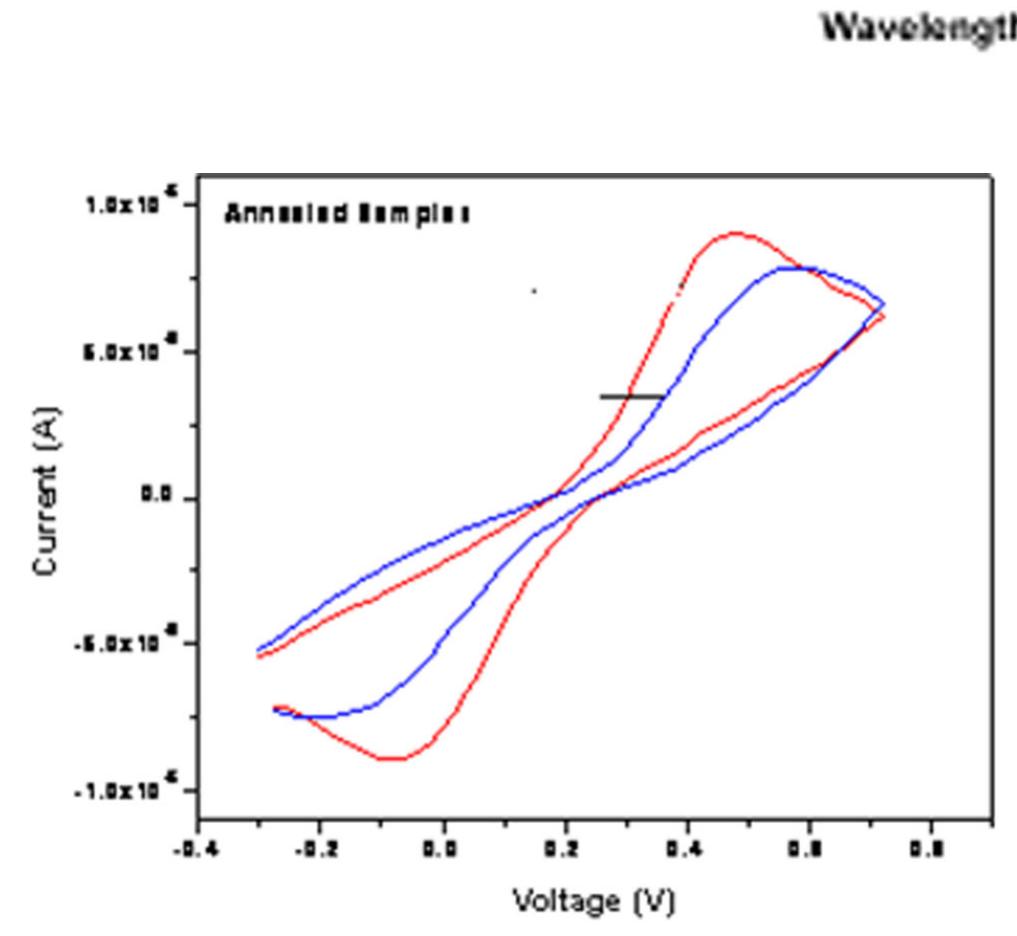
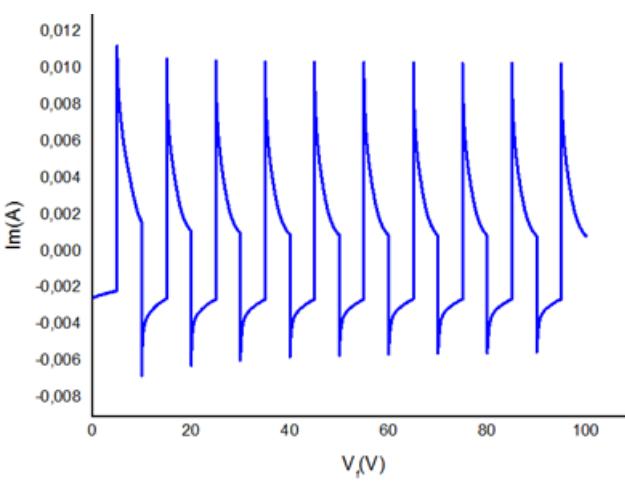
![The diffusion constant for the Li* ions was calculated as 4.2x10°!° cm?/s using the Randles-Servcik equation, for the intercalation process. The high diffusion coefficient for intercalation of Li* ions into WO; was in the range of 1.5x10"!?< D (Lit) < 5x10° cm/s; additionally, the diffusion coefficient was found to be in good agreement with the diffusion coefficient calculated by hydrothermal and CBD methods [25—27].The threshold voltage (Et) is indicated in Figure 3, which corresponds to the rapid surge of Li* ionic intercalation into the WOs; structures [26].](https://figures.academia-assets.com/88213827/figure_003.jpg)






![The o/w nanostructured fluid EAPC has been fully characterized in previous studies. The system’: water con SDS and 1 ent is ca. 73%, the rest being solvents (ethyl acetate, propylene carbonate), surfactan -pentanol (as a co-surfactant). A Small-Angle Neutron Scattering Study recently detailec the structure of this system, showing that EAPC is neither a “classical” microemulsion (i.e. where the solven simple mi s are only found as confined in nano-sized droplets dispersed in the aqueous phase) nor < cellar solution. In ] between t versatility surface of Three formulations o EAPC, both ethyl acetate and propylene carbonate are partitionec he continuous phase (water) and the dispersed droplets, which also accounts for the and the effectiveness of this fluid in removing different types of coatings from the works of art [22]. f semi-IPN pHEMA/PVP chemical hydrogels have also been characterized it the literatire 119 12] The farmiilatinne differ in the mnannmer/cerncdinker and nUERMUMA/PVUP rating](https://figures.academia-assets.com/67289317/figure_002.jpg)
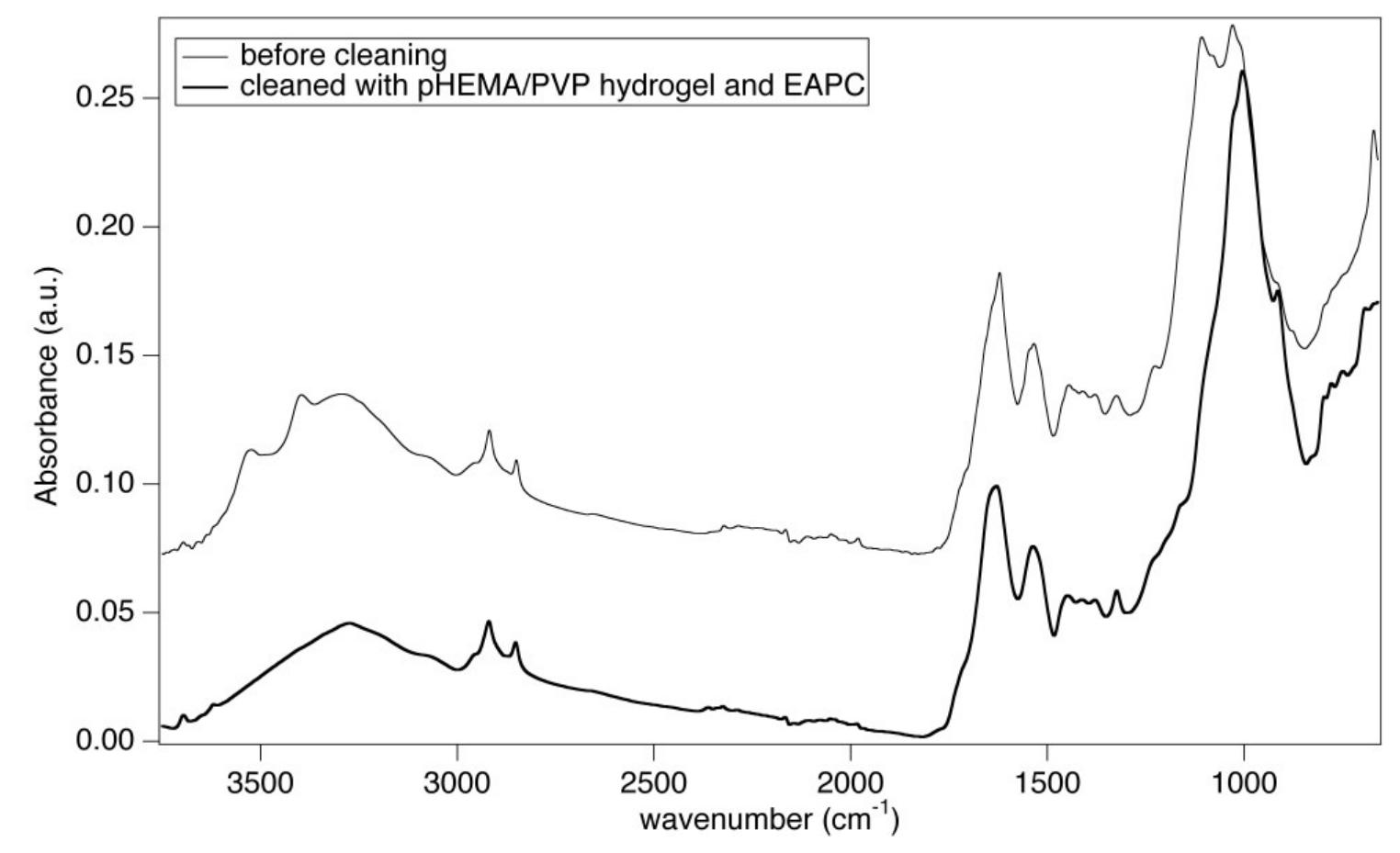
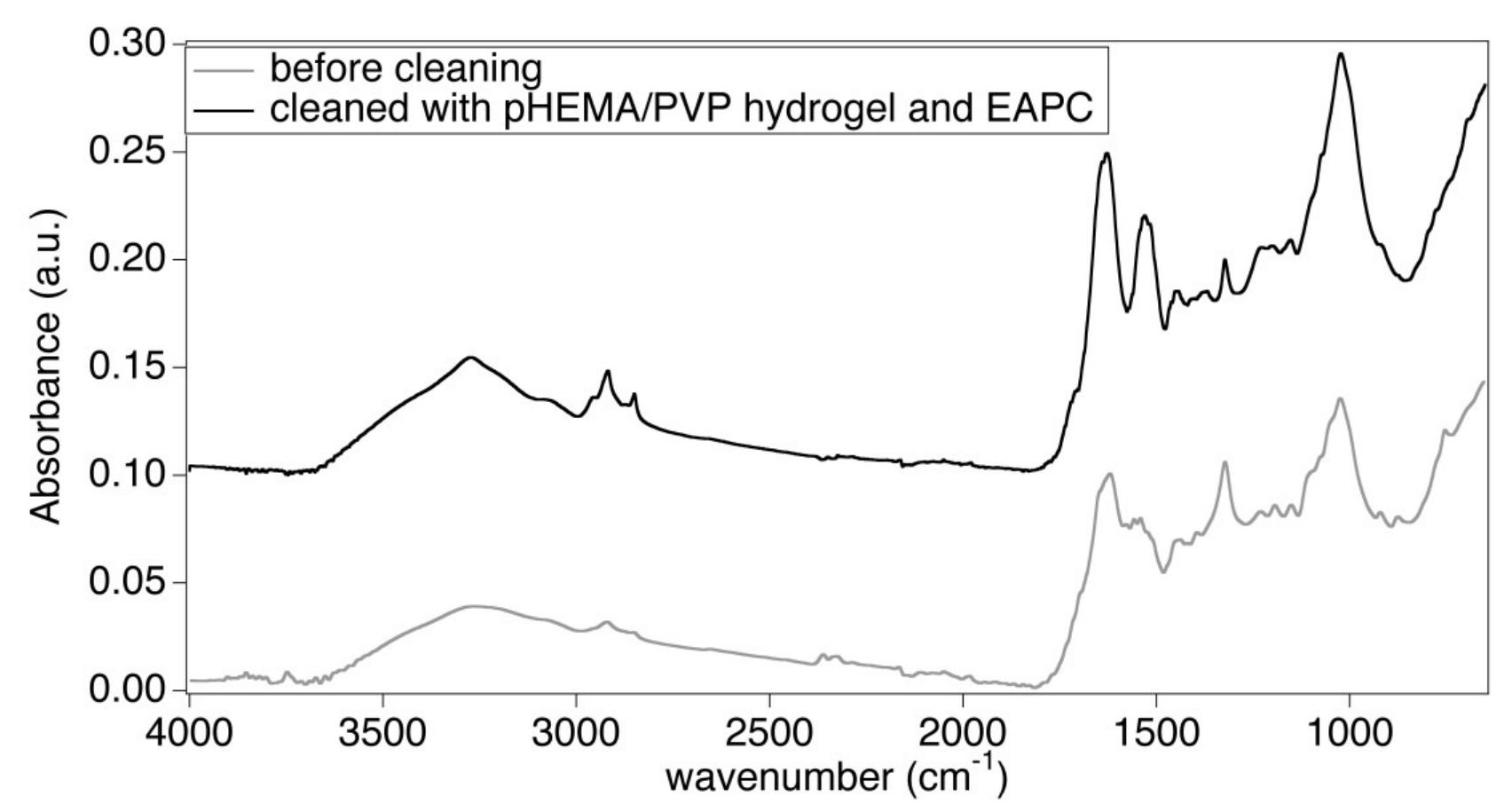
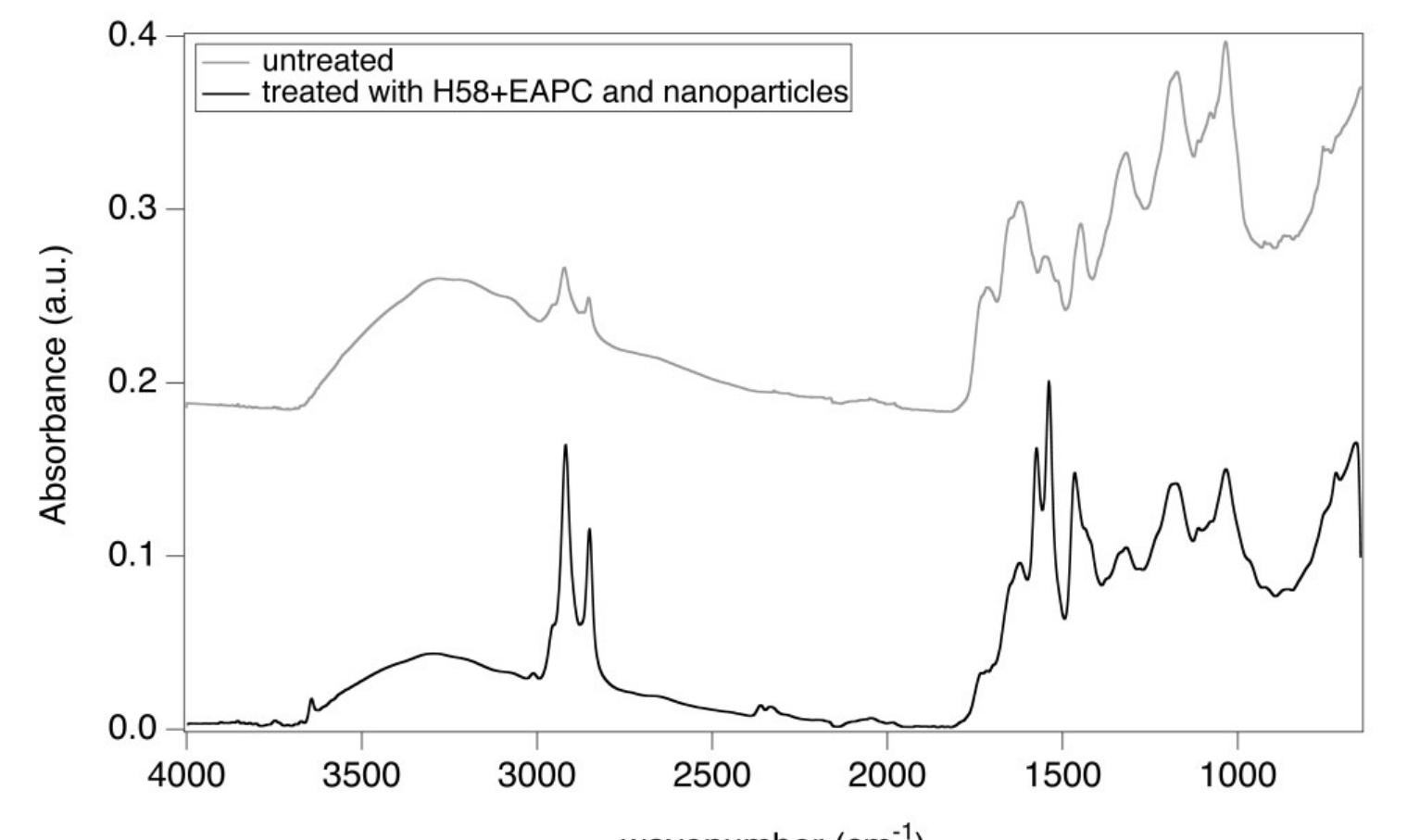
![Figure 6 H1 sample (Luther Bible cover, 1749 AD) before (bottom) and after (top) treatment with the pHEMA/PVP HS58 gel (loaded with |] EAPC) and a mixed calcium hydroxide — calcium lactate nanoparticles dispersion in 2-propanol. The area on the bottom shows the dull grayish surface patina of dirt, salts and pollutants. The area on the top shows the removal of the patina to reveal the underlying heterogeneous leather surface important to notice that the surface patina must be removed prior to application of the nanoparticles dispersion to favor the penetration of particles and avoid the formation of white veils. The ATR-FTIR spectrum of the sample before treatment (Figure 8) clearly shows the absorption bands of gypsum at 3525, 3398 cm” (vOH), 1619 cm”! (8OH), 1111 cm’! (asymmetric stretching vibrations of SO,”) and 669 cm” (symmetric bending of SO4”) [31,32]. These absorptions are no longer observable in the spectrum of the sample after treatment with the gel, while the bands of collagen and tannins can be clearly observed, which confirmed the effective removal of the surface patina. Also in this case, no bands ascribable to SDS (1084 em”, vs8O2; 1227 cm”! Vas9O2; 1471 cm’, CH) or to the H58 gel are observable in the spectrum.](https://figures.academia-assets.com/67289317/figure_006.jpg)