Key research themes
1. How does ion association, aggregation, and solvation influence the thermodynamic and transport properties of mixed electrolyte solutions?
This research area investigates the microscopic and thermodynamic mechanisms by which specific ions in mixed electrolyte solutions affect properties such as density, viscosity, conductivity, and phase behavior. Understanding ion pairing, aggregation beyond simple pairs, and ion solvation effects is critical for predicting electrolyte behavior in concentrated regimes relevant to industrial and biological systems.
2. What are the effects and optimization strategies of electrodialysis and bipolar membrane technologies for processing mixed electrolyte or brine solutions?
This theme centers on the application of electrodialysis (ED) and electrodialysis with bipolar membranes (EDBM) to treat complex mixed electrolyte solutions such as brines from desalination, lithium mining, and industrial wastewaters. Research focuses on membrane selection, control of ion transport (including selective ions like Li+, Ca2+), mitigating fouling and scaling from multivalent ions, and valorizing resulting acid/base streams, enabling sustainability and improved resource recovery.
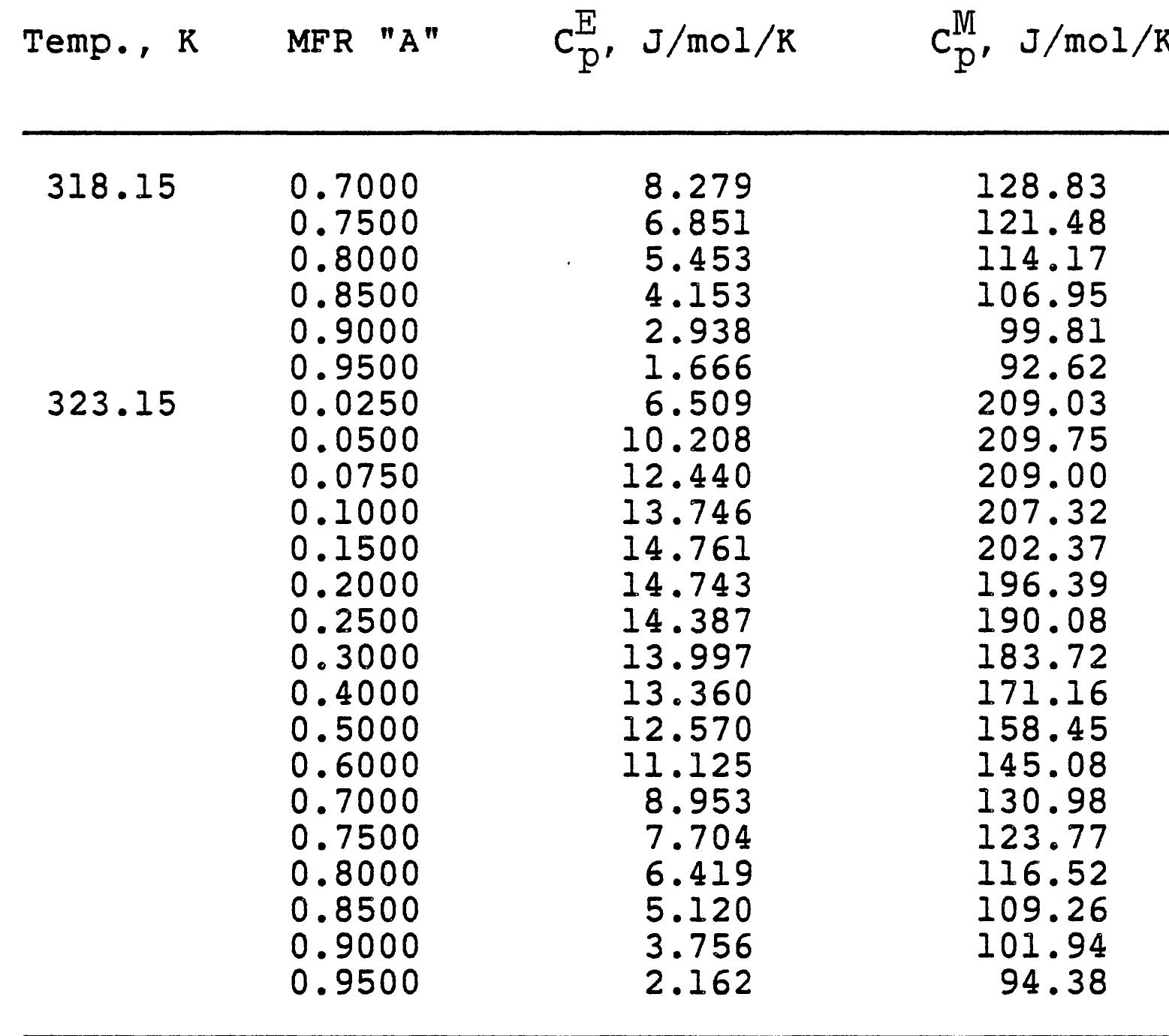
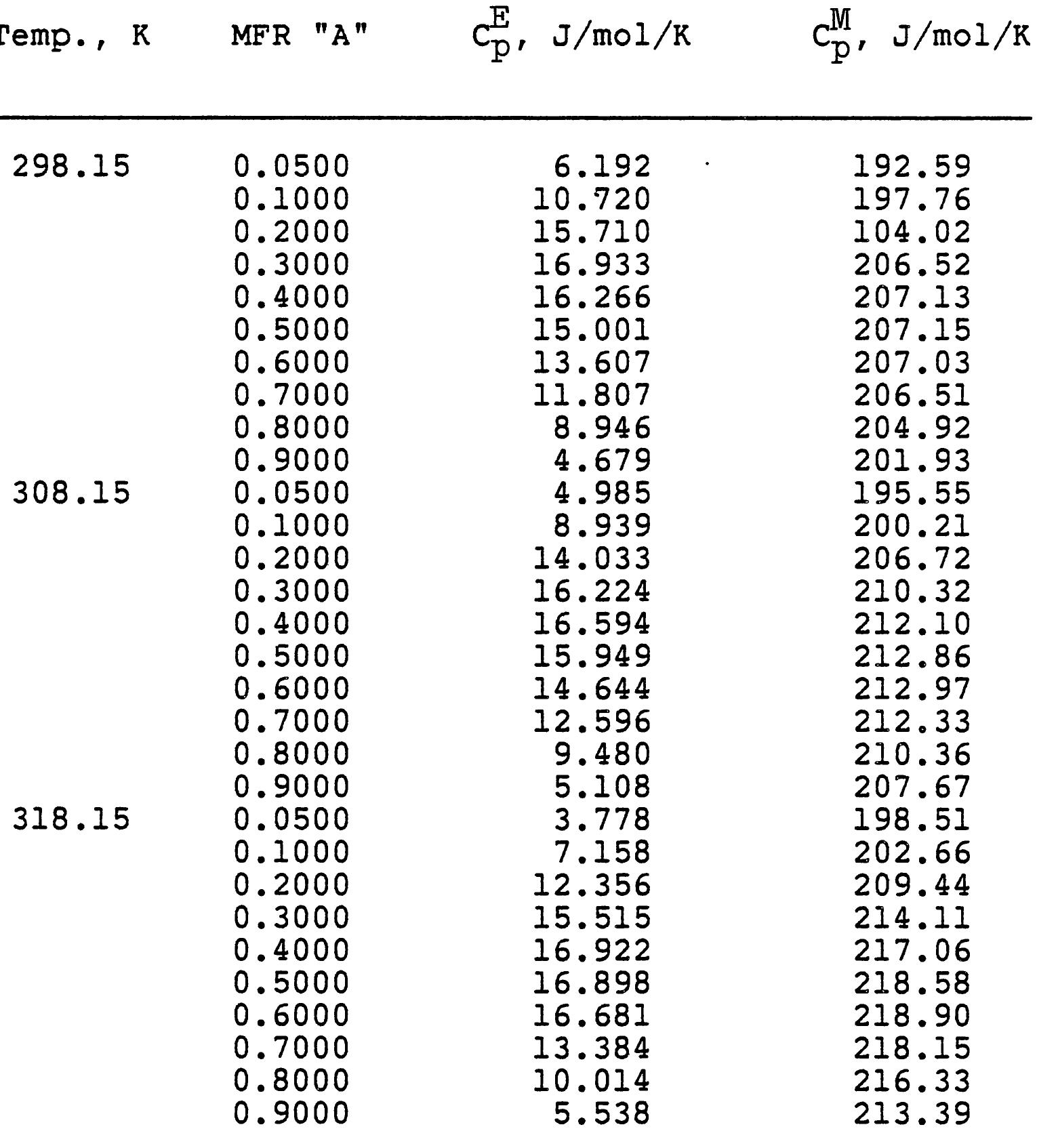
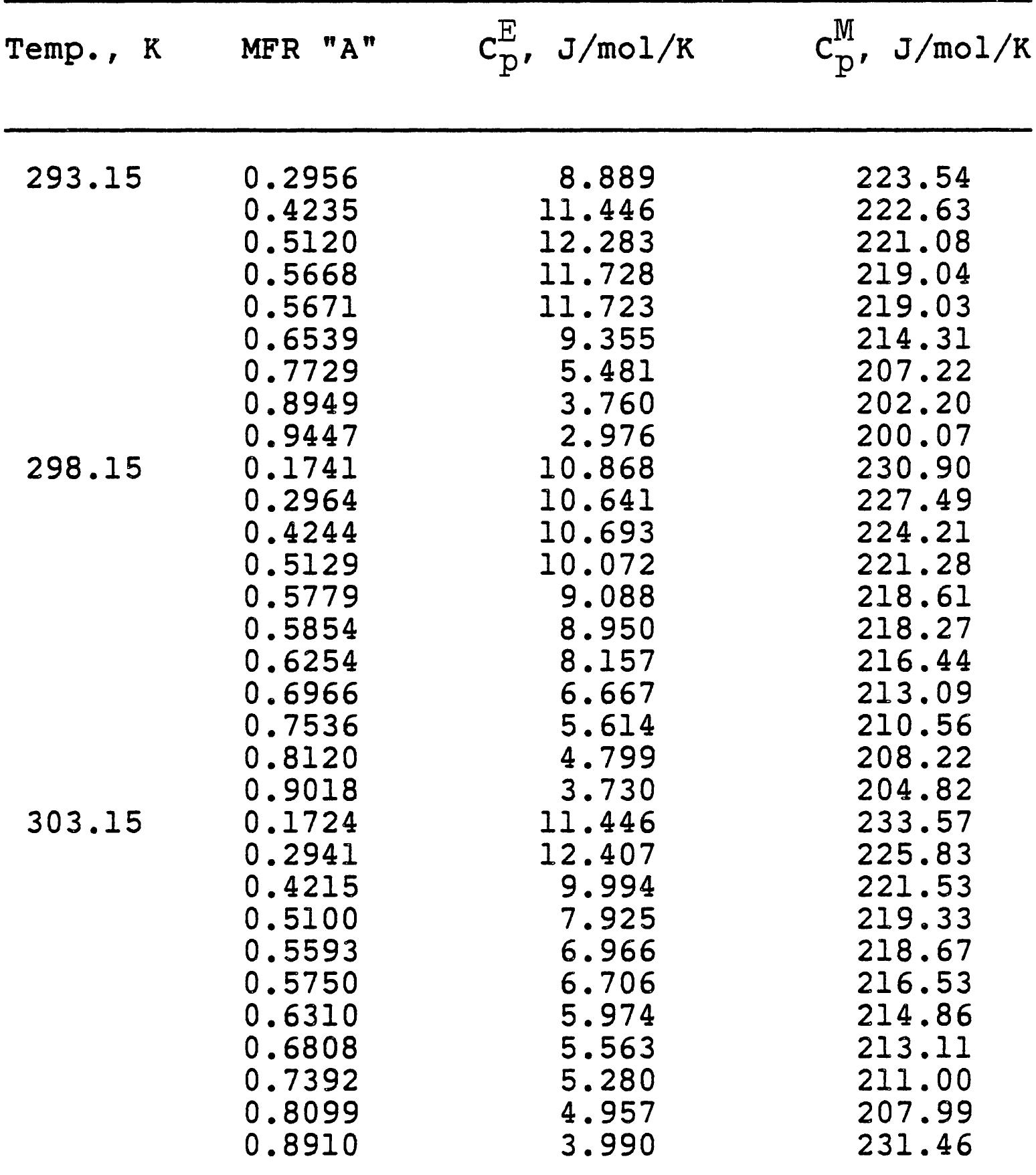
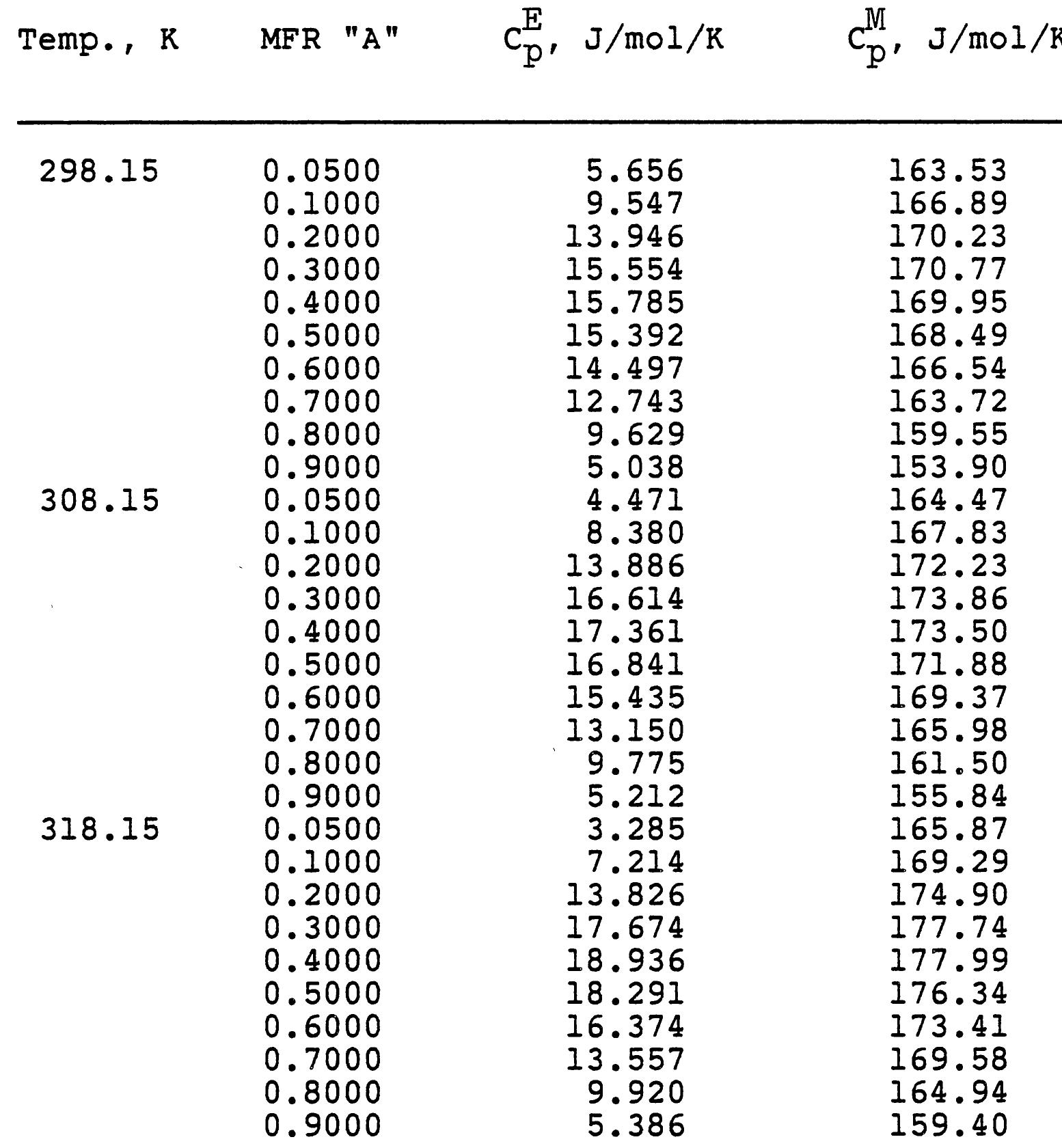
3. How do electrolyte composition and temperature empirically influence physical properties such as electrical conductivity, density, and viscosity in mixed electrolyte or solvent systems?
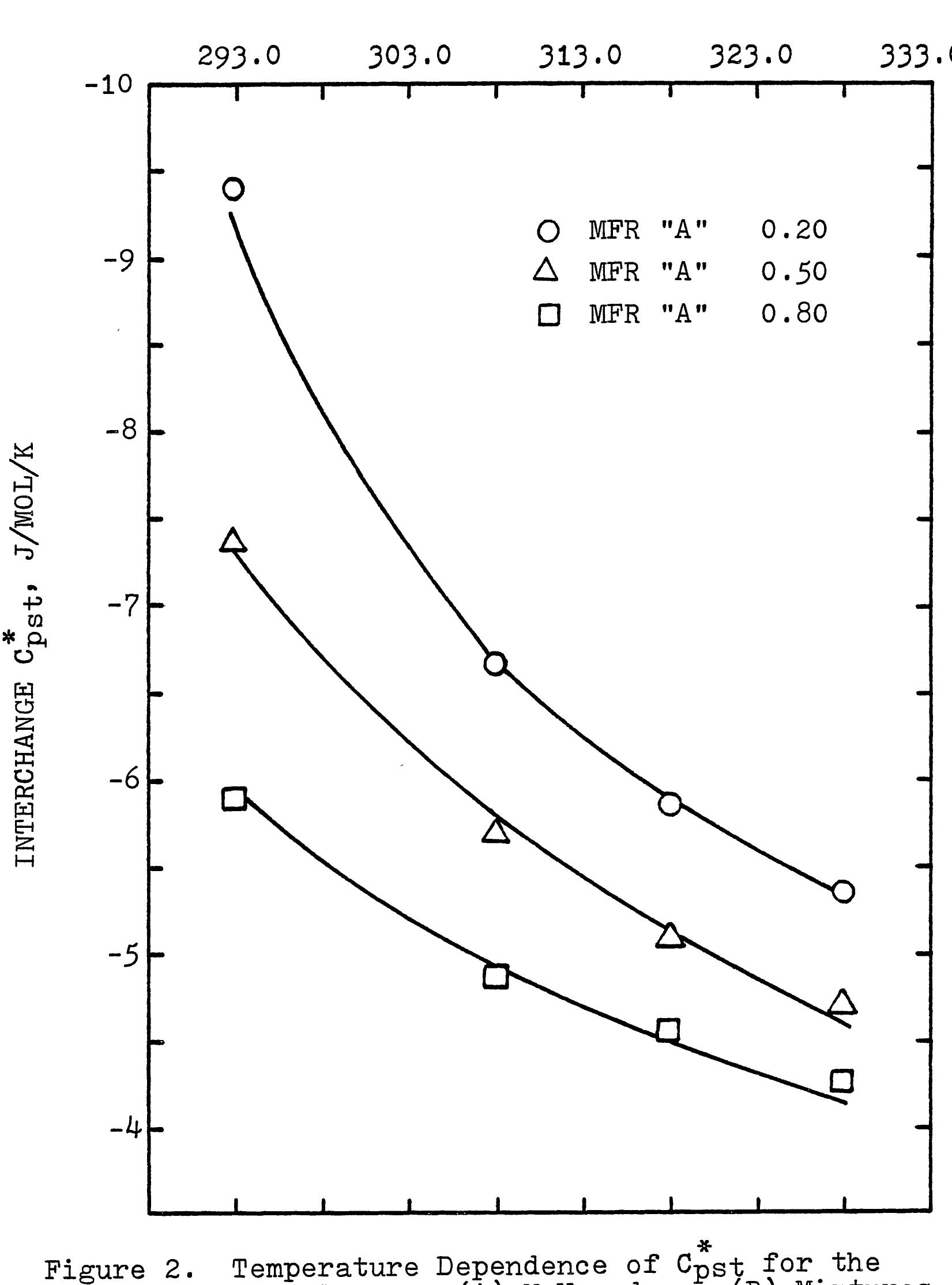
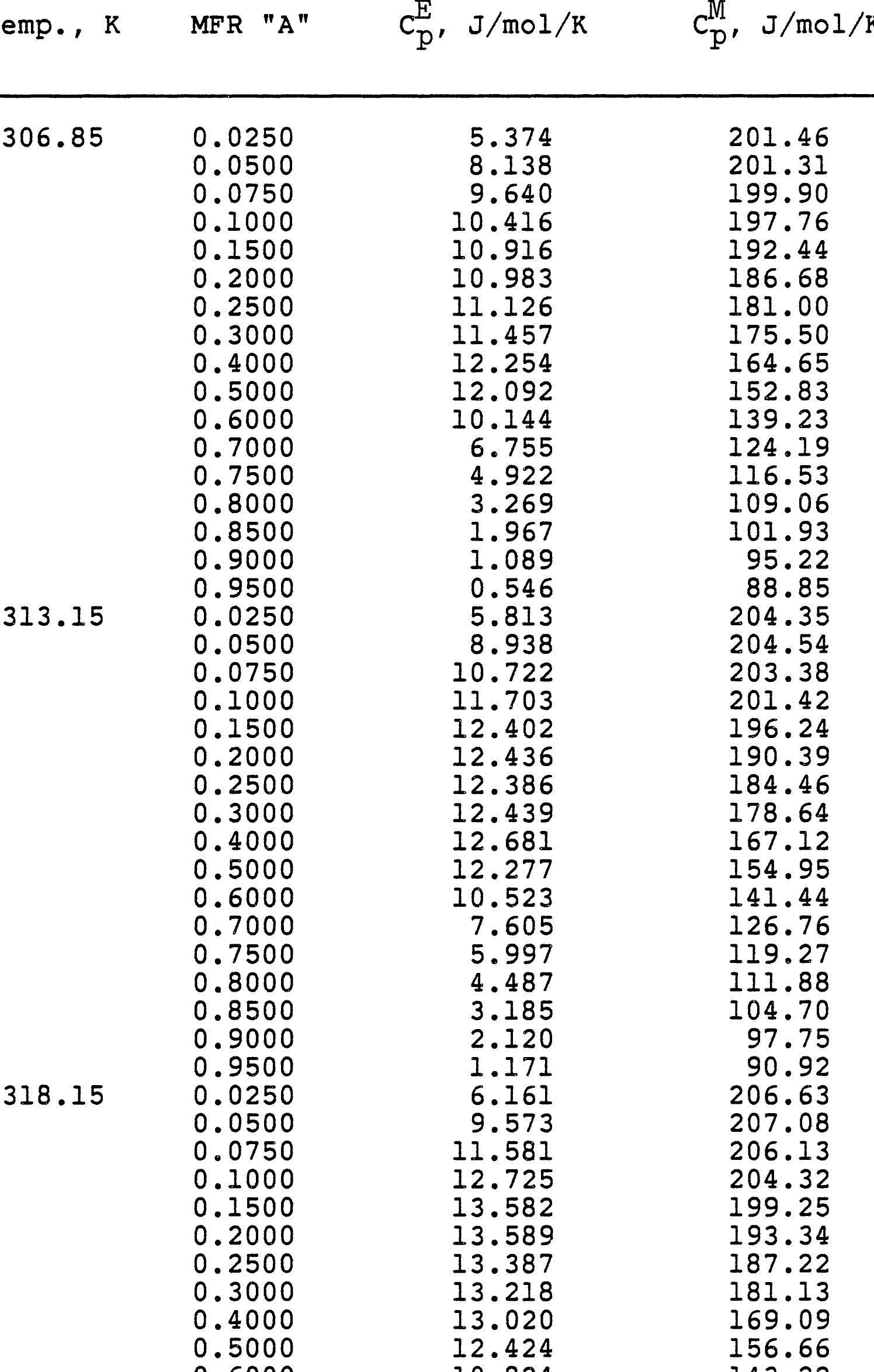
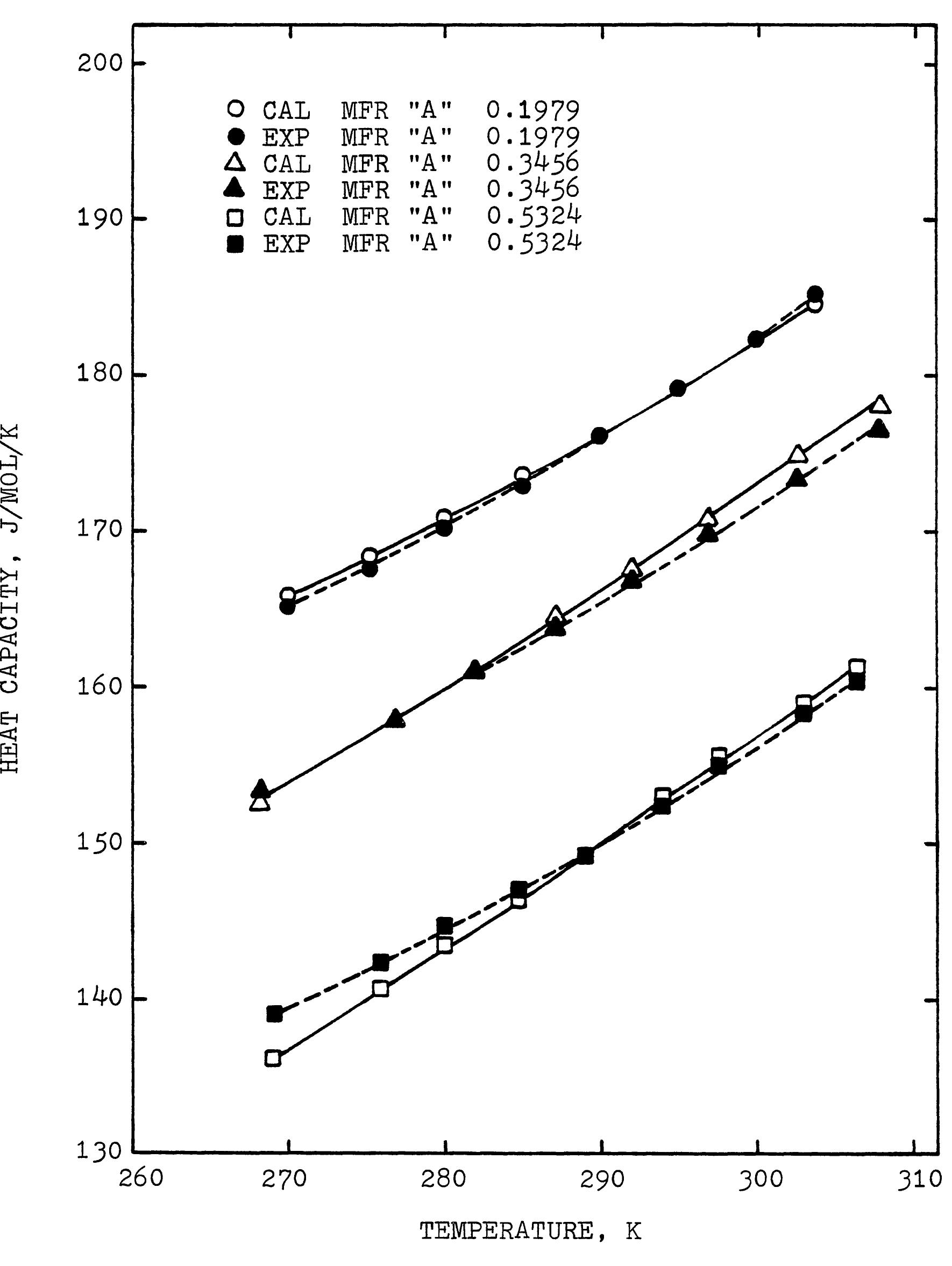
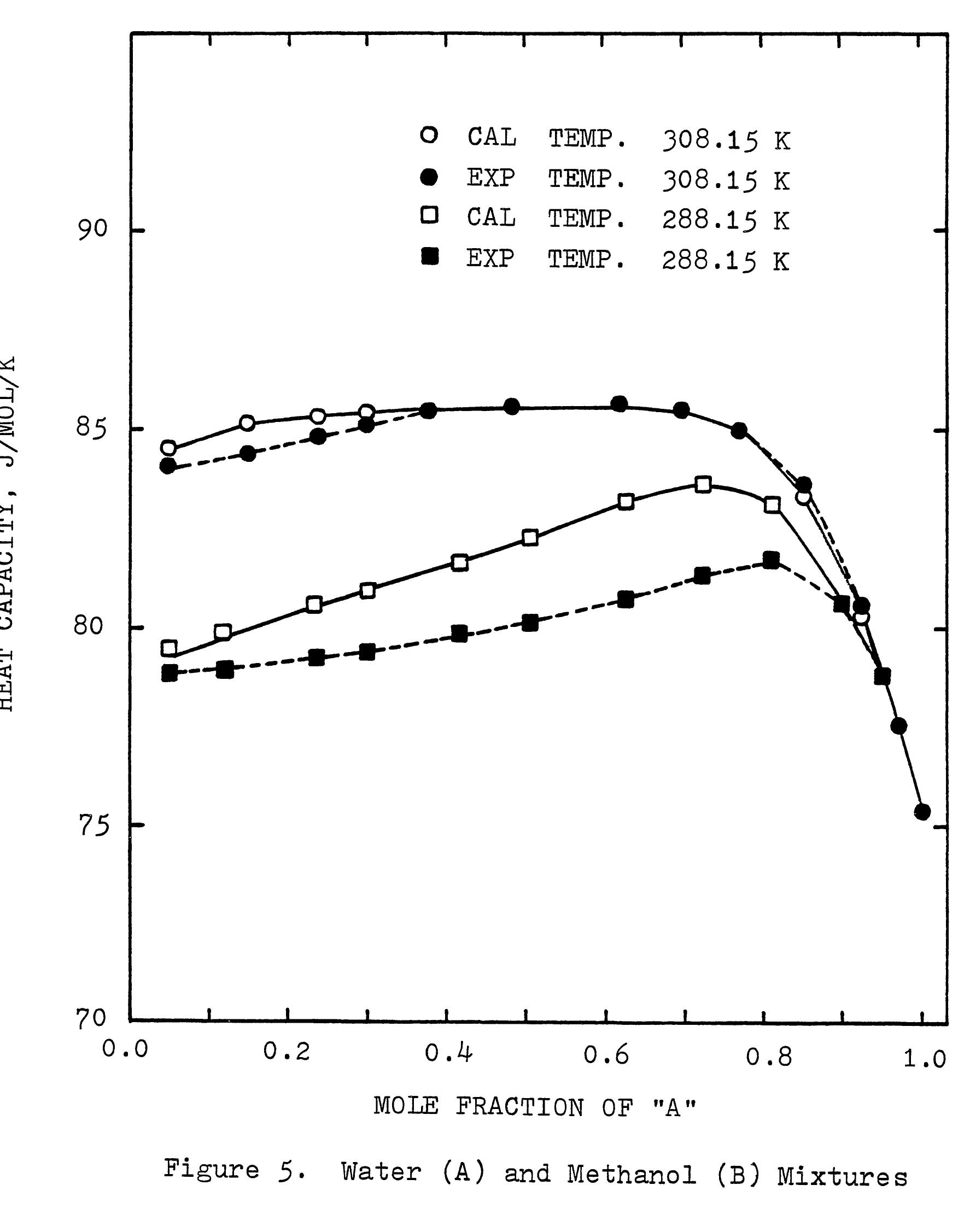
This research theme focuses on establishing empirical and semi-empirical relationships that describe how electrolyte concentration and temperature modulate physical properties critical to electrolyte performance, including electrical conductivity, density, and viscosity. Such relationships are essential for predicting behavior in mixed solvents and high concentration regimes relevant to industrial processes and energy storage.