Reduction of a colloidal suspension of exfoliated graphene oxide sheets in water with hydrazine hydrate results in their aggregation and subsequent formation of a high-surface-area carbon material which consists of thin graphene-based... more
The increasing demand of energy has been a prime source of motivation for the development of alternate fuels. The concept of biological degradation of organic wastes by anaerobic digestion for the generation of methane has been used by... more
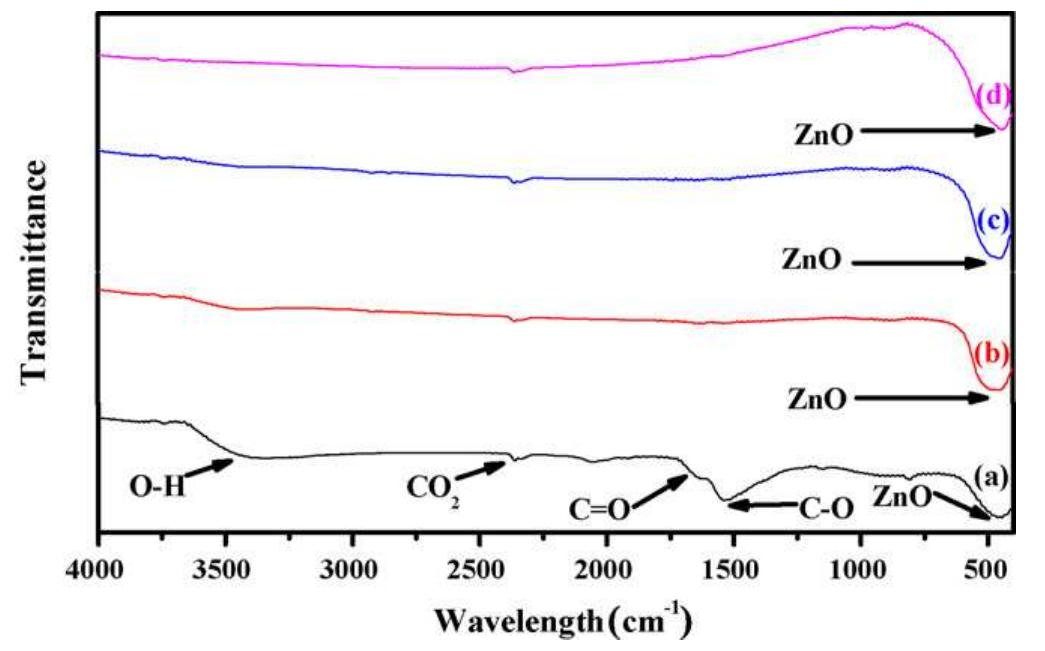
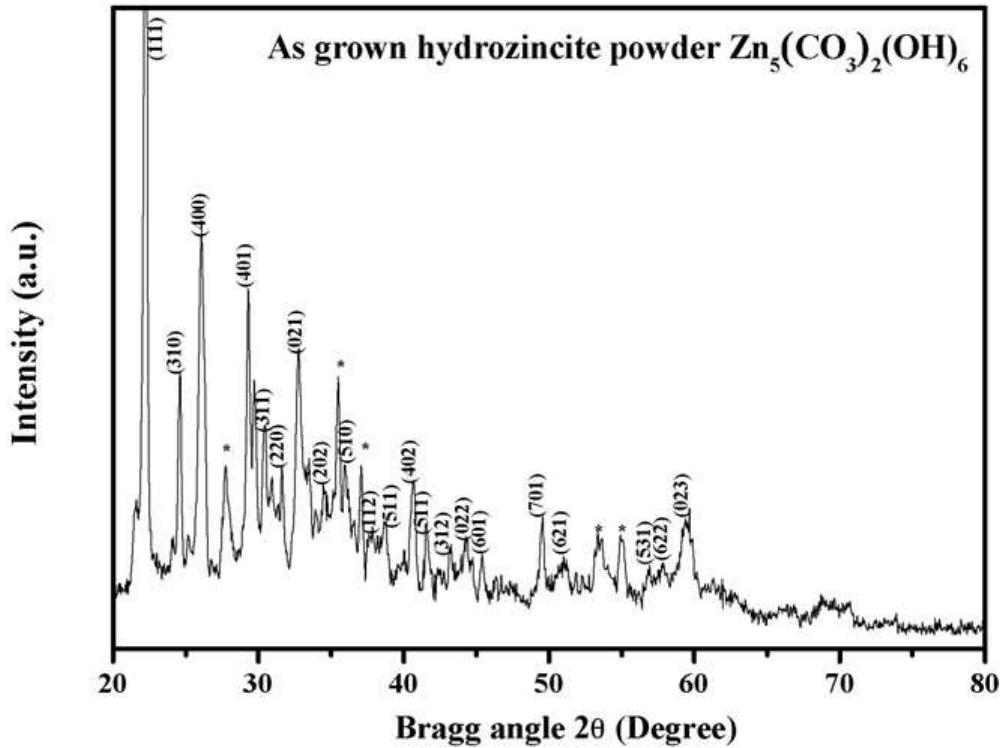
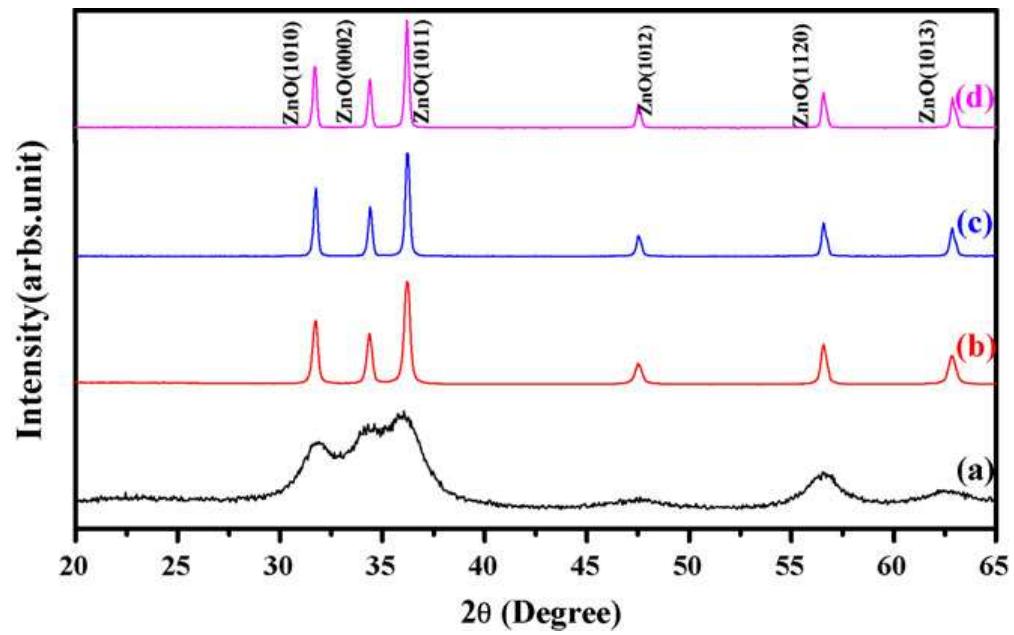
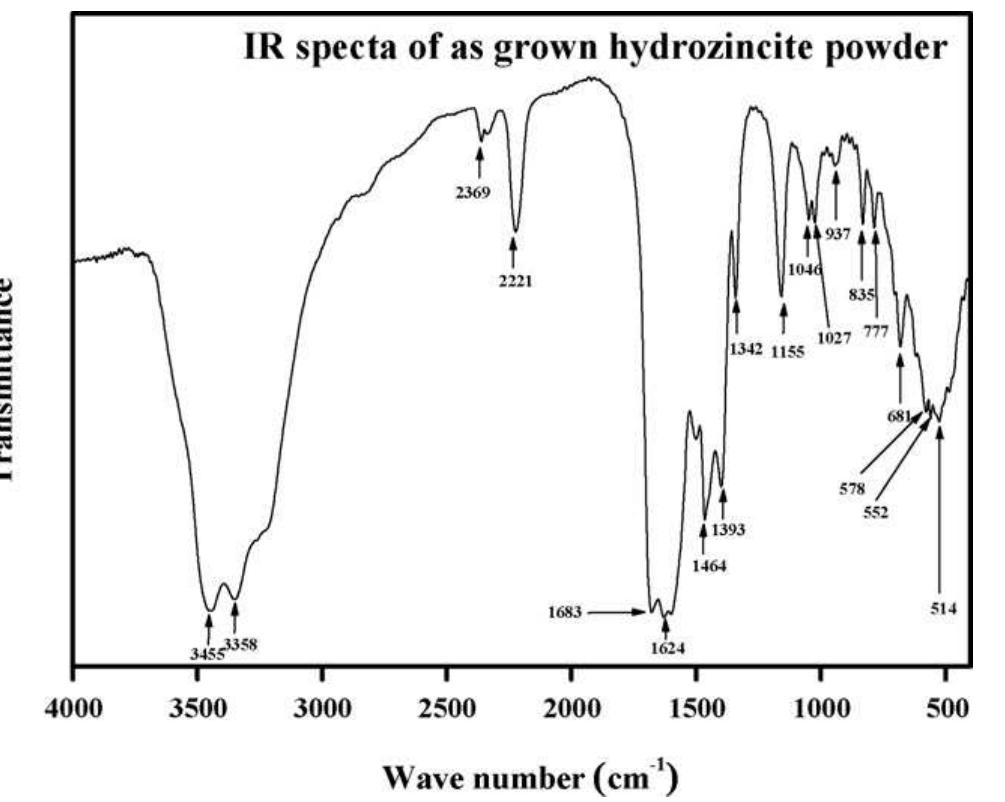
Faceted nanoparticles of zinc oxide were successfully achieved via thermal annealing of hydrozincite (Zn 5 (CO 3 ) 2 (OH) 6 ) powder at different annealing temperatures, i.e. 300, 500 700 and 900 • C in air for 2 h using sol-gel method by... more
Novel macroporous hydrogels were prepared by blending of cellulose and sodium alginate (SA) solution, and then cross-linking with epichlorohydrin. The resulting cellulose/SA hydrogels were characterized by solid-state 13 C NMR, wide-angle... more
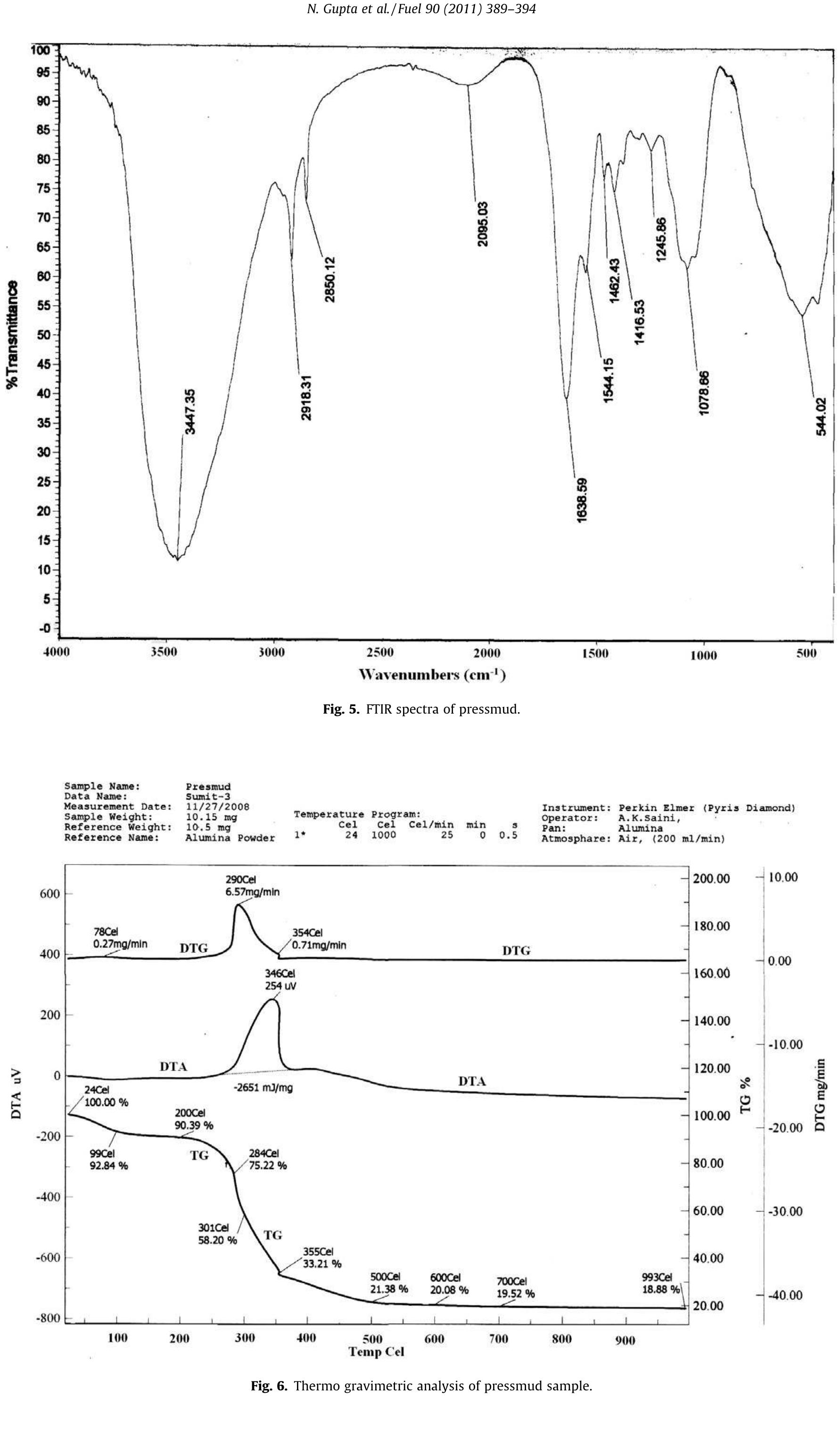
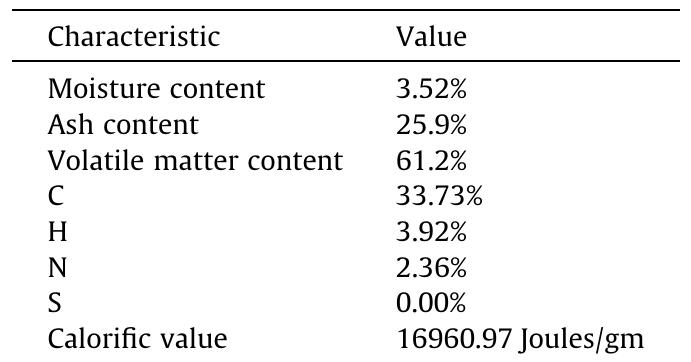
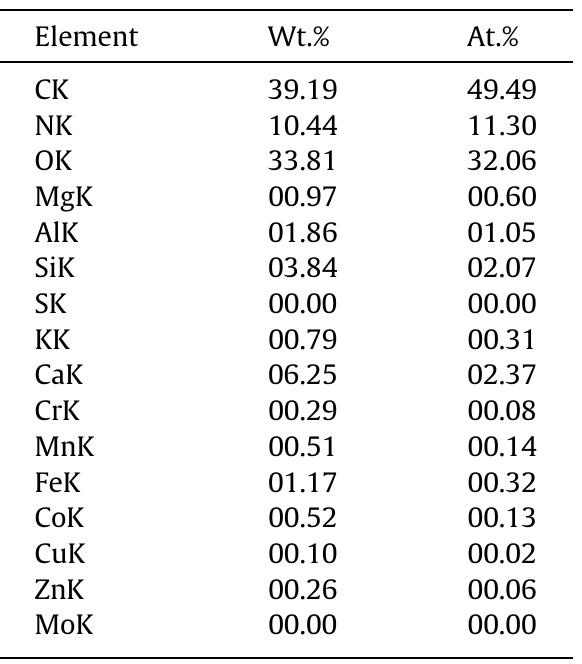
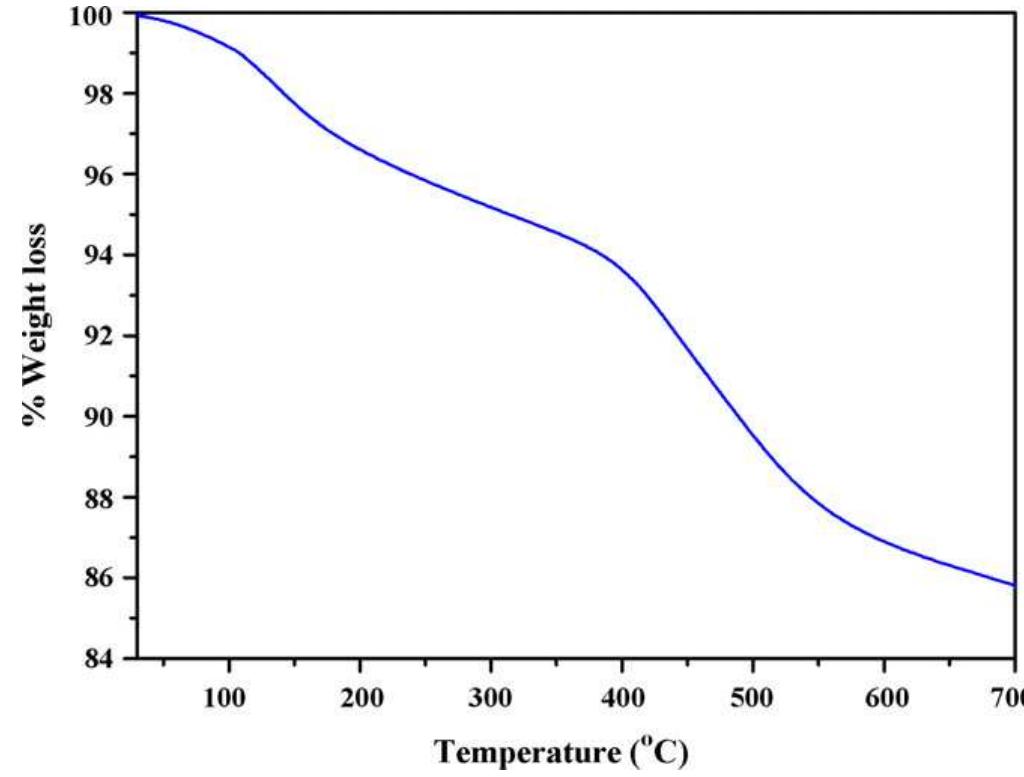
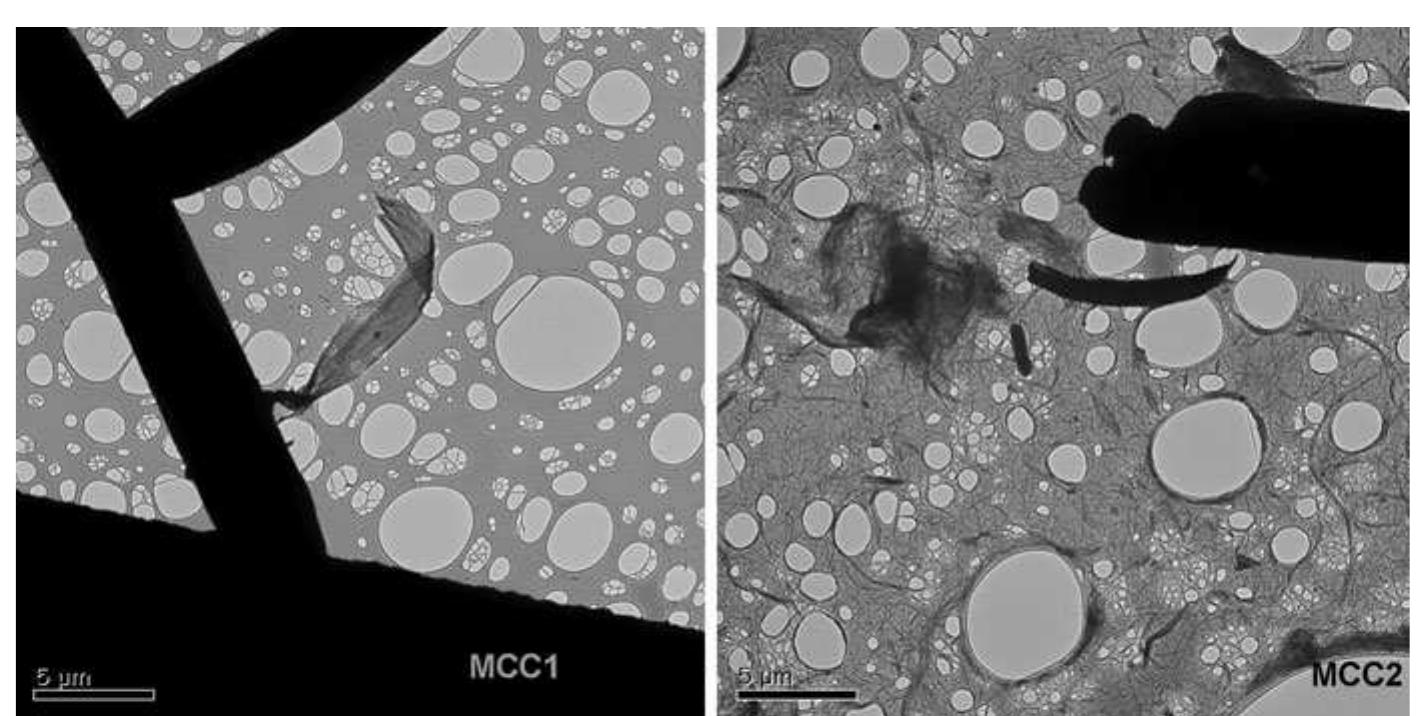
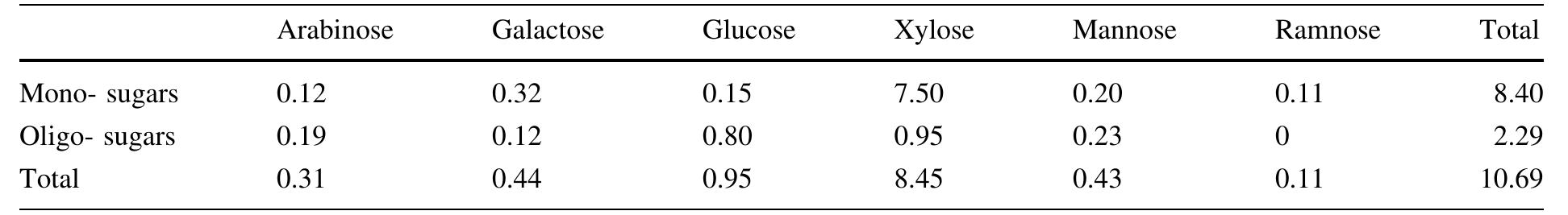
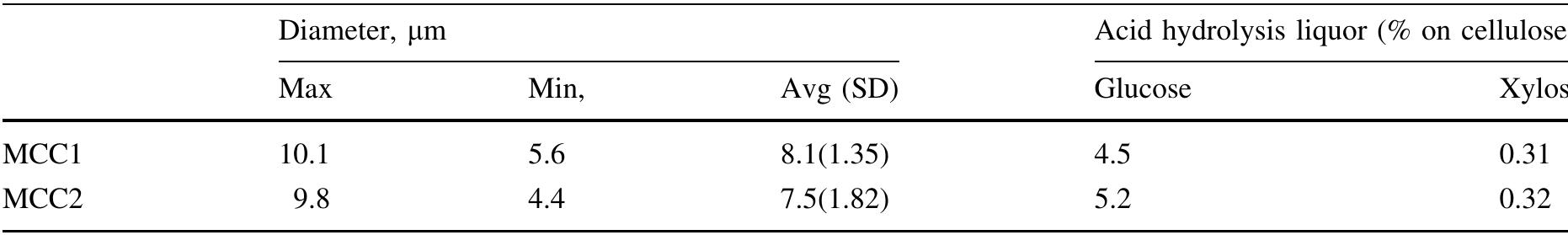
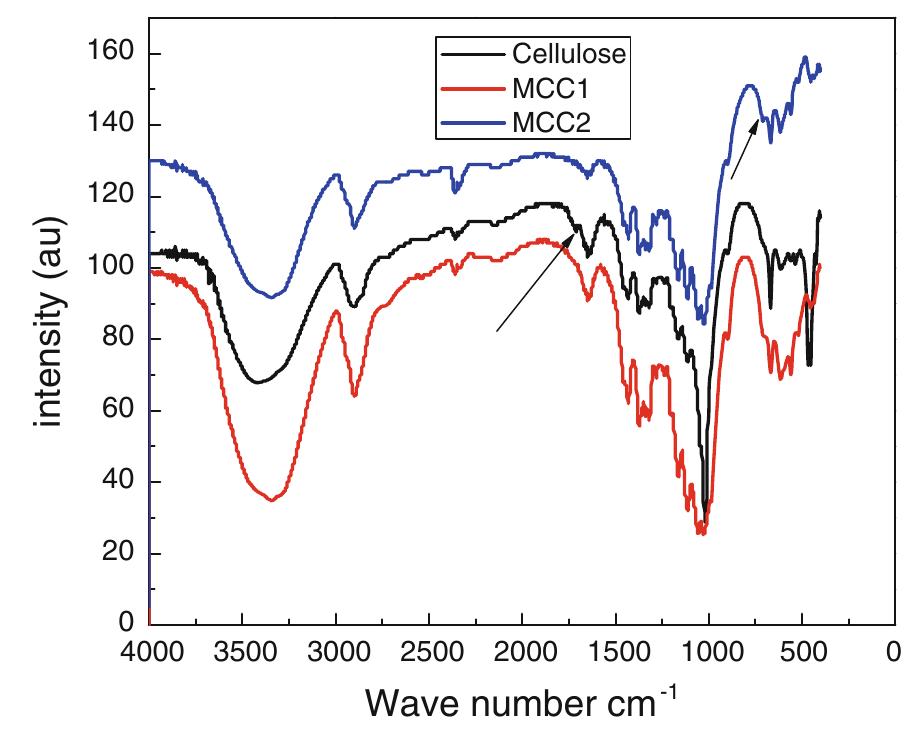
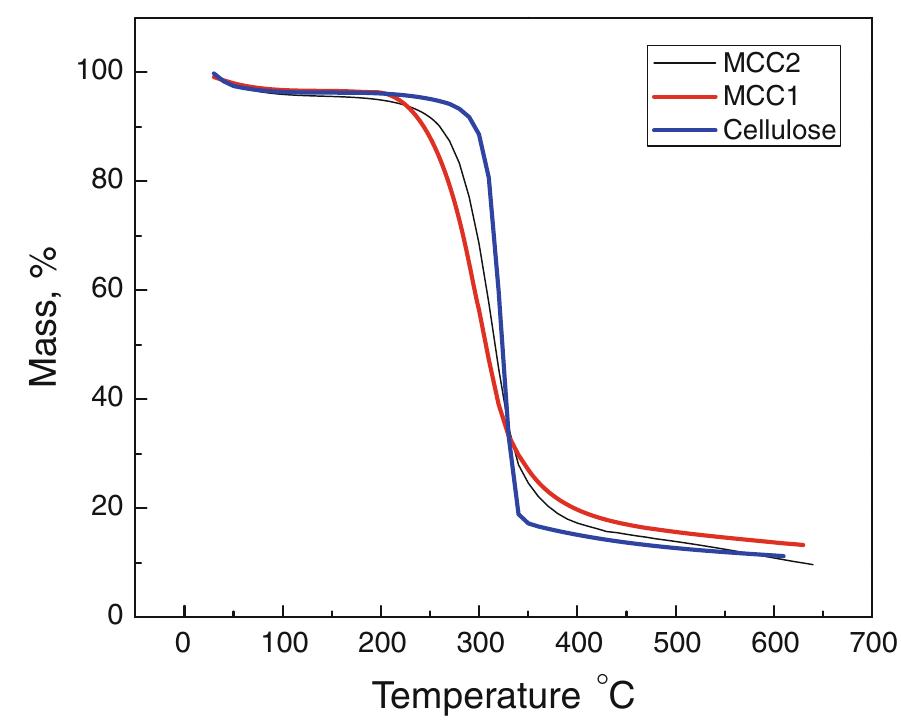
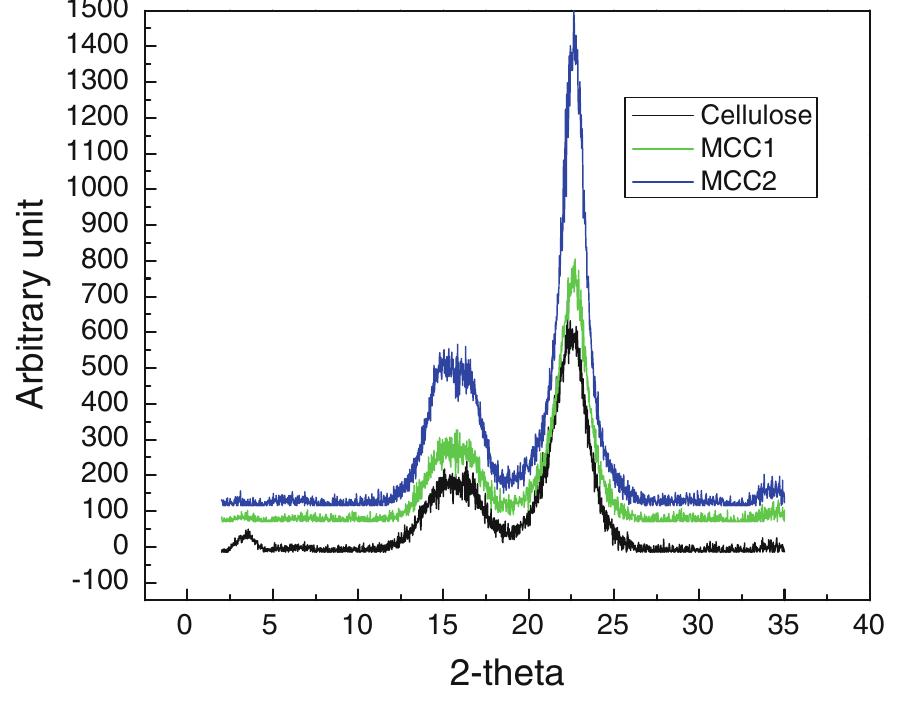
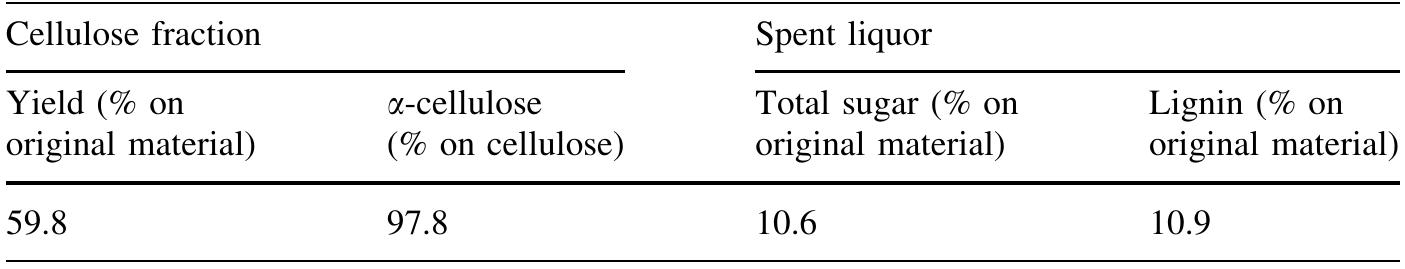
Cellulose was extracted at a yield of 59.8% from jute fibres based on the formic acid/ peroxyformic acid process at an atmospheric pressure. The amounts of dissolved lignin and hemicelluloses were determined in the spent liquor. The... more
The antibacterial effect of addition of silver oxide to Na2O·CaO·2SiO2 glass have been studied. Silver containing and silver free Na2O·CaO·2SiO2 glasses have been prepared by sol–gel synthesis using tetramethil orthosilicate, sodium... more
Polypropylene (PP)/cenosphere based composites were fabricated and characterized for their structural/ morphological and mechanical properties such as tensile, flexural, impact and dynamic mechanical properties such as storage and loss... more
The present paper reports the results about a study of mechanical, thermal, dynamic mechanical and electrical properties of housing (weather shed) materials for outdoor polymeric insulators. Silicone rubber, ethylene-propylenediene... more
Non-stoichiometric nickel oxide thin films were prepared by pyrolytic decomposition of aerosol droplets of aqueous nickel acetate solution. Conventional un-nebulized spray pyrolysis system was used for the synthesis of thin films. The... more
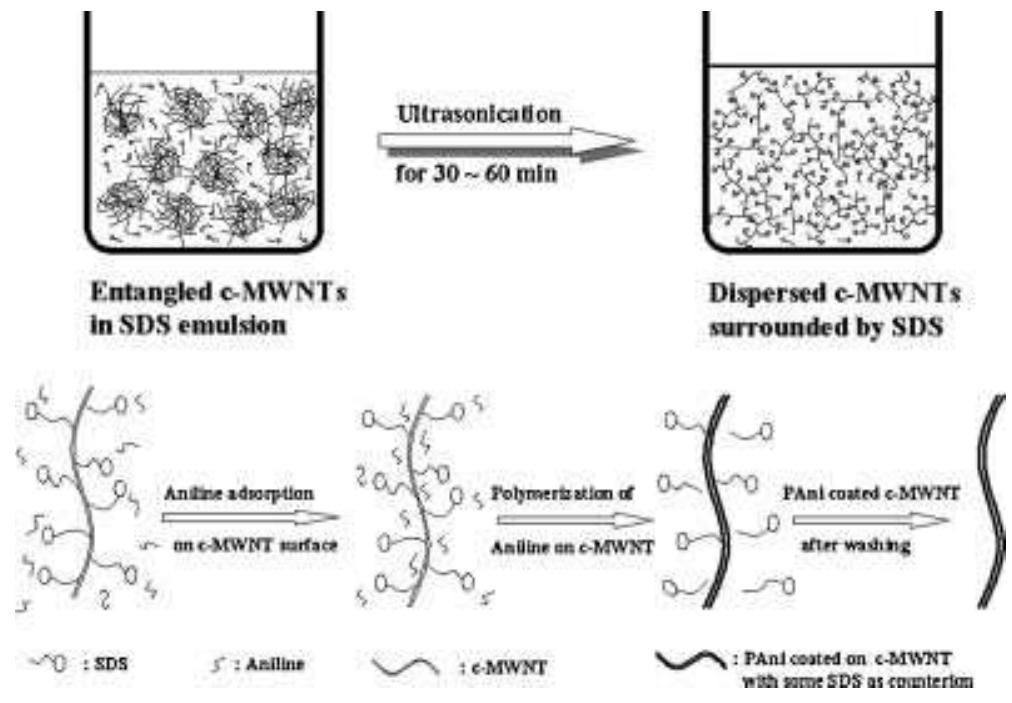
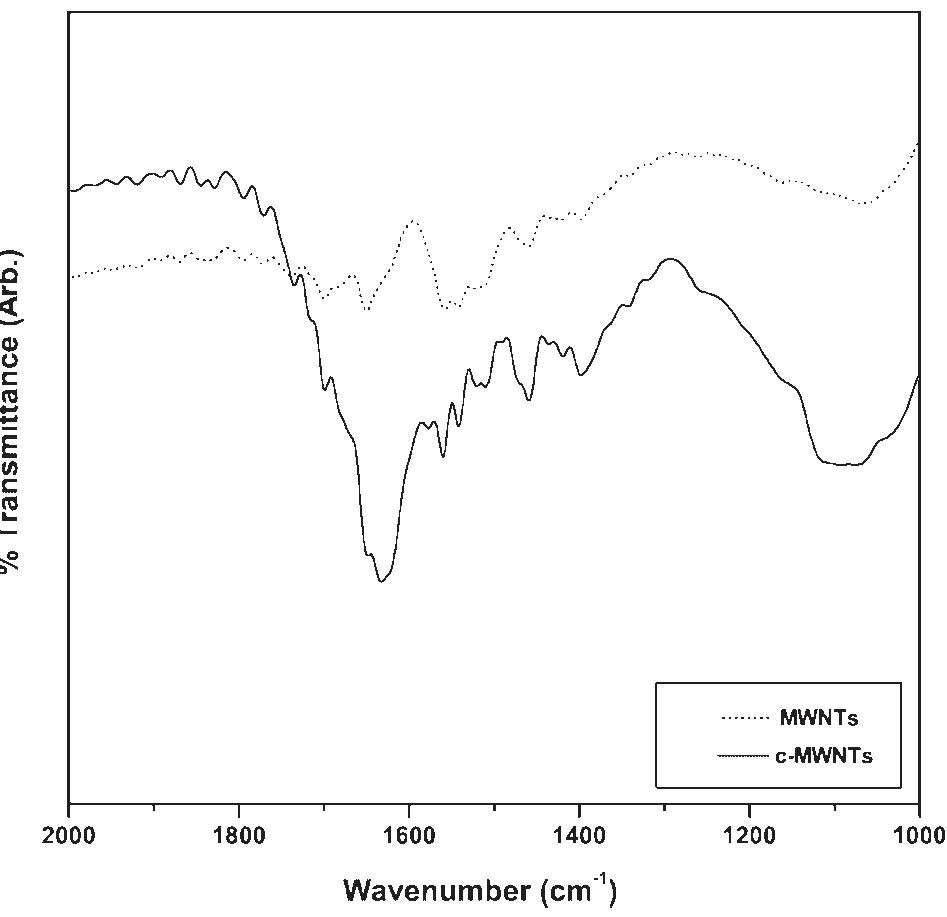
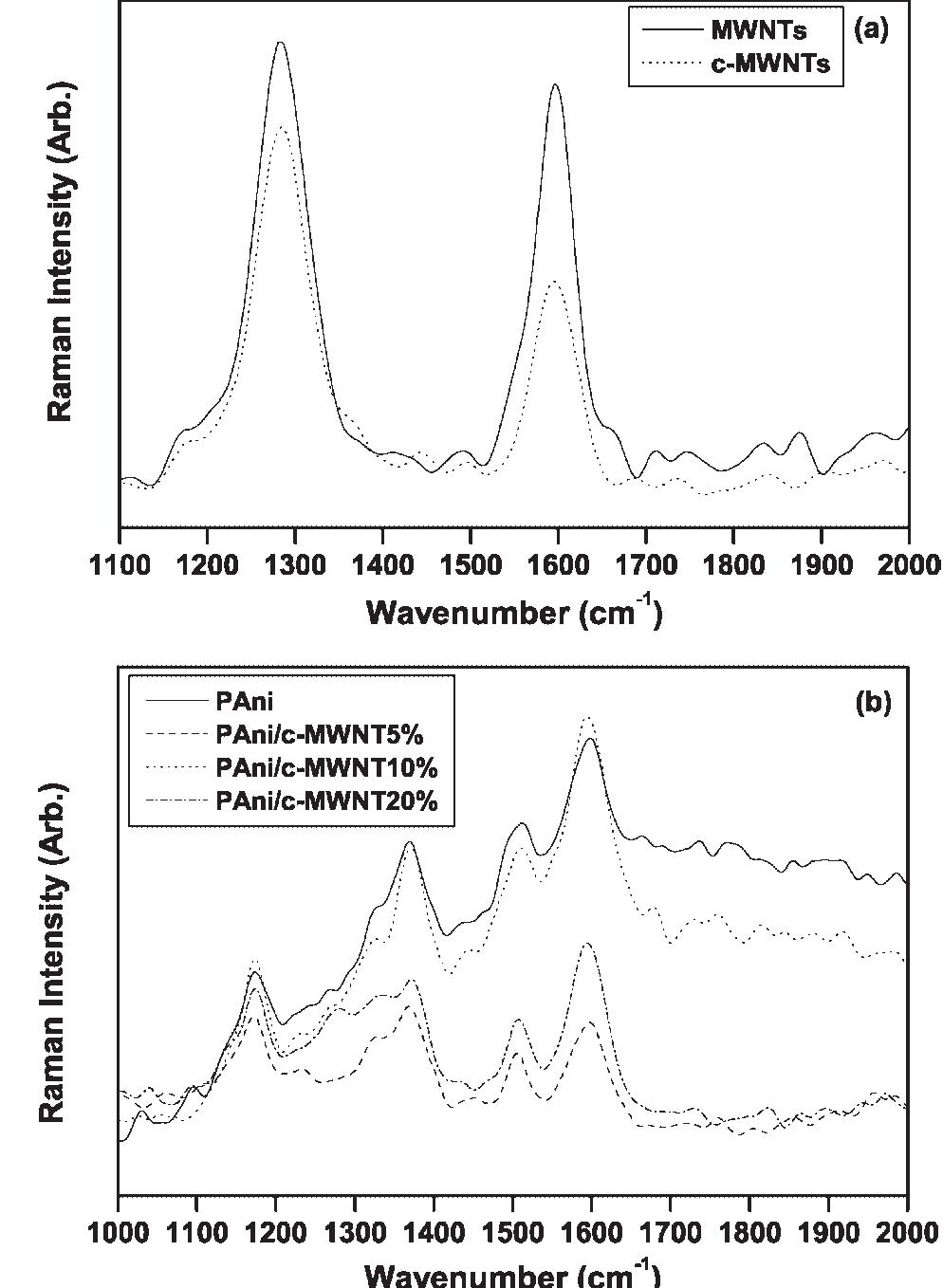
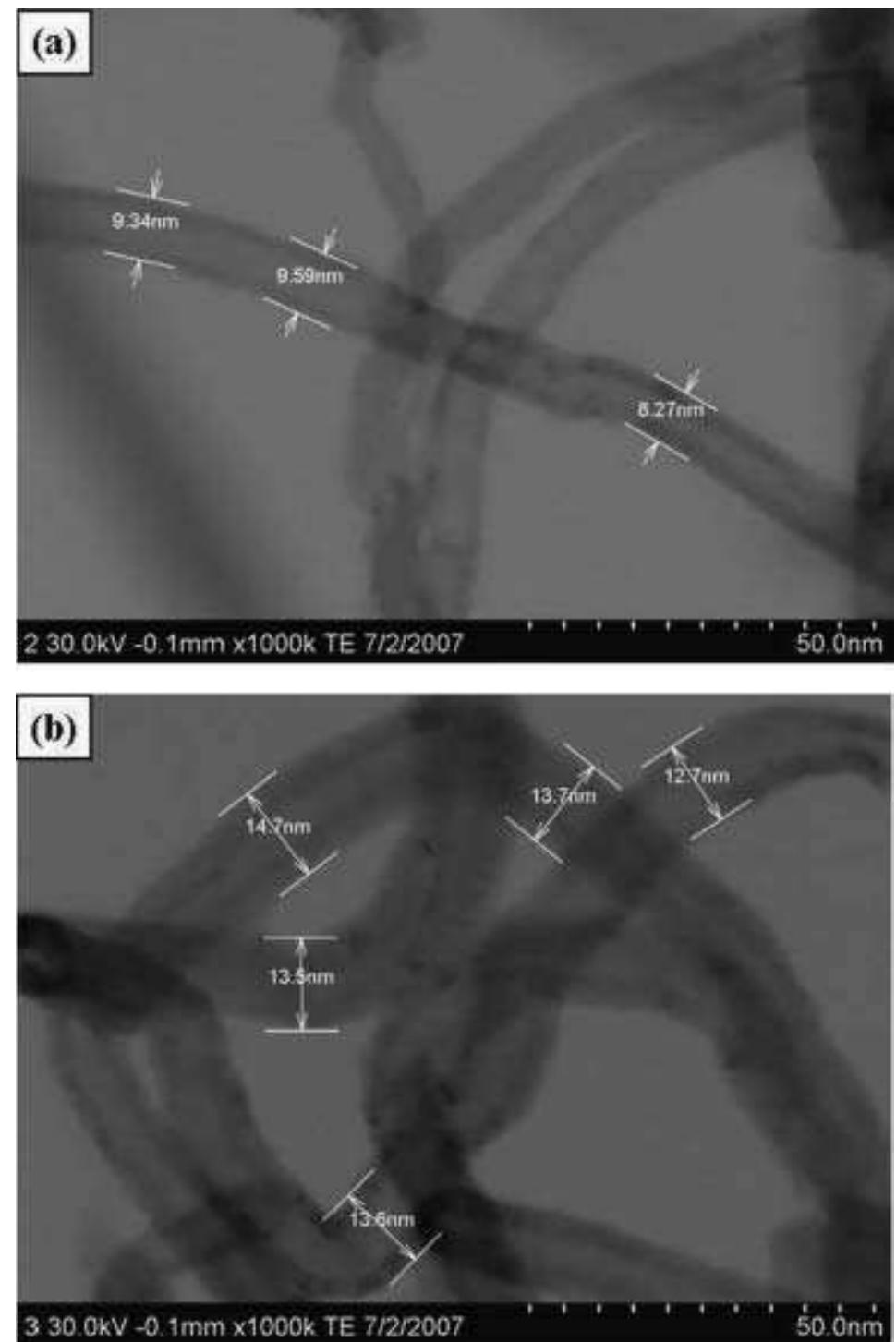
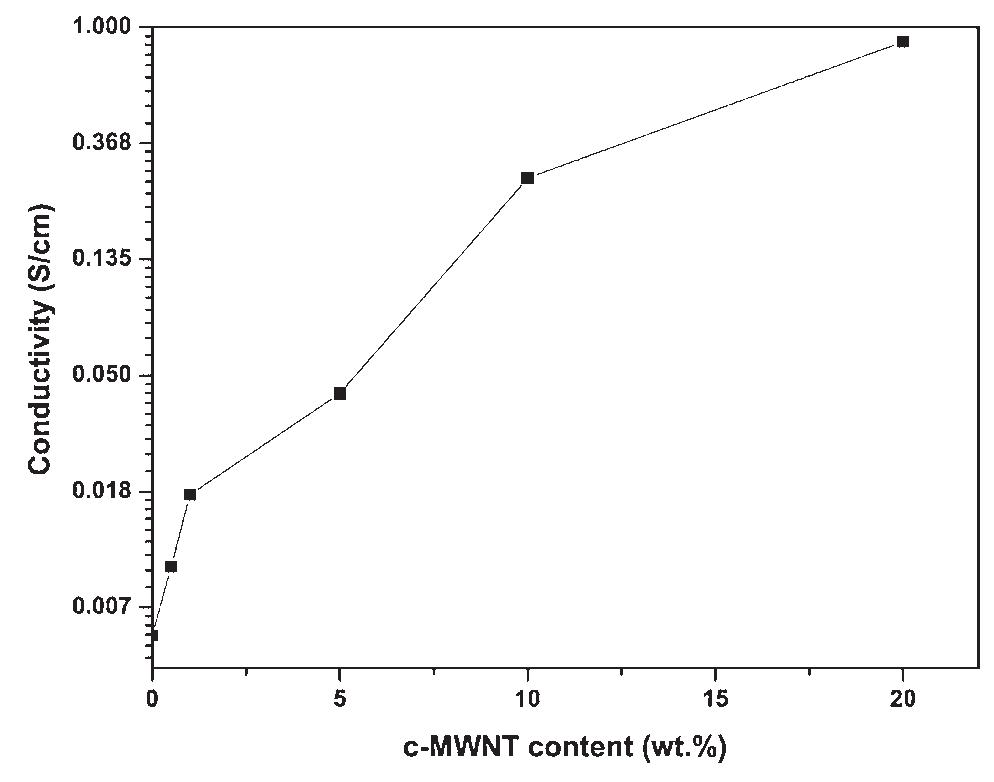
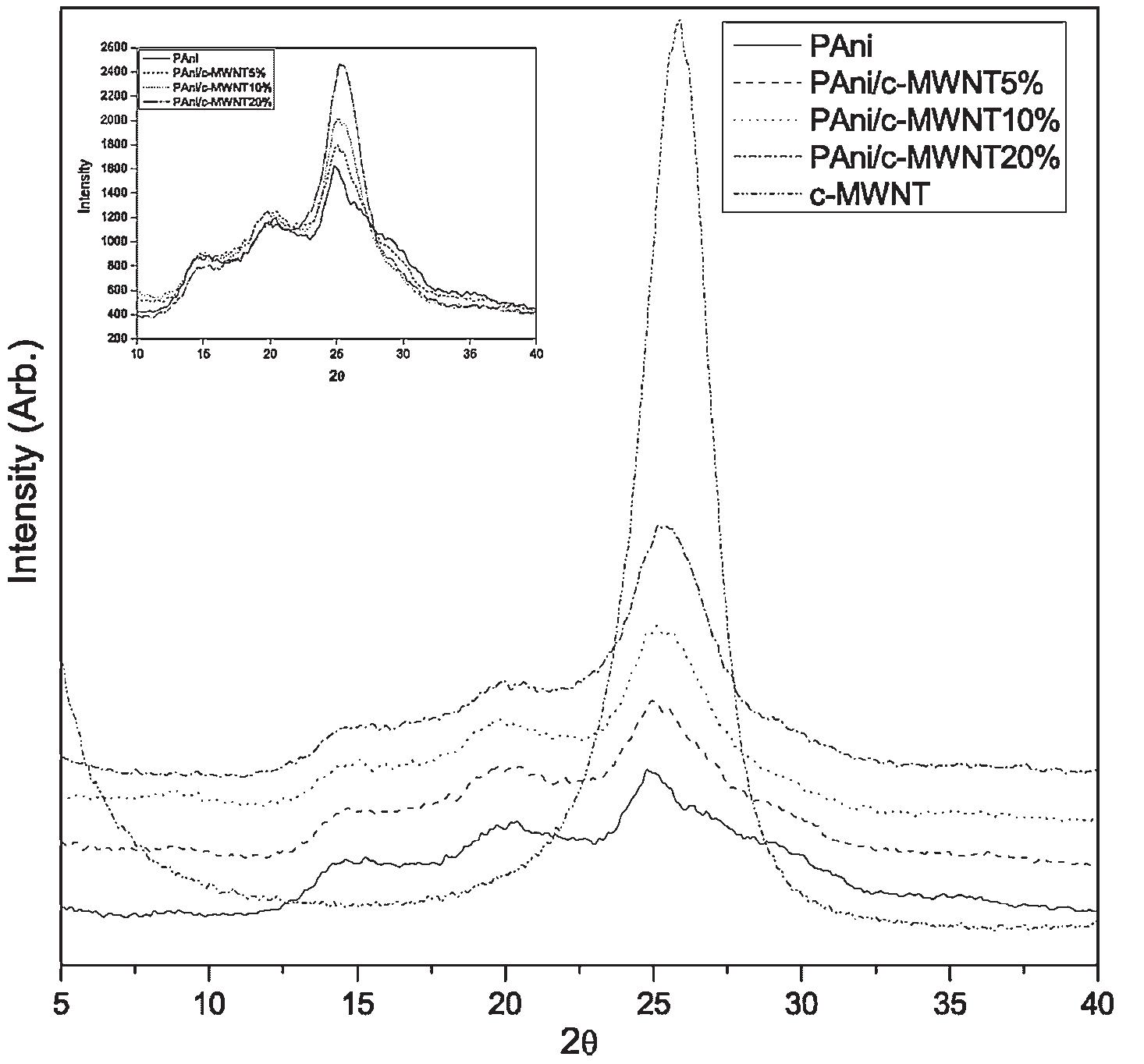
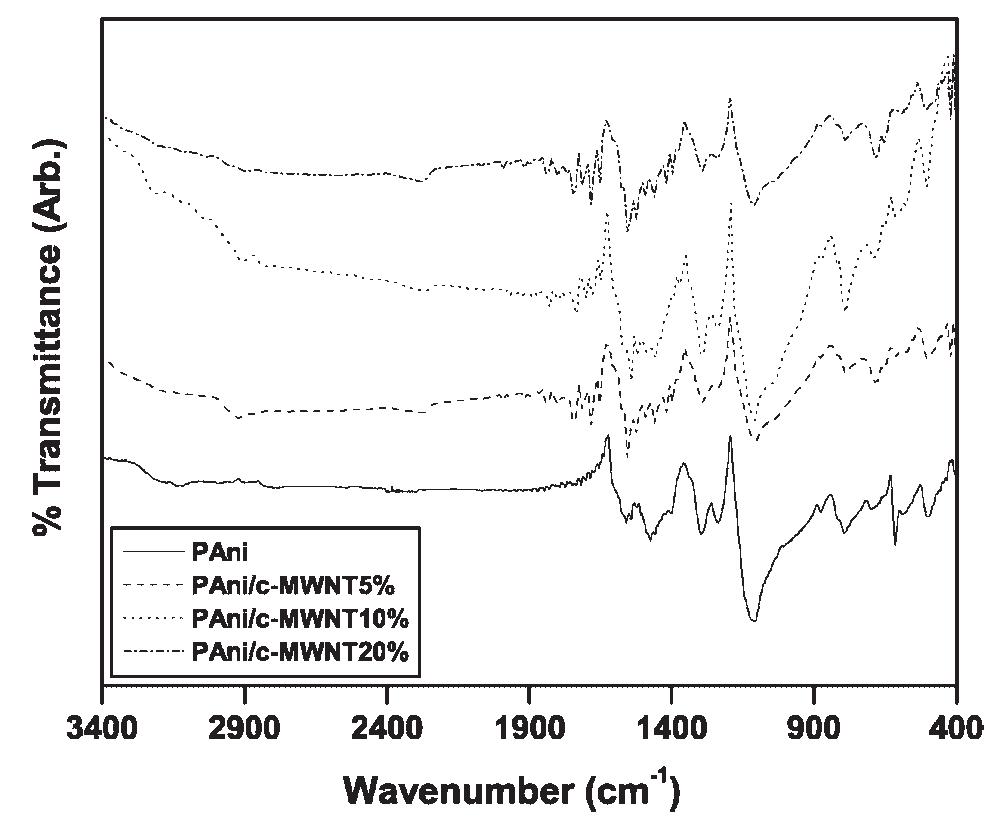
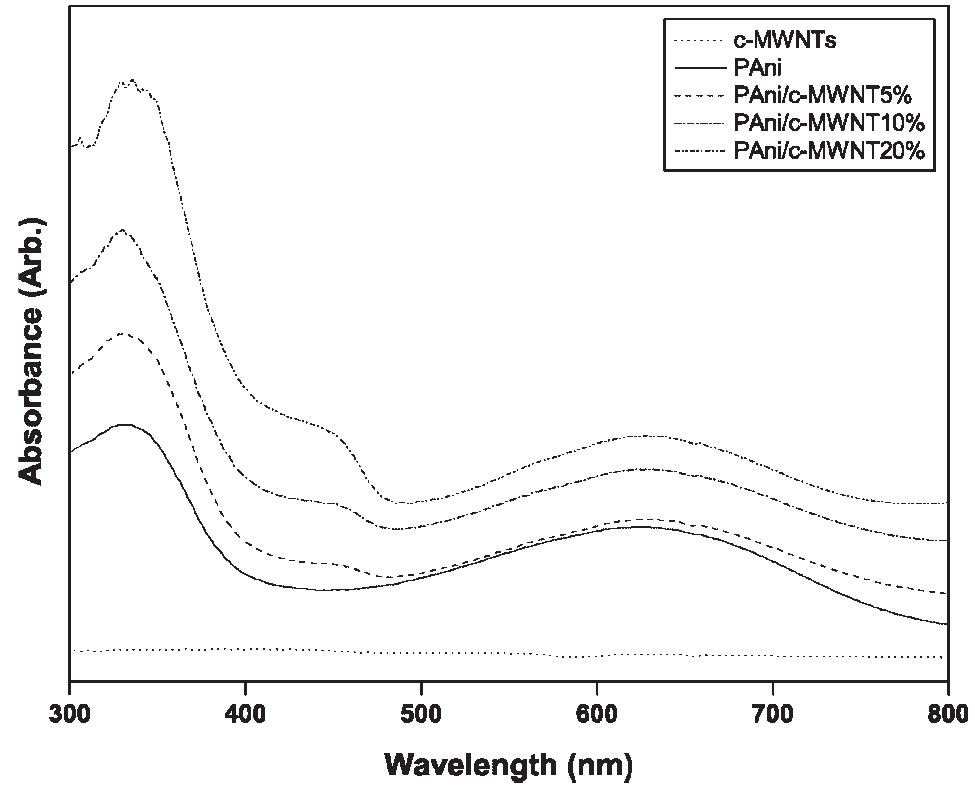
Polyaniline-carboxylic acid functionalized multi-walled carbon nanotube (PAni/c-MWNT) nanocomposites were prepared in sodium dodecyl sulfate (SDS) emulsion. First, the c-MWNTs were dispersed in SDS emulsion then the aniline was... more
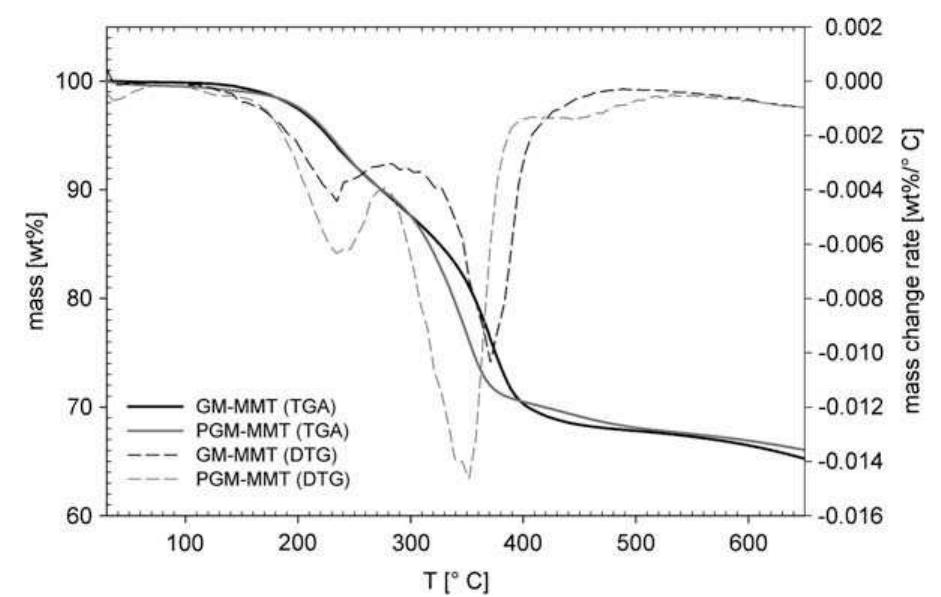
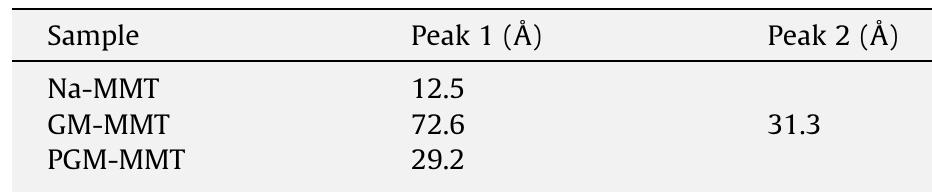
Vitamin B 1 (thiamine hydrochloride, VB 1 ) intercalated into montmorillonite (MMT), which was characterized by X-ray diffraction (XRD), Fourier transformed infrared spectroscopy (FT-IR), and thermo gravimetric analysis (TGA). The... more
The novel N-heterocyclic chitosan aerogel derivatives were prepared by reacting 79% deacetylated chitosan separately with 4-pyridinecarboxaldehyde and 2,6-pyridinedicarboxaldehyde followed by subsequent solvent exchange into acetone,... more
The solvothermal reactions of 1,4-bezenedicarboxylic acid (H 2 BDC) or azobenzene-4,4 -dicarboxylic acid (H 2 ABD) with zinc ions/clusters lead to the formation of four crystalline materials. All of these compounds were characterized by... more
Ricinus communis (RC) oil-based materials are currently receiving increasing attention because of economic and environmental concerns. In the present work, RC oil—a natural triol has been utilized for the development of an advanced... more
Chitosan-N-2-methylhydroxypyridine-6-methylcorboxylate (Ch-PDC) and chitosan-N-2methylhydroxypyridine-6-methylhydroxy thiocarbohydrazide (Ch-PDC-Th) were synthesized for the first time using chitosan as precursor. Chitosan, Ch-PDC,... more
The effects of nanoclay on the structure and final properties of high density polyethylene (HDPE)/thermoplastic starch (TPS) blends were investigated. Neat blends as well as nanoclay containing samples were prepared by melt blending in an... more
In the field of thermal shielding for aerospace applications C f /SiC composites are raising great interest, provided that they are protected from oxidation by suitable coatings. Conversely, ultra high temperature ceramics, and in... more
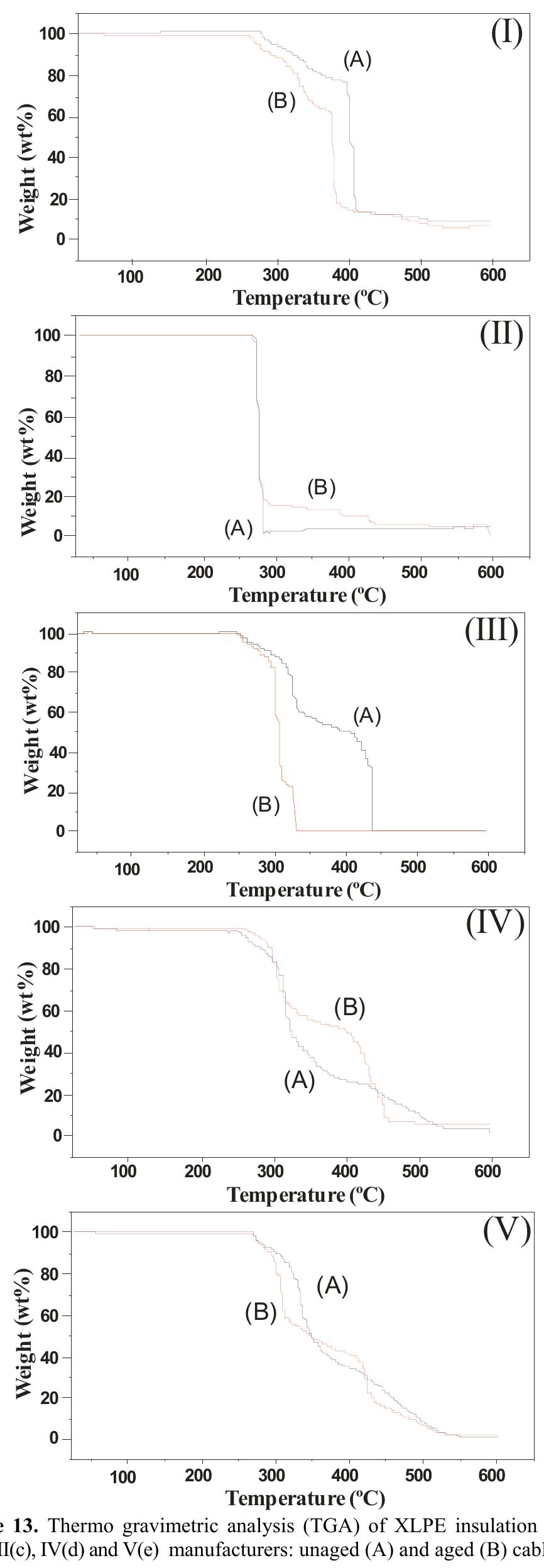
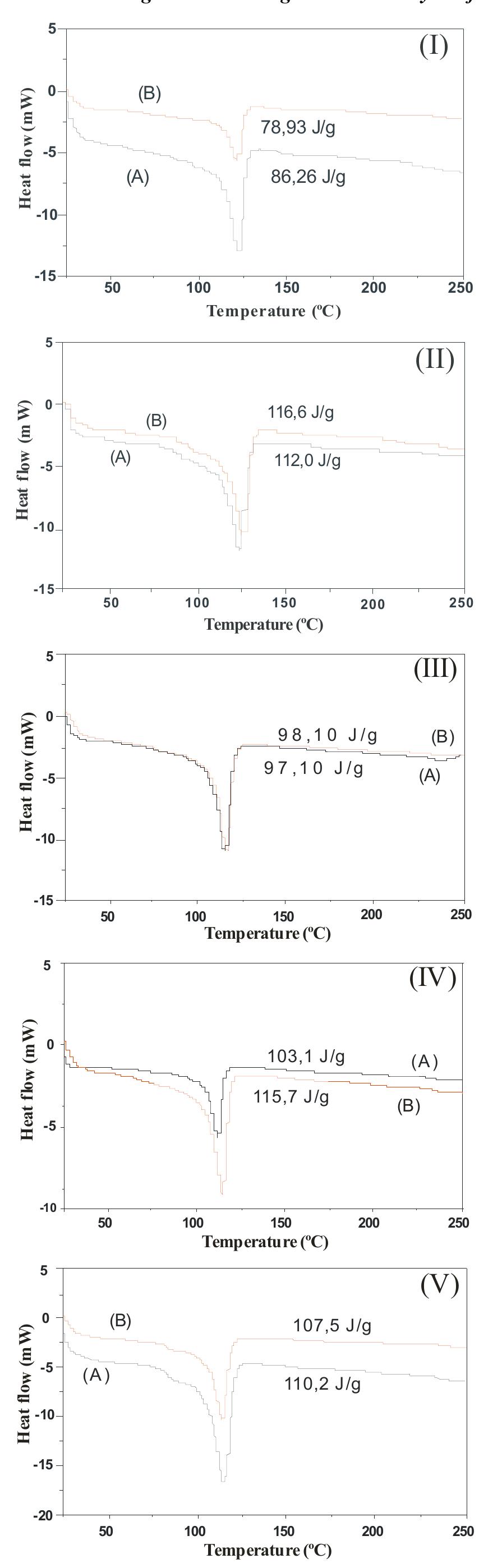
Today the knowledge of the ageing status of an electrical installation and its components is of fundamental interest for correctly inferring on the network economics. In Brazil, covered conductors must withstand an ageing test combining... more
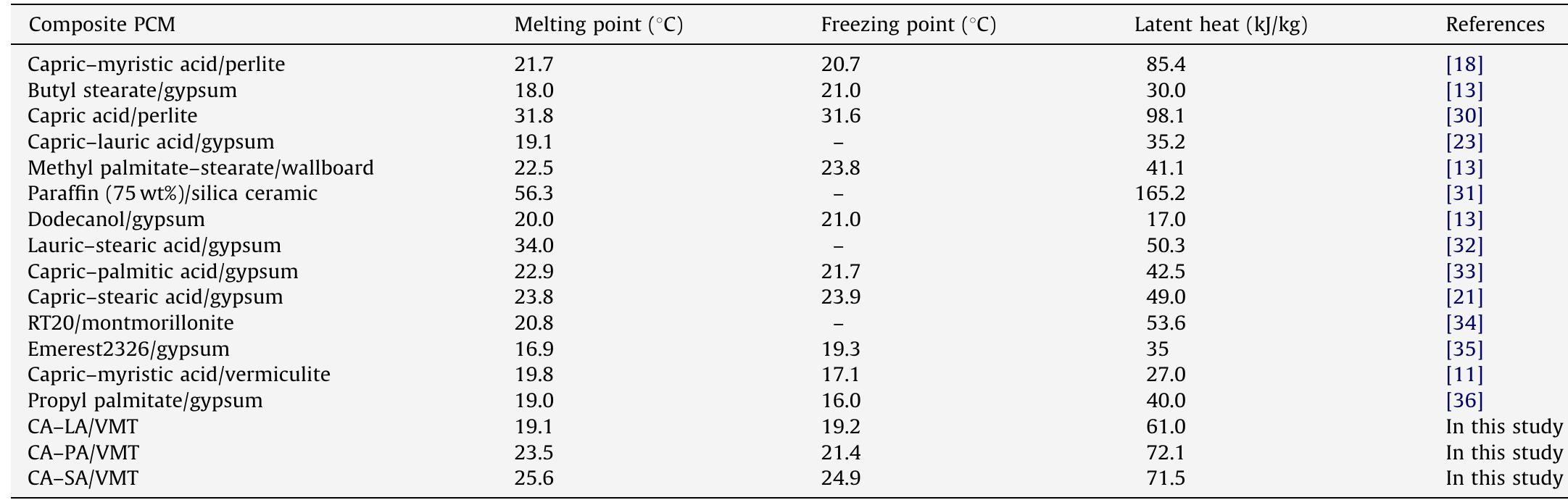
This paper deals with the preparation, characterization, thermal properties and thermal reliability of novel form-stable composite phase change materials (PCMs) composed of eutectic mixtures of fatty acids and expanded vermiculite for... more
Polypropylene (PP)emontmorillonite nanocomposites have been prepared using isotactic PP homopolymers with different rheological properties, and a maleic anhydride grafted PP. Morphology and structure of the composites were investigated by... more
Poly(o-methylaniline) (POTO), poly(o-methoxyaniline) (POAS), poly(2,5-dimethylaniline) (PDMA), poly-(2,5-dimethoxyaniline) (PDOA), and nanocomposite based on multi-walled carbon nanotubes (MWNTs) and single-walled carbon nanotubes (SWNTs)... more
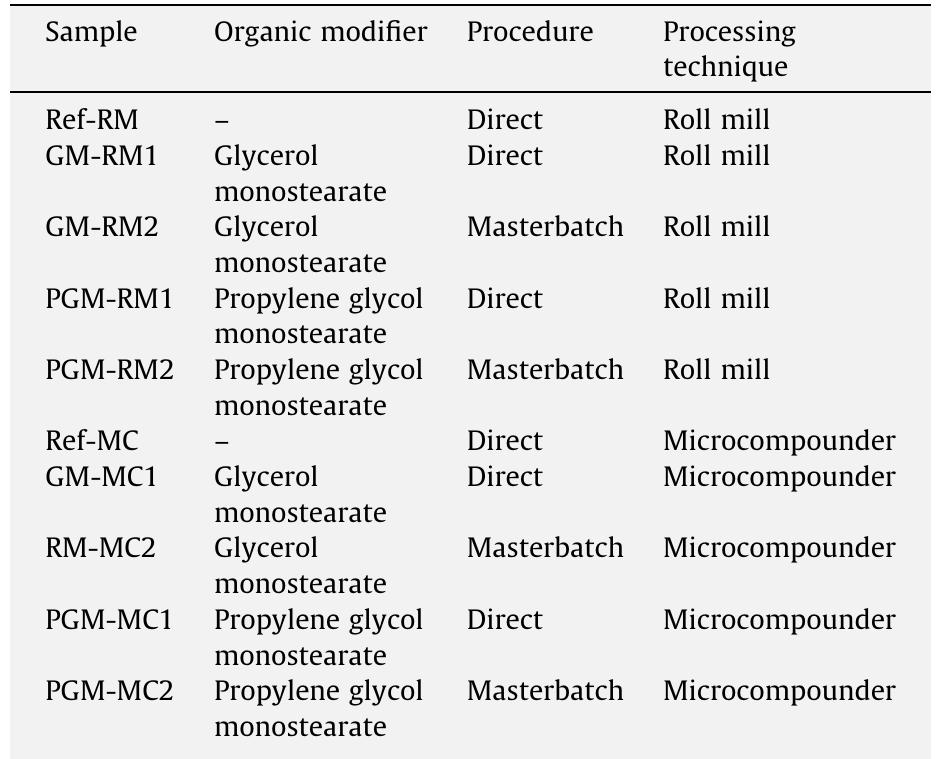
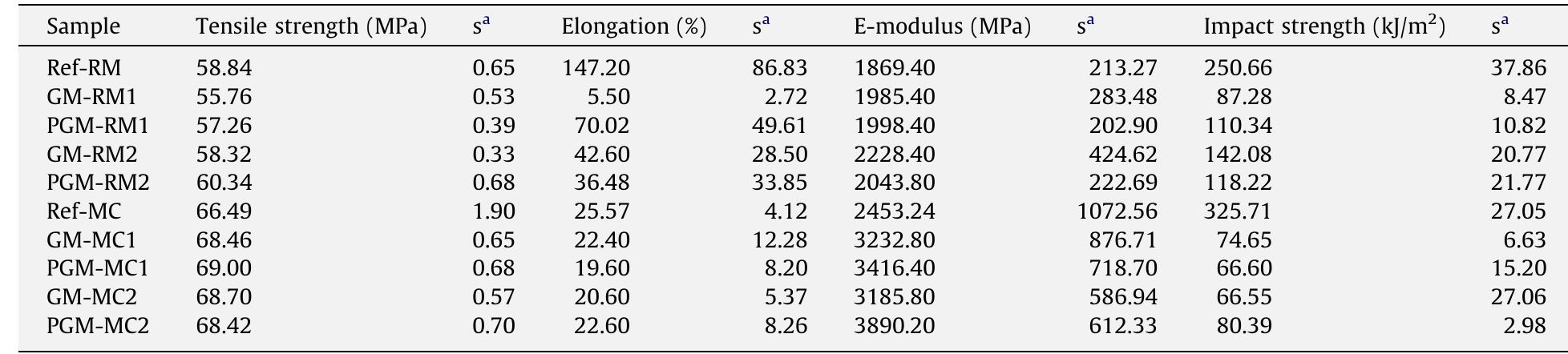
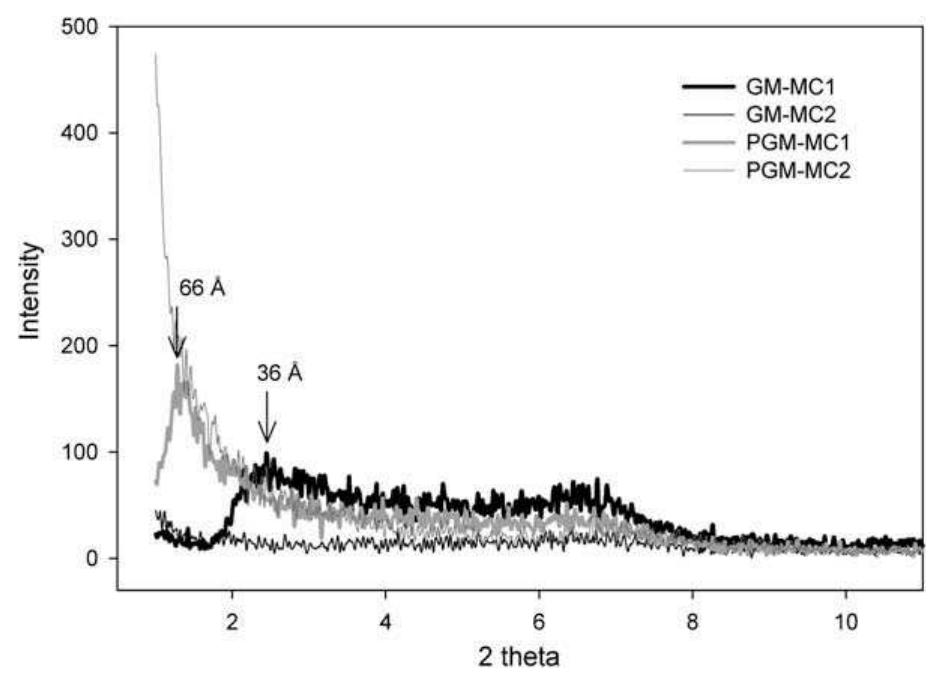
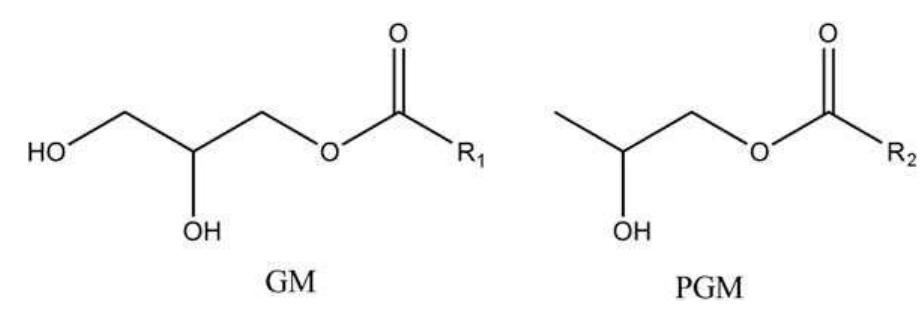
This study aims to compare the influence of processing techniques and procedures on the morphology and mechanical properties of polyvinyl chloride (PVC) nanocomposites. PVC nanocomposites with 5 phr montmorillonite (MMT) clay were... more
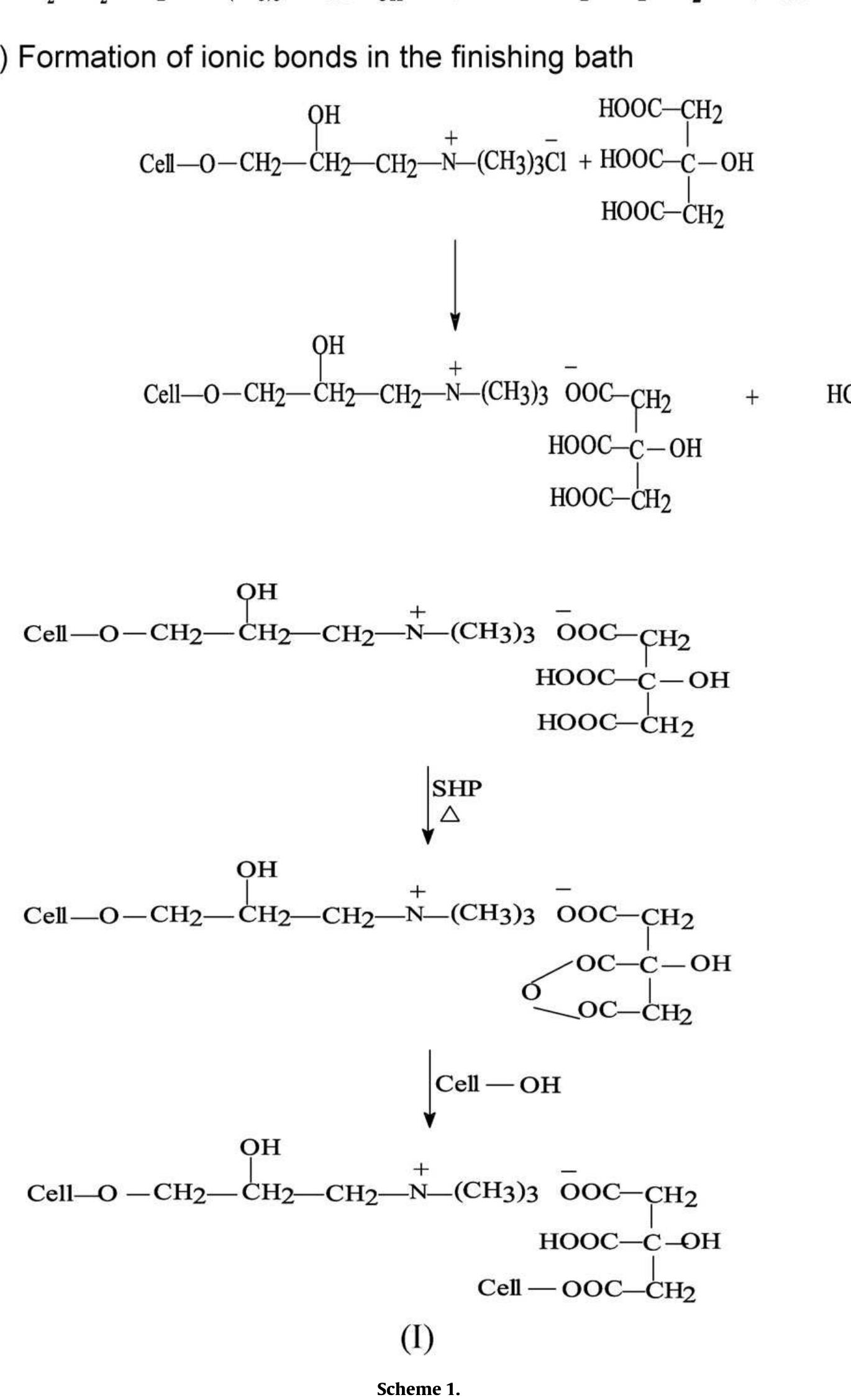
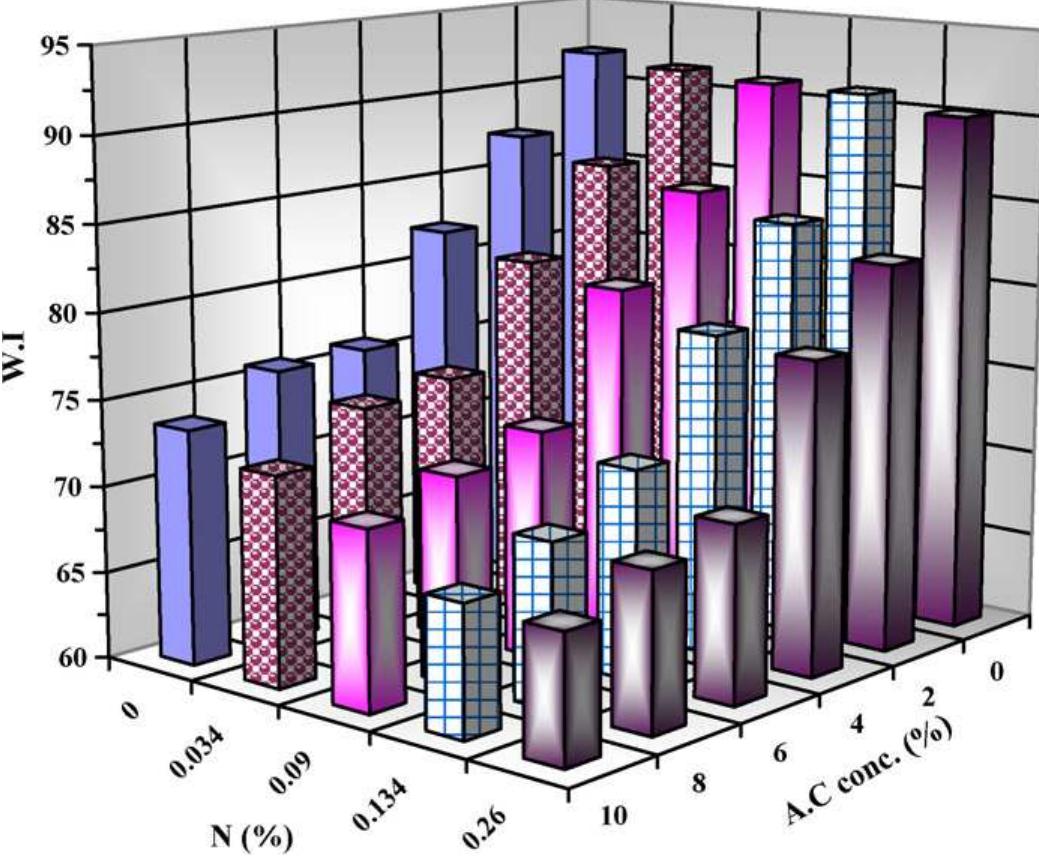
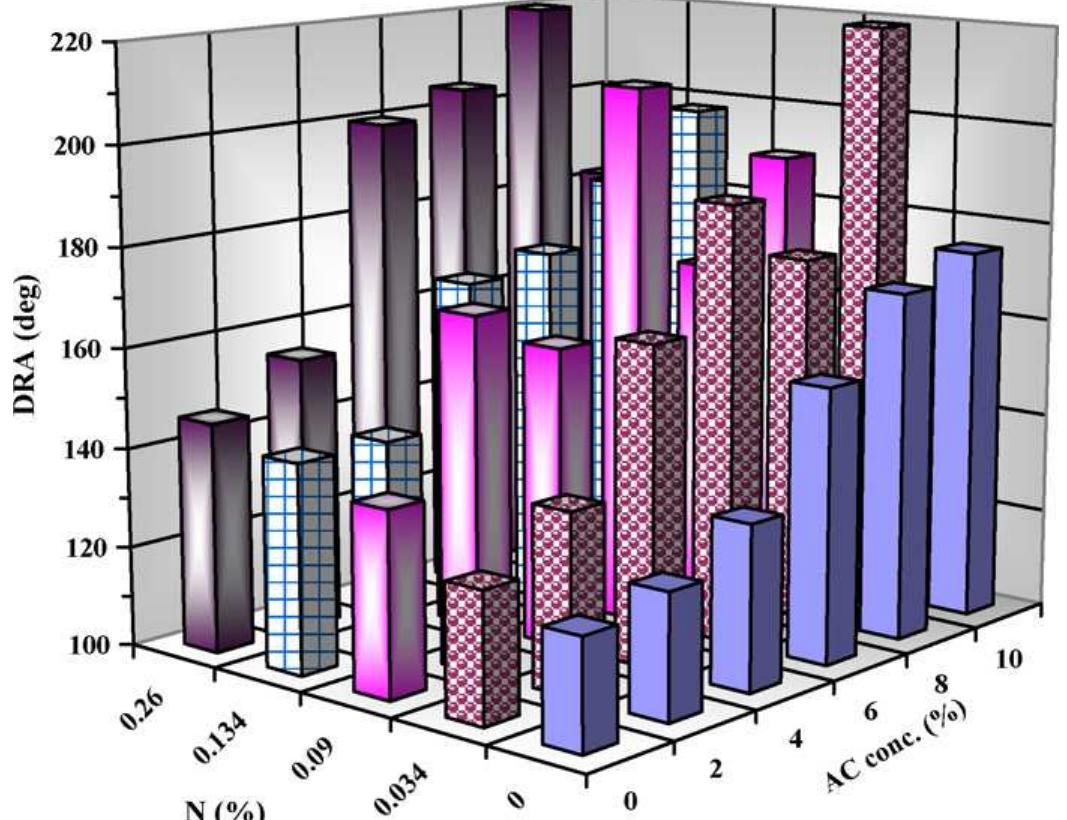
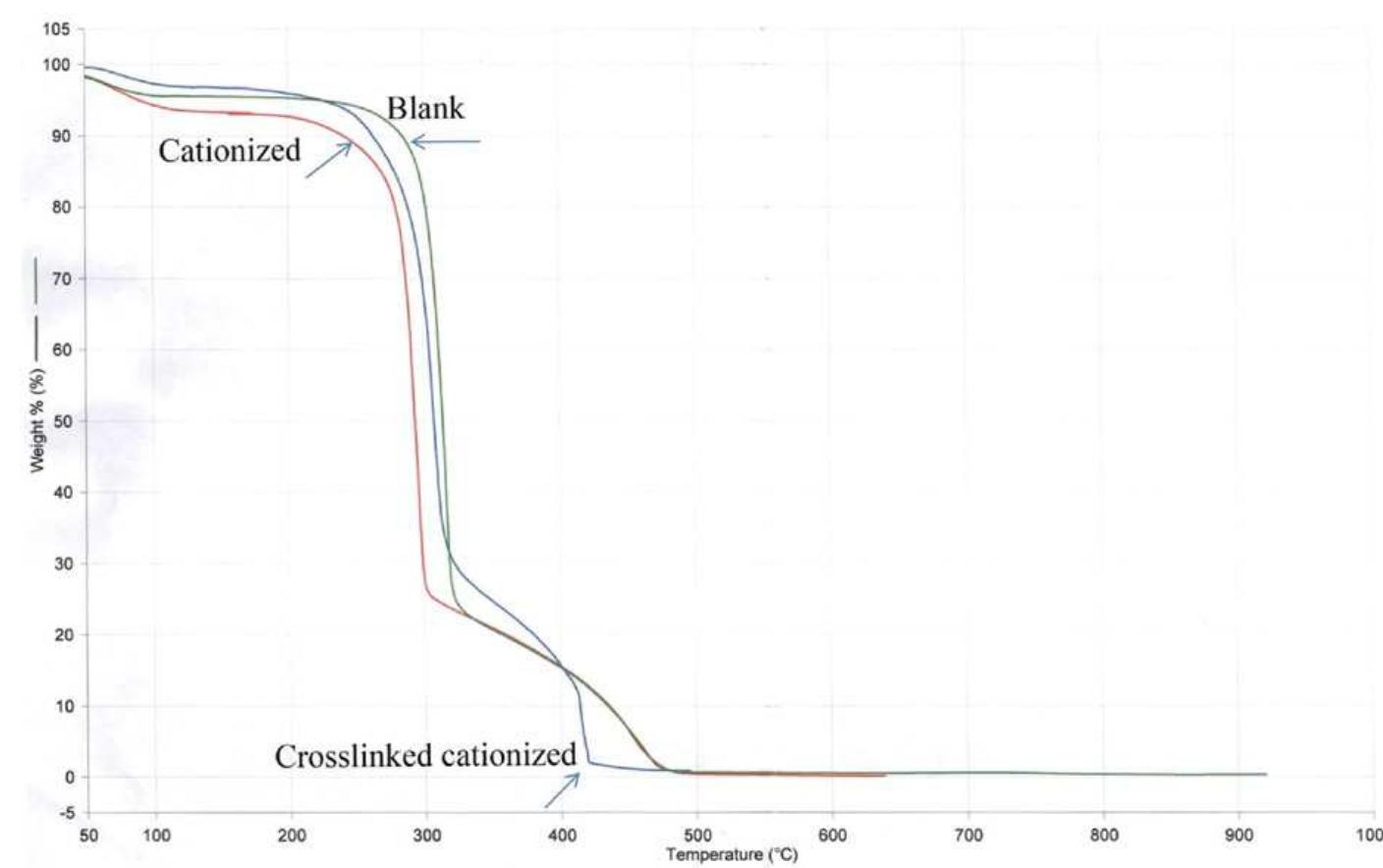
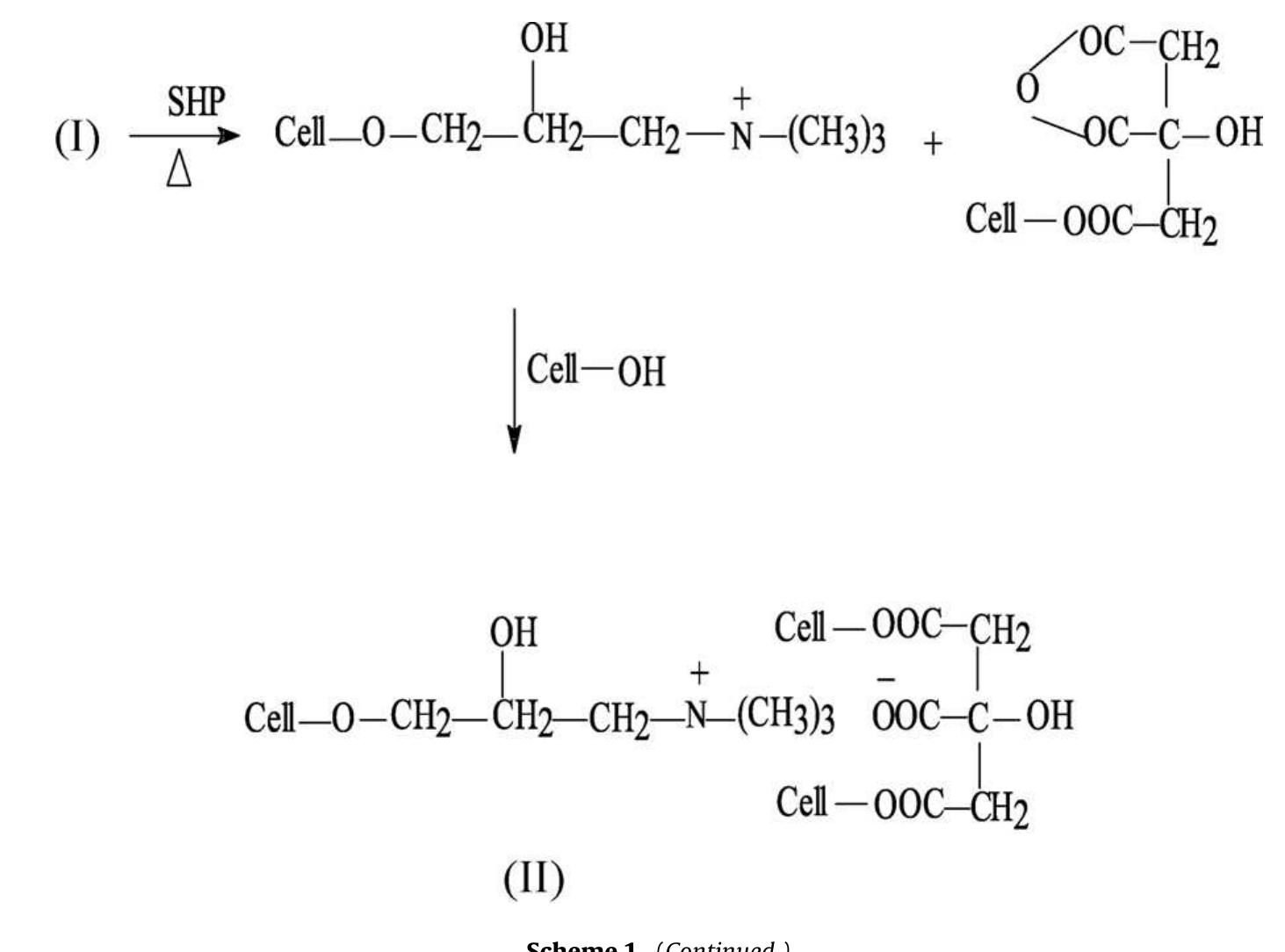
To enhance the easy care finishing properties of cotton fabric along with maintaining its mechanical properties, a new route based on ionic crosslinking was employed. In this research work, pre-cationized cotton fabric was crosslinked... more
A laboratory scale fluidized bed pyrolysis system has been designed and fabricated for obtaining liquid fuel from biomass solid wastes. The components of the system are: fluidized bed reactor, gas preheating chamber, reactor feeder,... more
Bioplastics based on glycerol and different proteins (wheat gluten, albumen, rice and albumen/gluten blends) have been manufactured to determine the effect that processing and further thermal treatments exert on different... more
Discovery of hydrogels has resulted in developing competent controlled-release drug delivery systems. Present study describes the synthesis and characterization of novel pH responsive hydrogels of chitosan, hydroxyl ethyl cellulose (HEC)... more
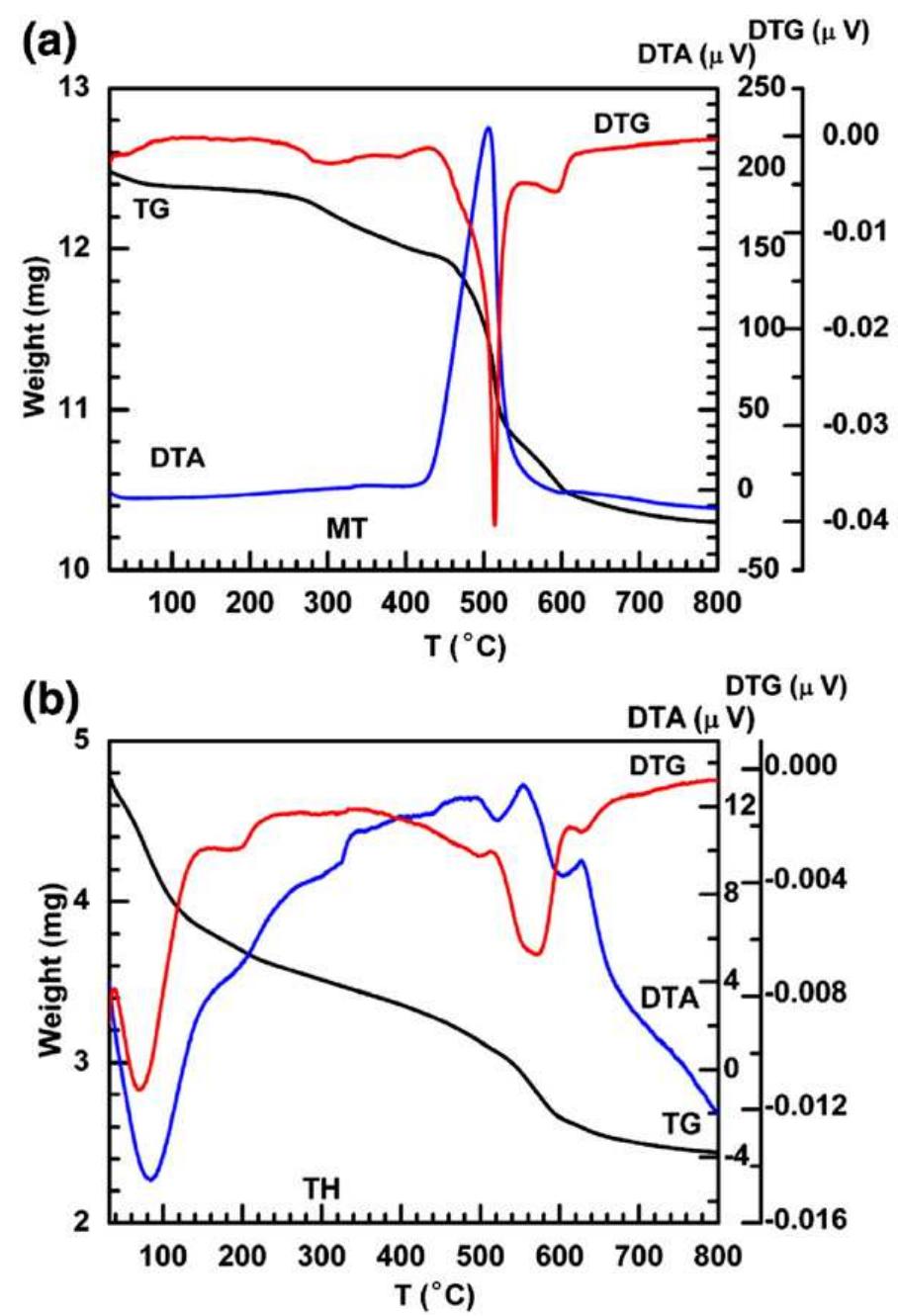
Many workers [1–9] studied the kinetics of dolomite decomposition to study the effects of different parameters like, gas (CO2, N2 etc.) pressures, water vapor, presence of other impurities, particle size and grain size of the dolomite... more
The thermal properties of micro-sized boron nitride (BN) and nano-sized BN dispersed high density polyethylene (HDPE) composites were investigated by means of differential scanning calorimetry (DSC) and thermo-gravimetric analysis (TGA).... more
Carbon nanofibers were prepared through electrospinning a blend solution of polyacrylonitrile and polypyrrole, followed by carbonization at 700 • C. Structural features of electrospun polyacrylonitrile/polypyrrole bicomponent nanofibers... more
• Surfactant stabilized AgCl nanoparticles in aqueous medium were synthesized.
The influence of paraffin wax characteristics on the formulation of wax-based binders has been examined. Four paraffin waxes (PW 1 , PW 2 , PW 3 and PW 4) were physically and molecularly evaluated and four binder systems 1-4 were... more
Spinel MgGa 2 O 4 ceramic materials were synthesized by solid-state method. MgGa 2 O 4 powders were investigated with X-ray powder diffraction (XRD), Fourier transform infrared (FT-IR) spectrum, Raman spectrum, X-ray photoelectron... more
This paper explores the feasibility of converting waste Rohu fish (Labeo rohita) scale into a high-performance, reusable, low-cost heterogeneous catalyst for synthesis of biodiesel from soybean oil. The thermo-gravimetric analysis (TGA)... more
Synthesis of oleic acid capped copper nano-particles has been carried out by use of sodium formaldehyde sulfoxylate (SFS) in aqueous medium. Nano-copper can be effectively coated with polyvinyl alcohol (PVA). Phase-pure nano-Cu can be... more
Nanosized zinc ferrite (ZnFe 2 O 4 ) has been prepared directly from metallic iron (Fe) and zinc (Zn) powders by using wet-milling technique. Structural and magnetic properties of the samples were investigated by X-ray diffraction (XRD),... more
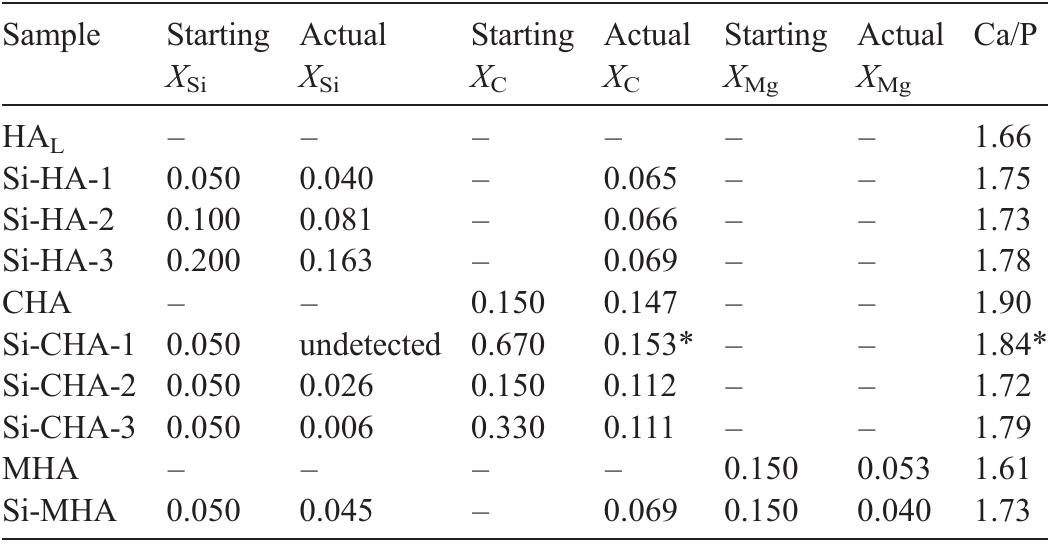
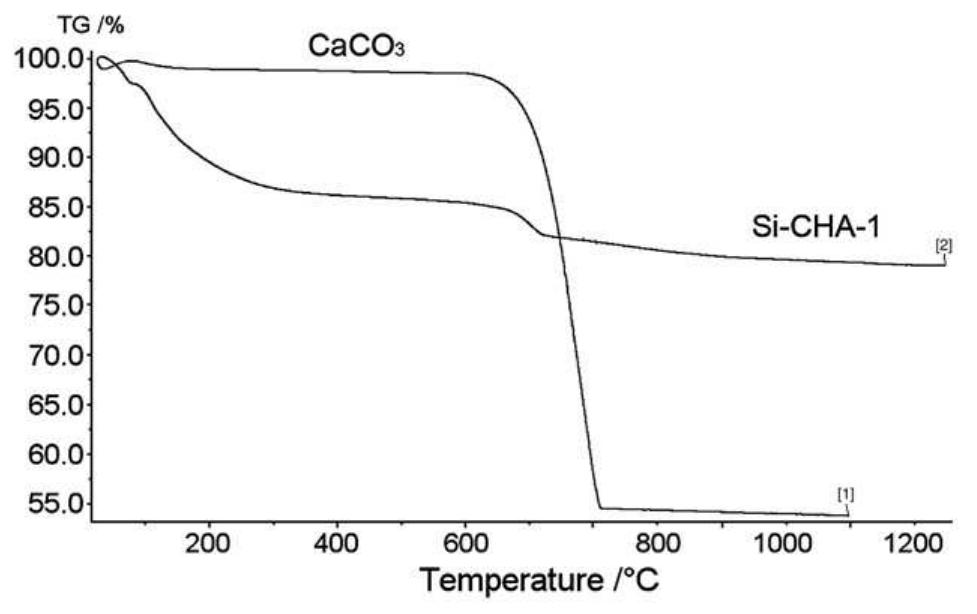
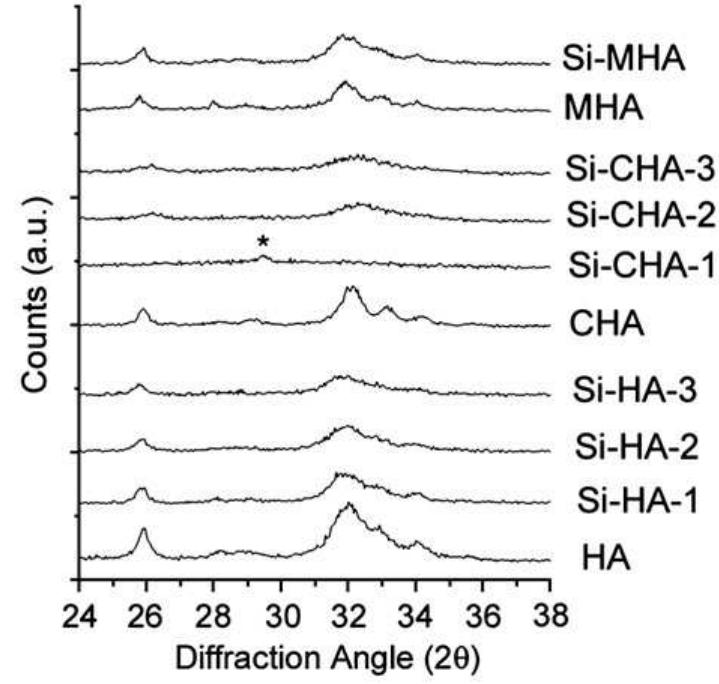
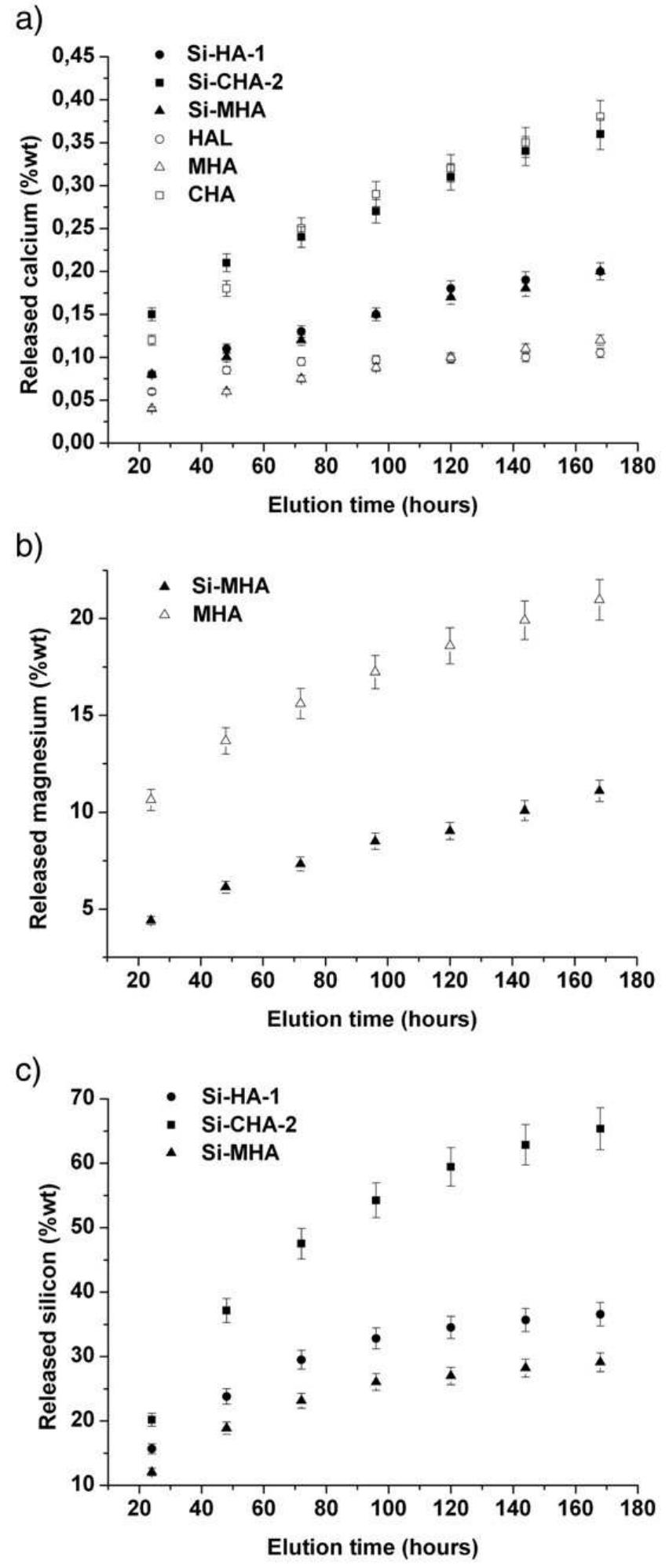
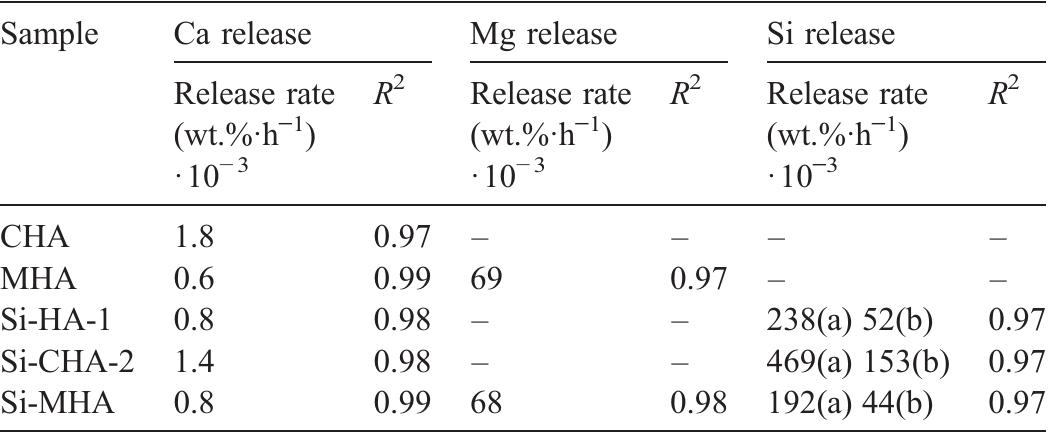
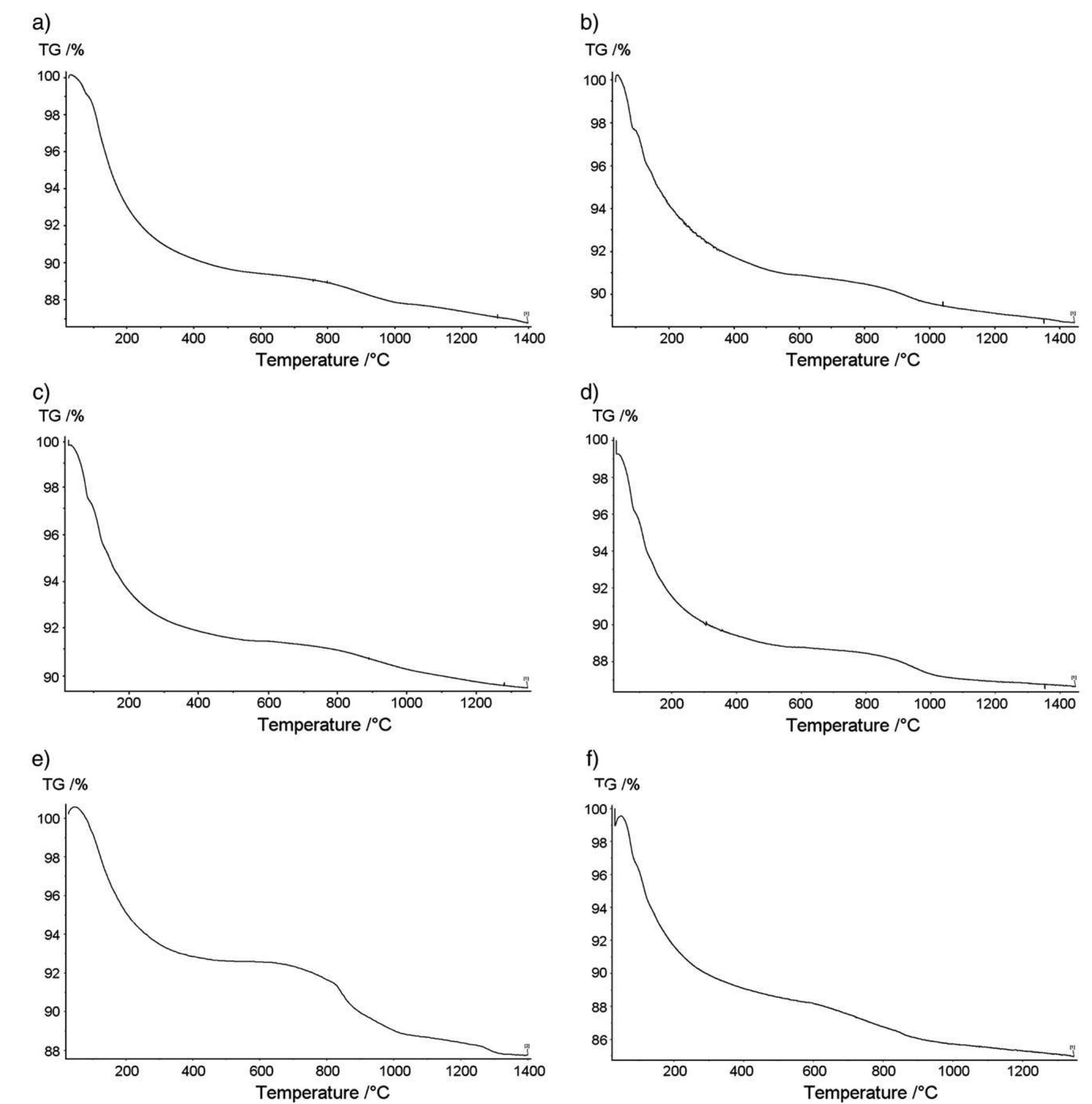
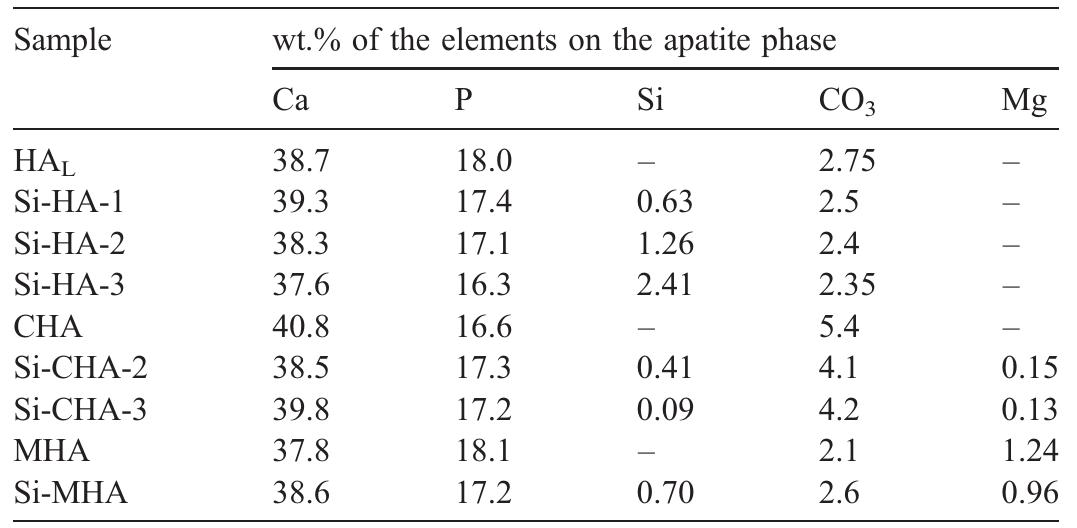
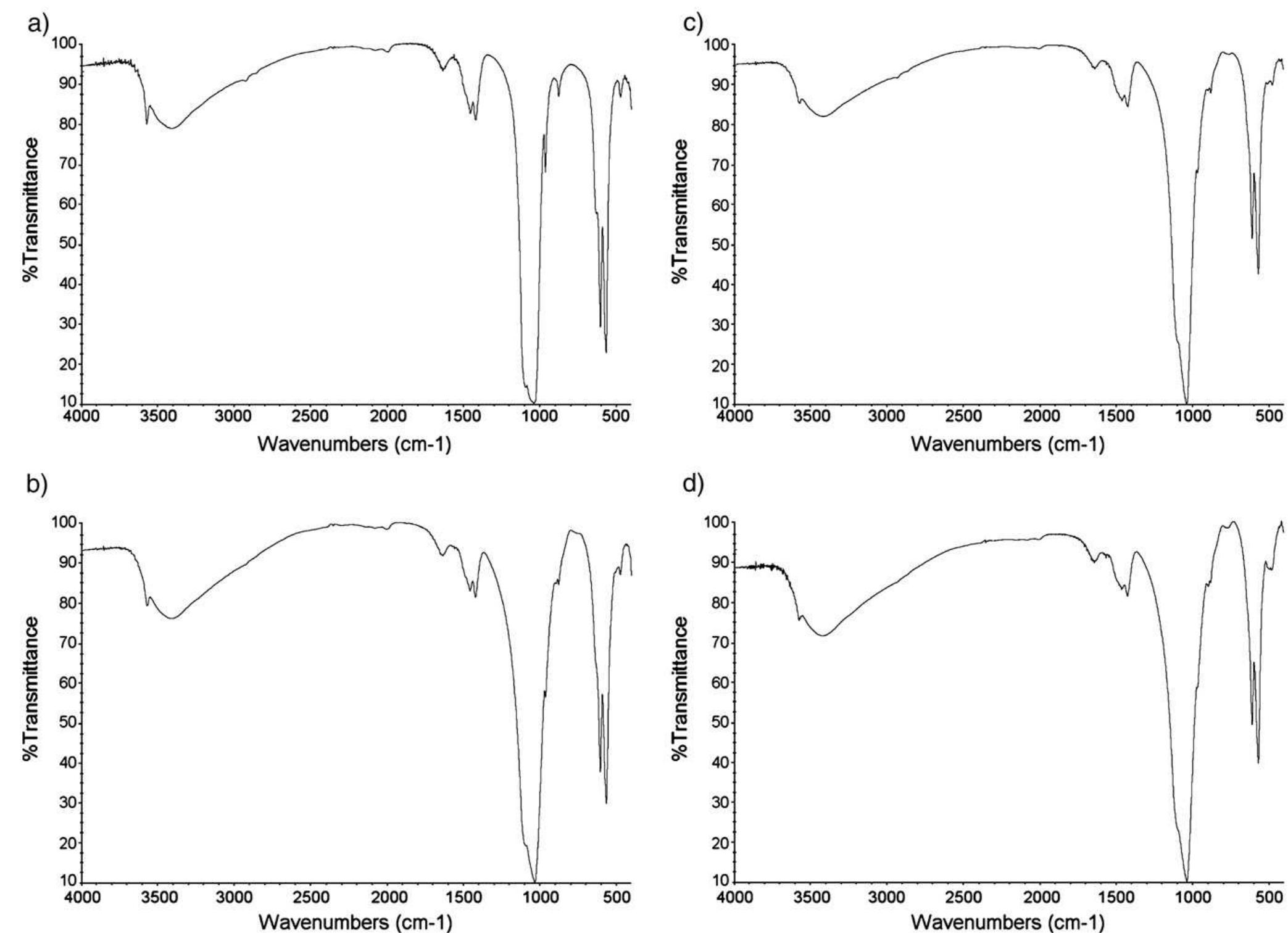
Hydroxyapatite powders characterized by ionic substitutions both in anionic and cationic sites were successfully prepared by synthesis in aqueous medium. The process parameters were set up to allow the simultaneous substitution of the... more
Most plastics, at present, are petroleum-based and do not degrade over many decades under normal environmental conditions. As a result, efforts towards developing environment-friendly and biodegradable 'green' plastics for various... more
Aluminum terephthalate, MIL-53(Al), metalorganic framework synthesized hydrothermally and purified by solvent extraction method was used as an adsorbent for gas adsorption studies. The synthesized MIL-53(Al) was characterized by powder... more
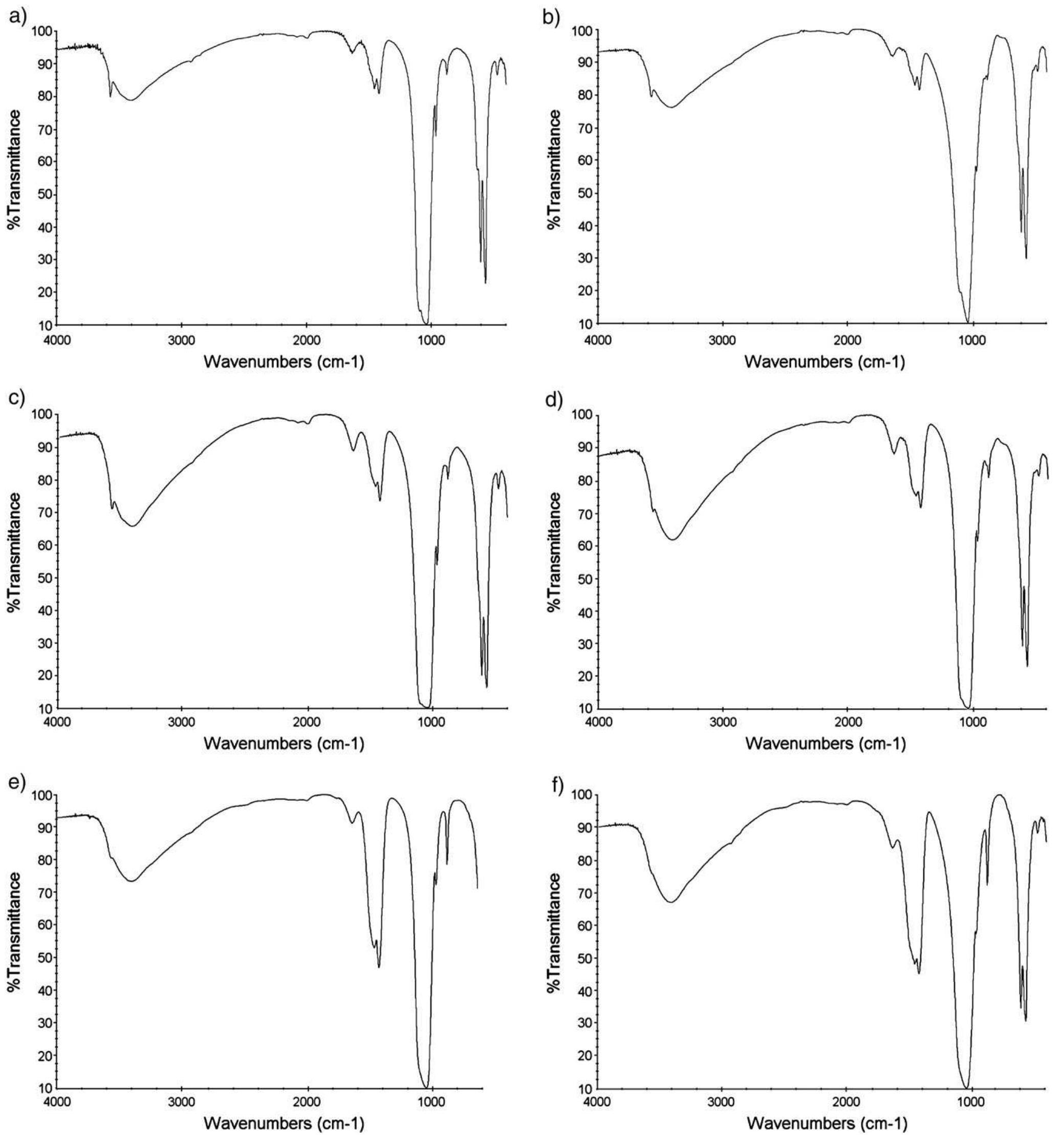
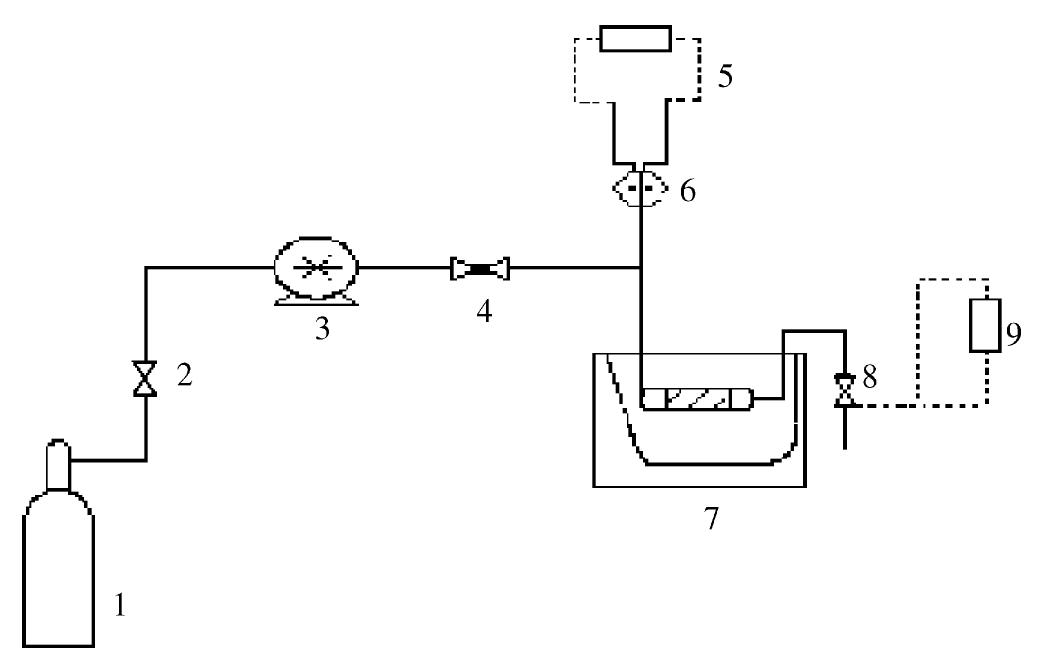
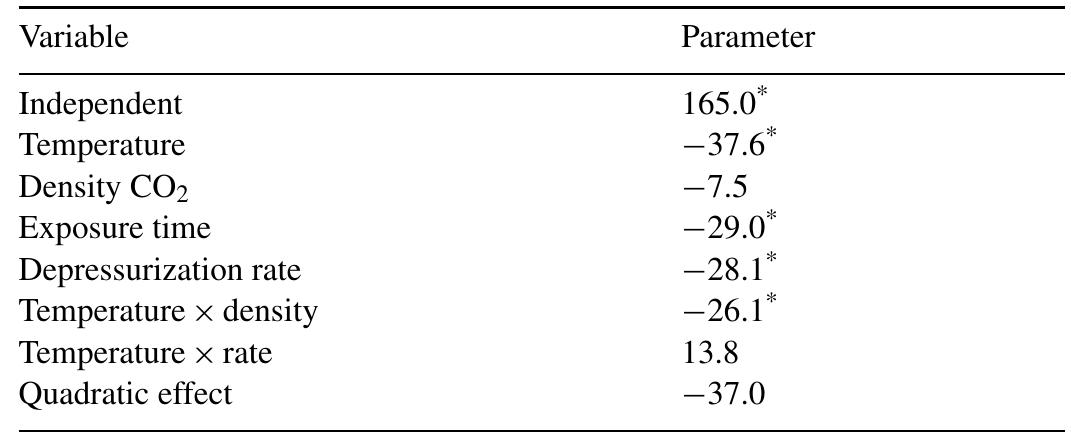
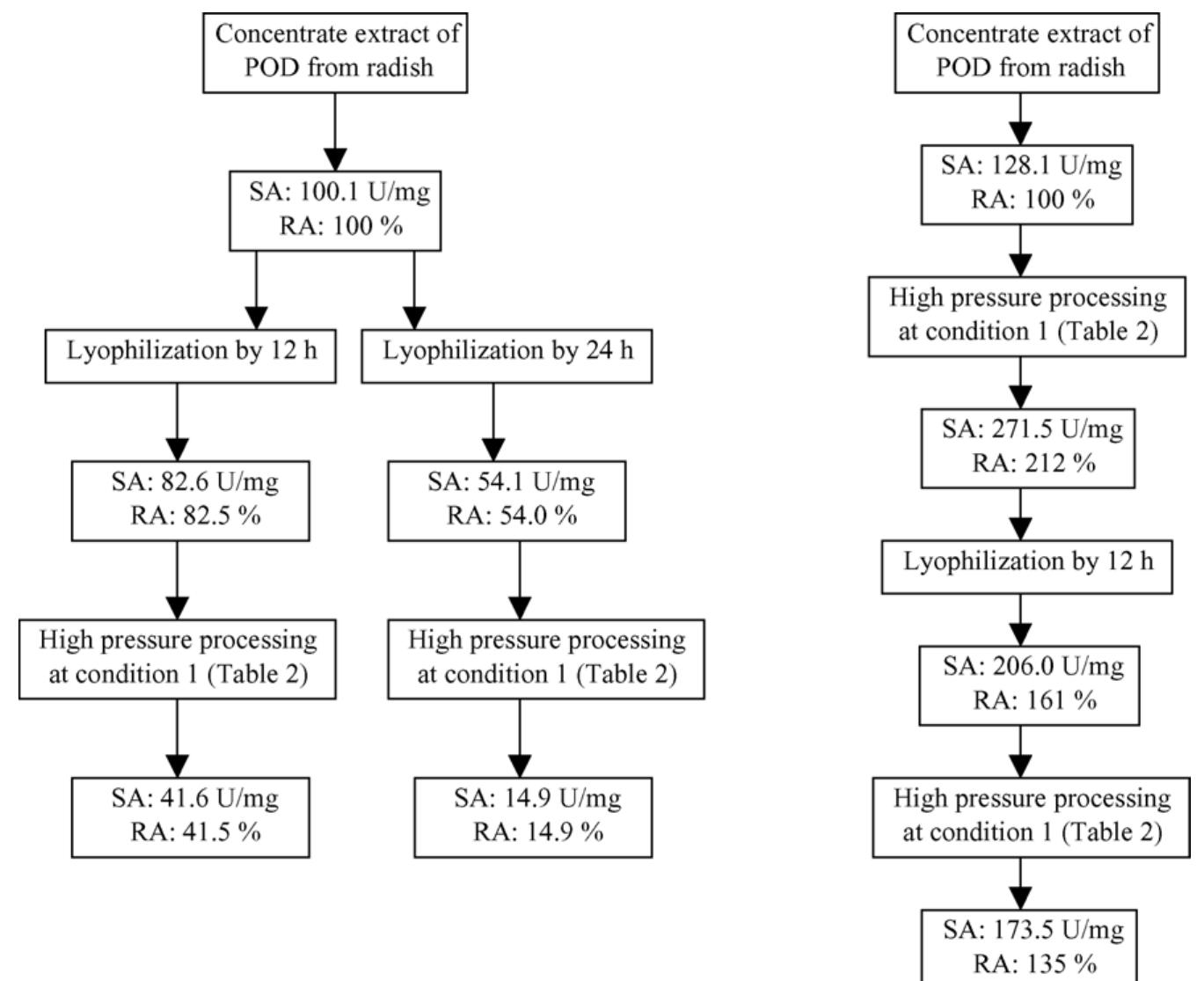
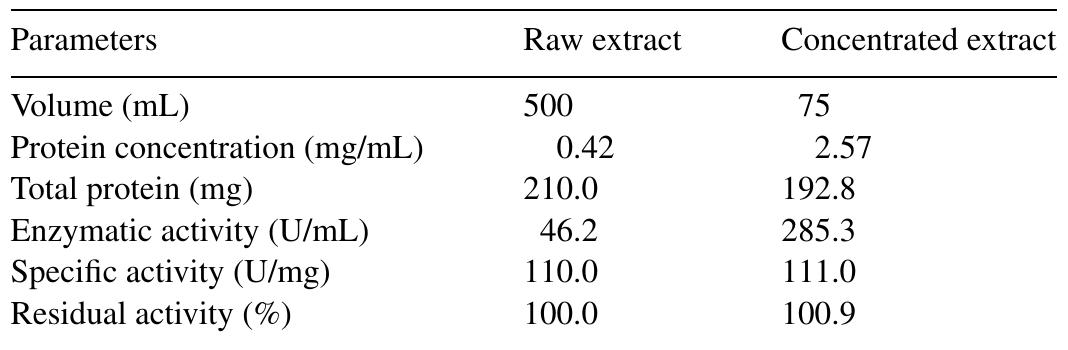
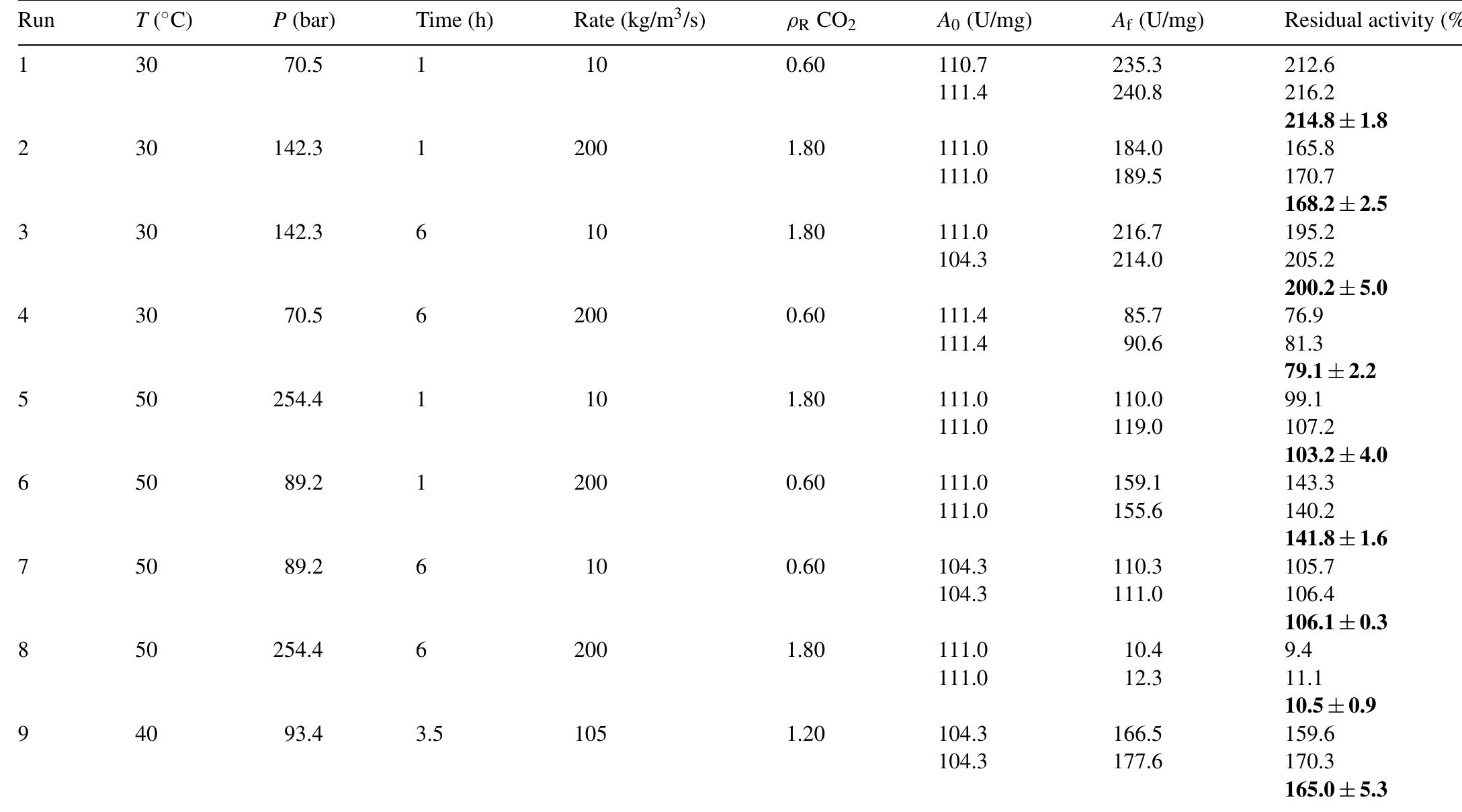
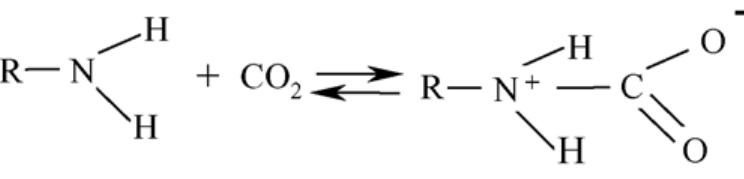
The objective of this work was to assess the influence of compressed CO 2 on the specific activity of peroxidase (POD) from radish (Raphanus sativus L.). The protein concentrate, in phosphate buffer, was obtained through the precipitation... more
Cerium titanium oxide nanocontainers were synthesized through a two-step process and then loaded with corrosion inhibitors 2-mercaptobenzothiazole (2-MB) and 8-hydroxyquinoline (8-HQ). First, polystyrene nanospheres (PS) were produced... more
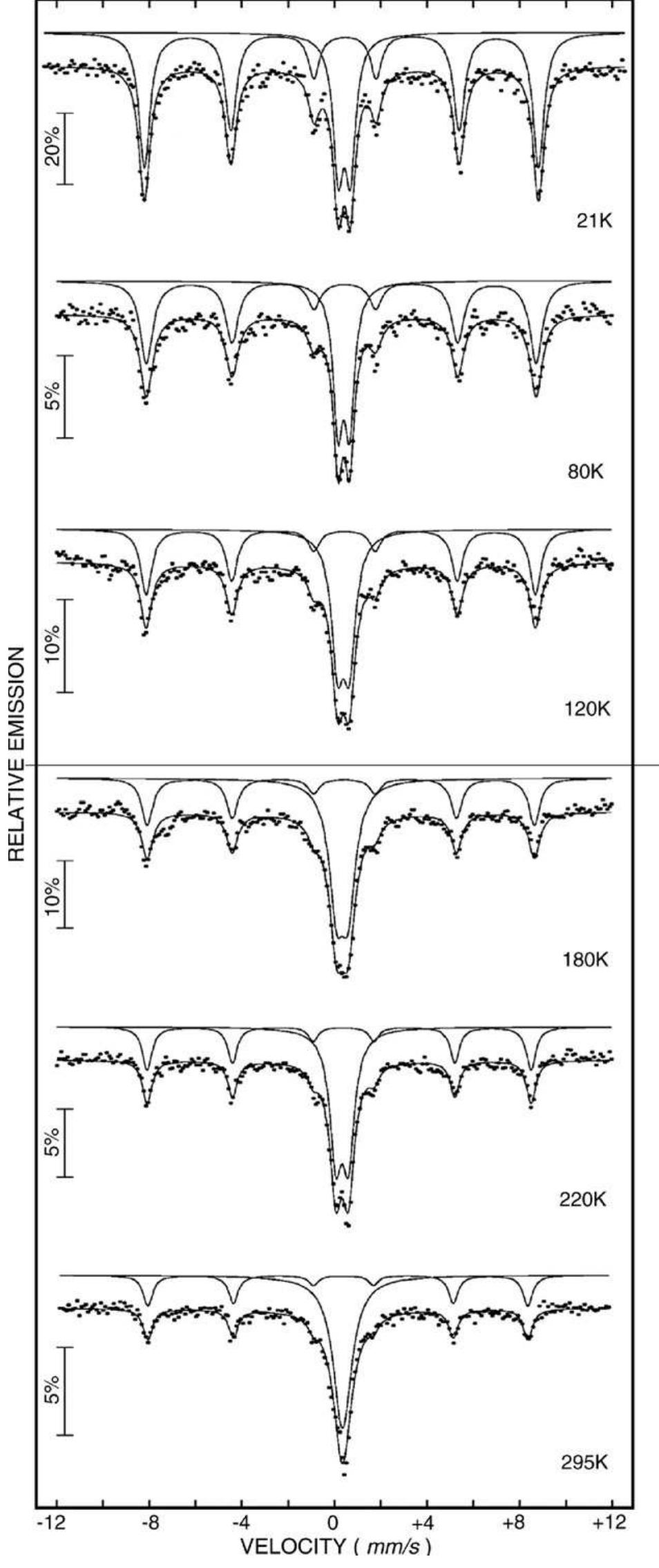
This study deals with the temperature and composition dependence of magnetization and magnetic anisotropy of Cu 2 þ -Cr 3 þ co-substituted magnesium ferrite, Mg 1 À x Cu x Cr x Fe 2 À x O 4 (x ¼0.0-0.5). The synthesized materials are... more
a b s t r a c t Nanostructured Cr/Co-Al coatings were deposited on Superni-718 substrate by DC/RF magnetron sputtering in the present work. Hot corrosion studies were conducted on bare substrate and the Cr/Co-Al coated Superni-718 after... more
Microwave technique was adopted for preparation of tin dioxide nanoparticles with particles size ranging from 10 to 11 nm within 10 min. The formation of monocrystalline SnO 2 nanoparticles was confirmed by the XRD (X-Ray Diffraction) and... more
Poly (l-aspartic acid-citric acid) green copolymers were developed using thermal polymerization of aspartic acid (ASP) and citric acid (CA) followed by direct bulk melt condensation technique. Antibacterial properties of copolymer of... more
A promising technique to develop polymer matrix composites by reinforcing carbon nanoparticles/whiskers through an extrusion process is described. Linear low density polyethylene (LLDPE) powder and carbon nanoparticles/whiskers are first... more
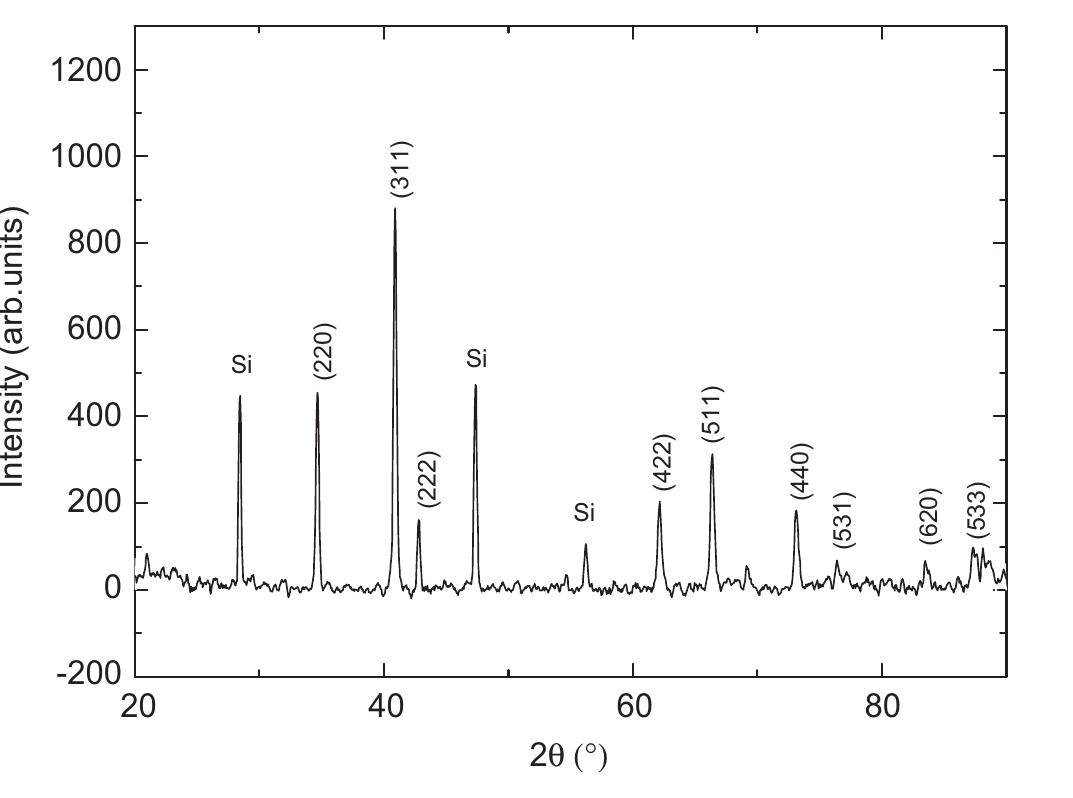
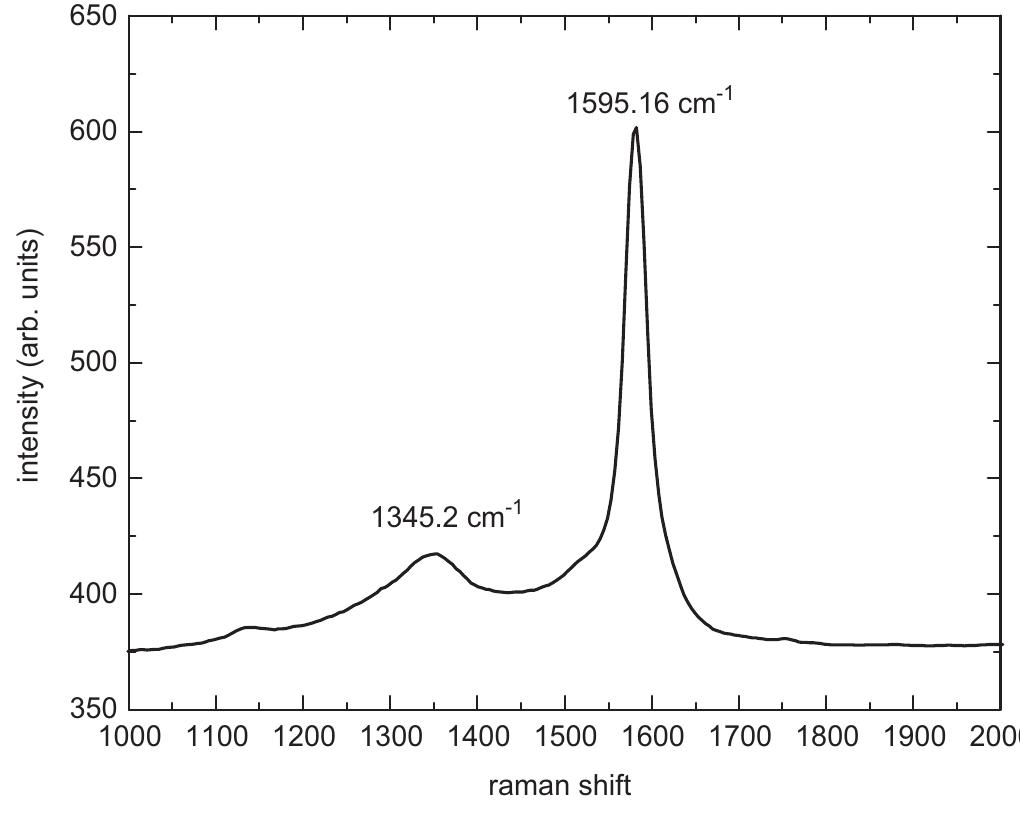
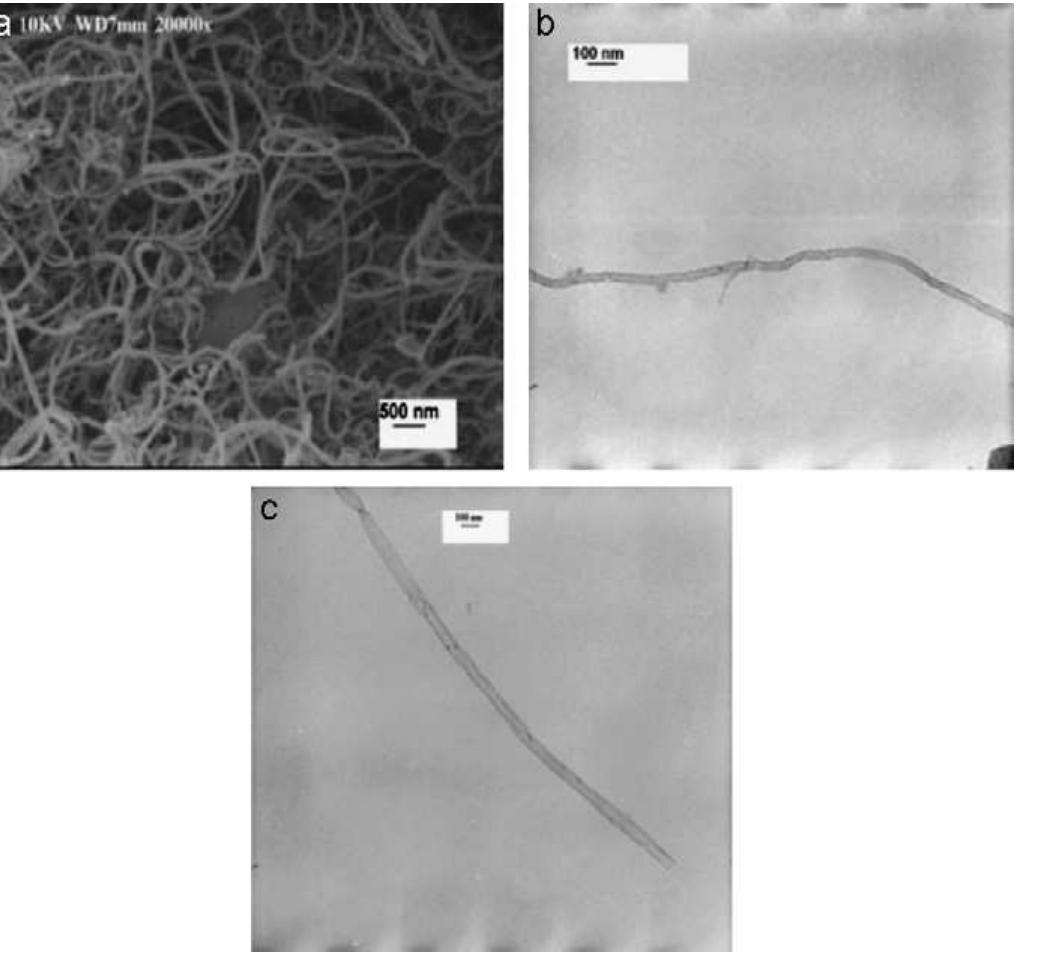
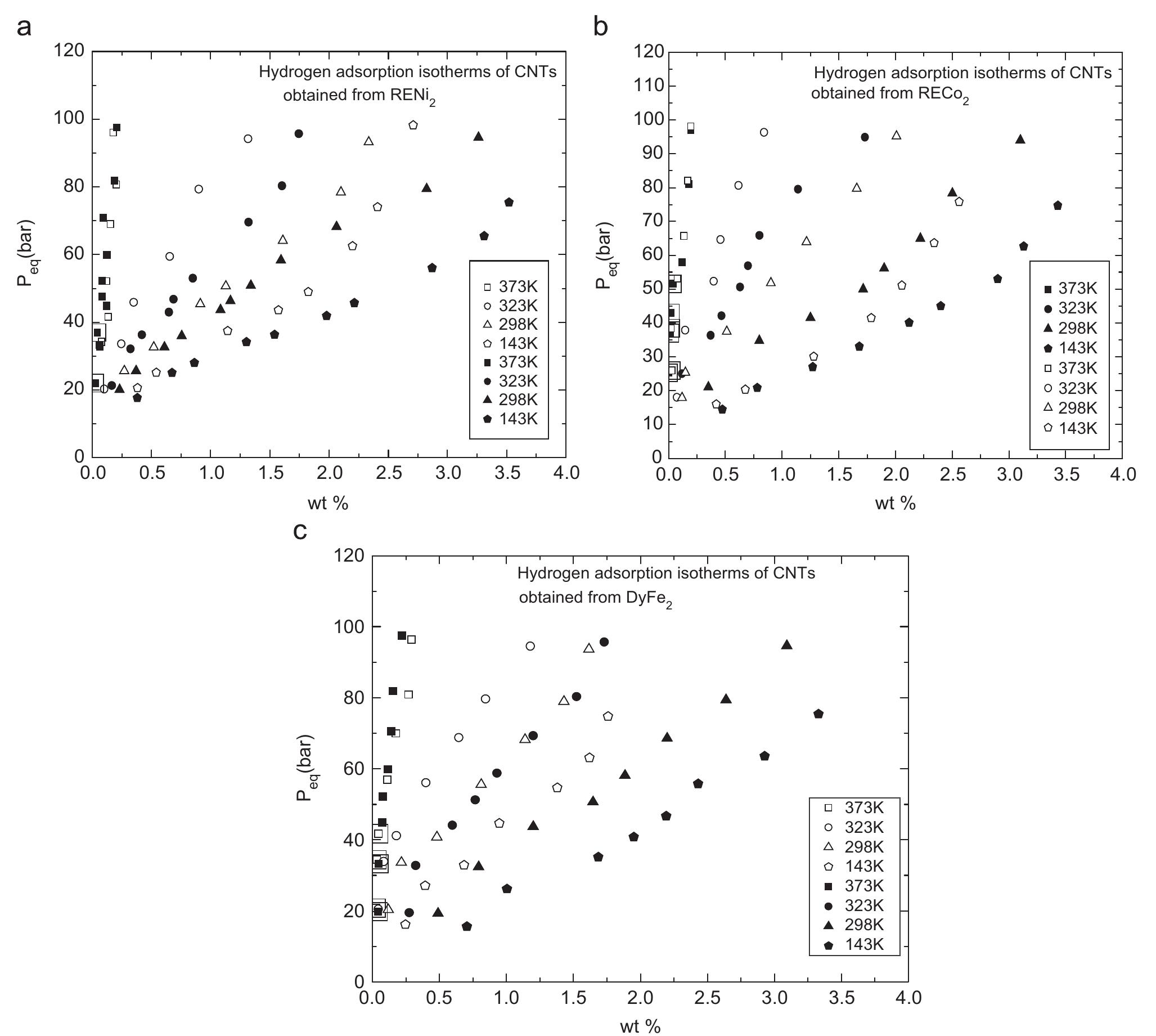
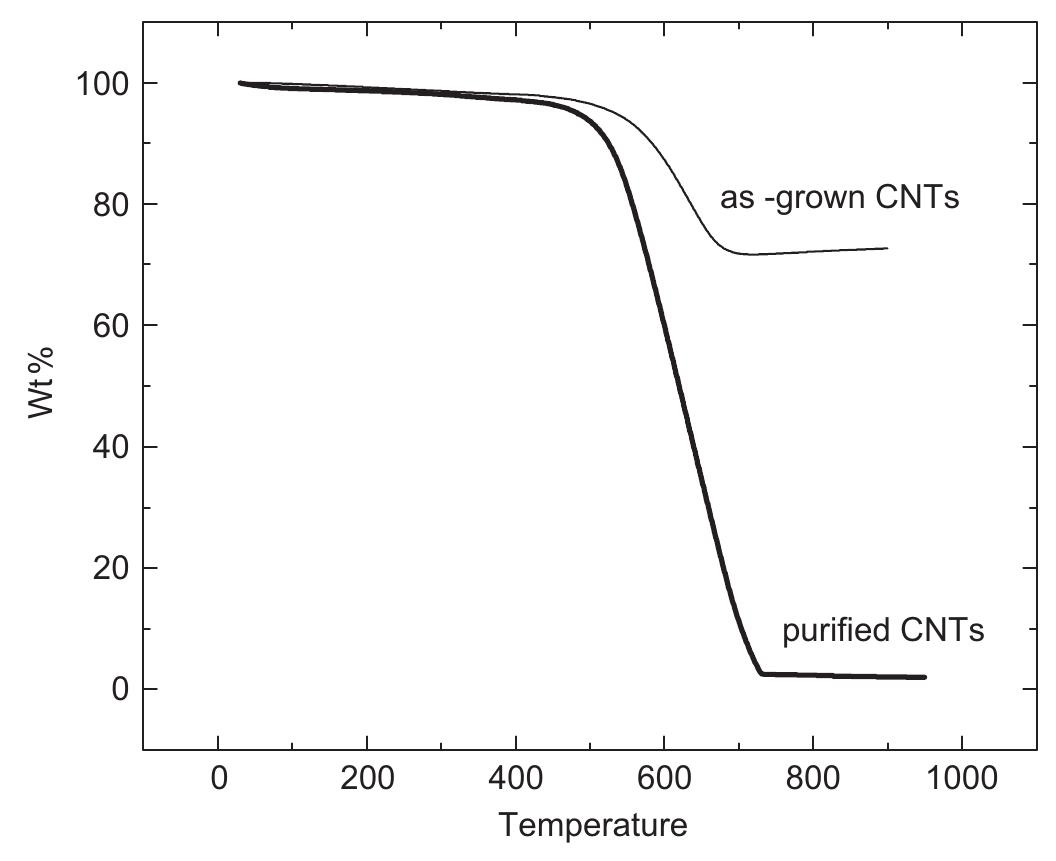
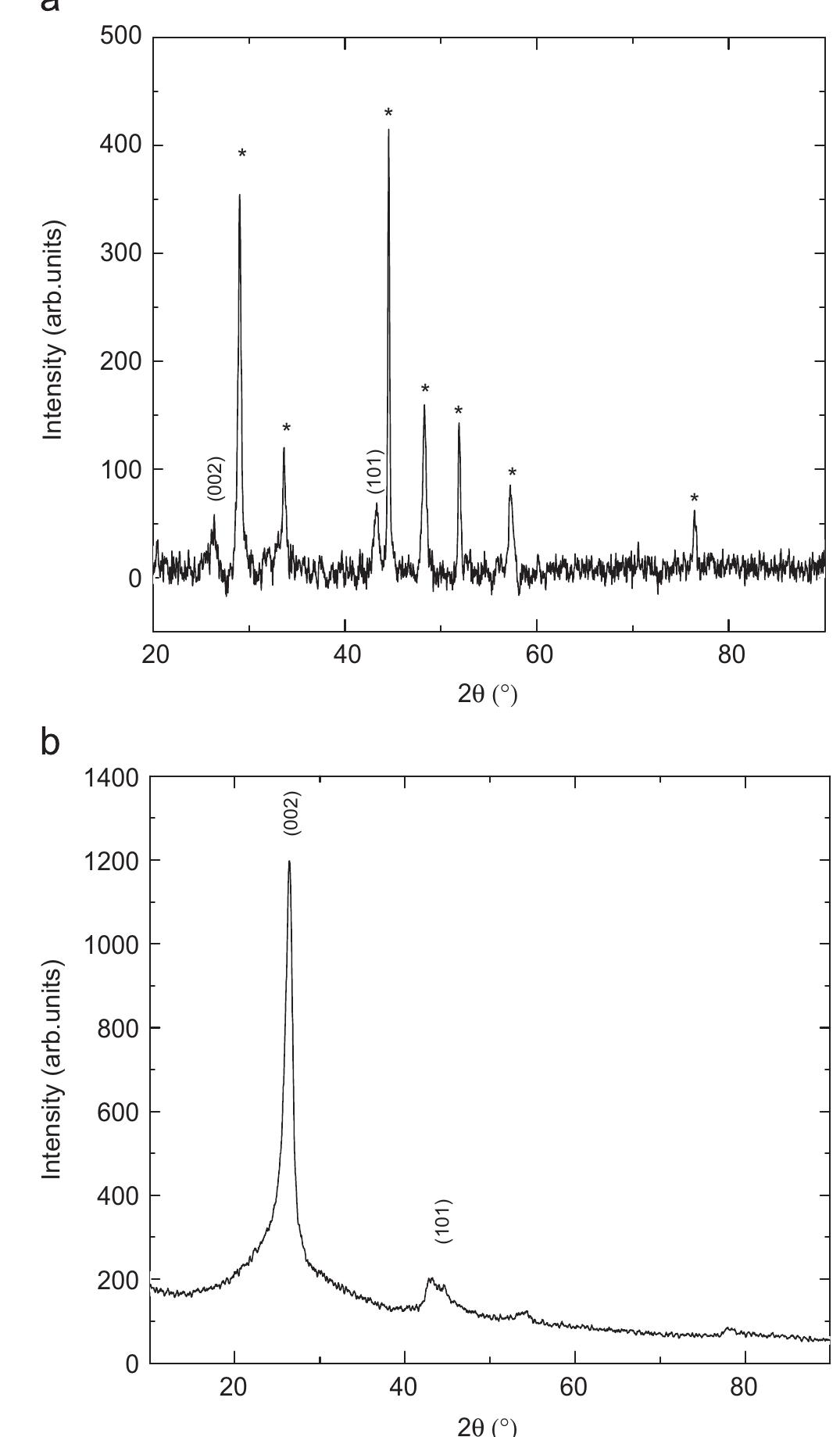
Multi-walled carbon nanotubes (MWNT) have been synthesized by chemical vapor decomposition of acetylene over rare-earth (RE) based AB2AB2 alloy hydride catalysts. The AB2AB2 alloy hydride catalysts have been prepared by hydrogen... more
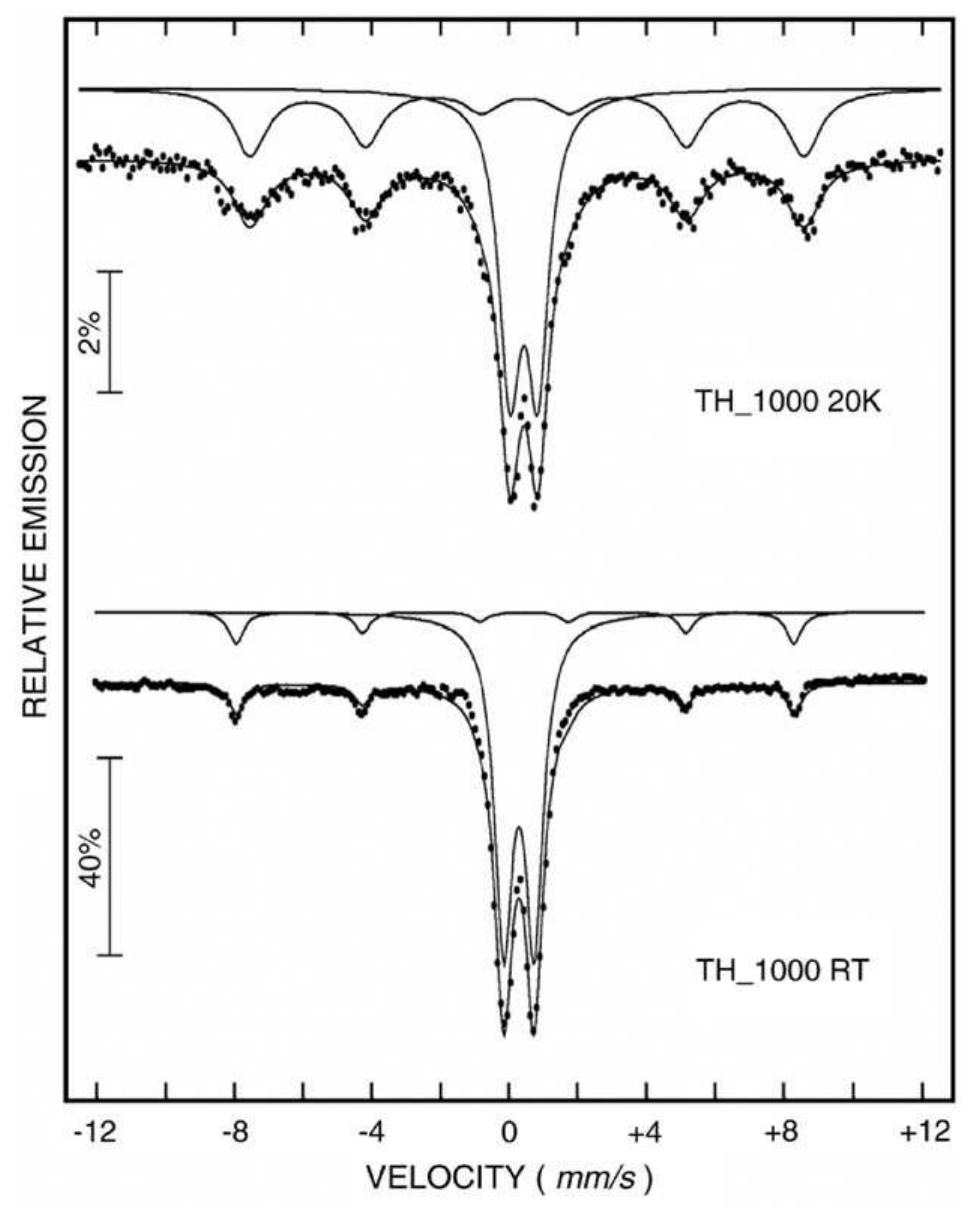
Fe x O y -SiO 2 nanocomposites were prepared by sol-gel method by using two SiO 2 sources and Fe(SO 4 ) 2 ·7H 2 O as raw materials. The amorphous gels were thermally treated up to 1000°C. The initial gel and the thermally treated samples... more
Quantum chemical calculations of molecular geometries, vibrational wavenumbers and thermodynamical properties of 3′-bromopropiophenone and 4′-bromo-3-chloropropiophenone were carried out using Hartree-Fock (HF) and density functional... more
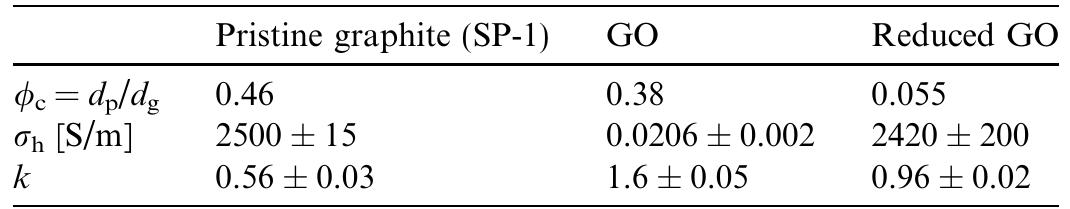
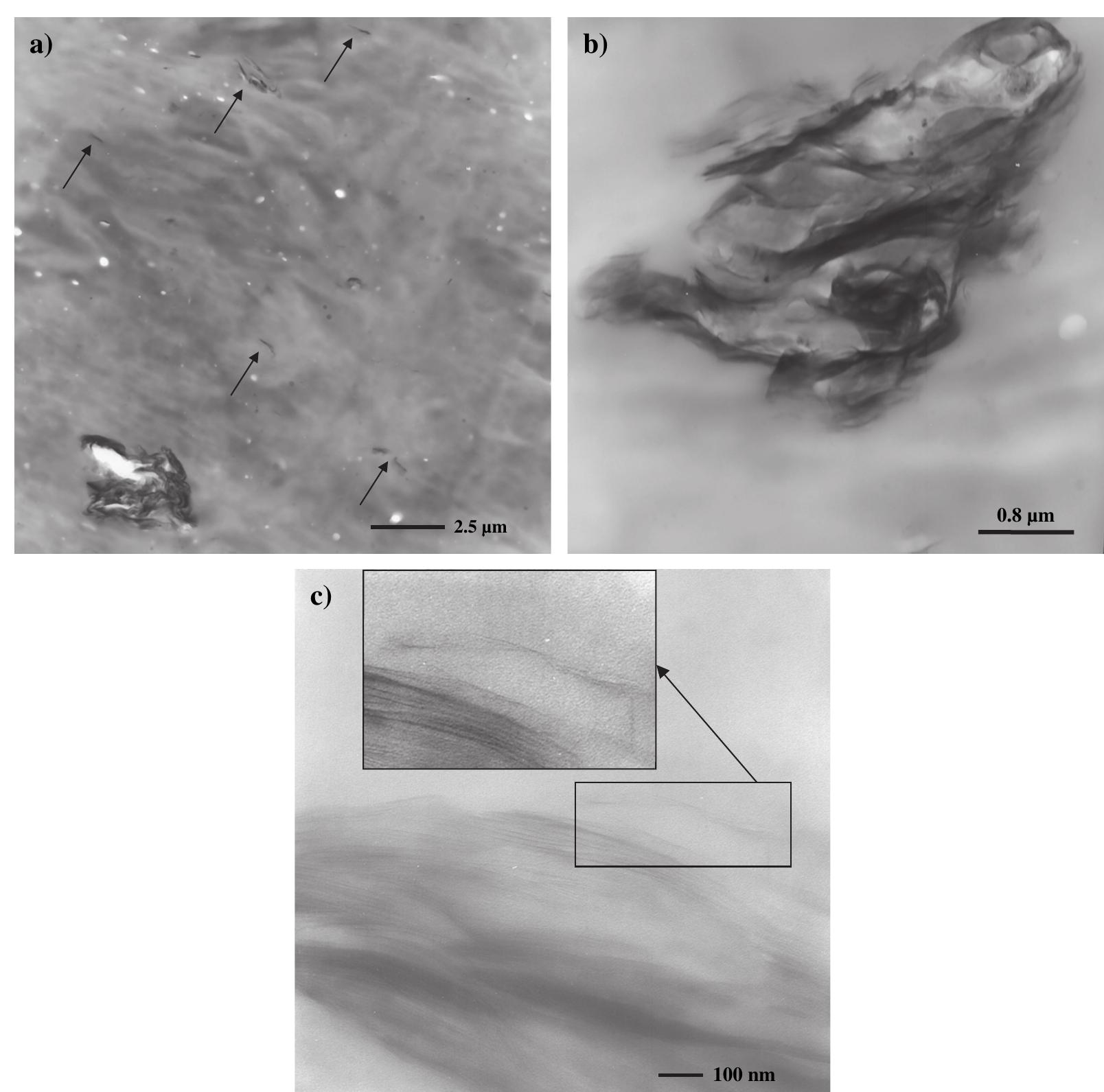
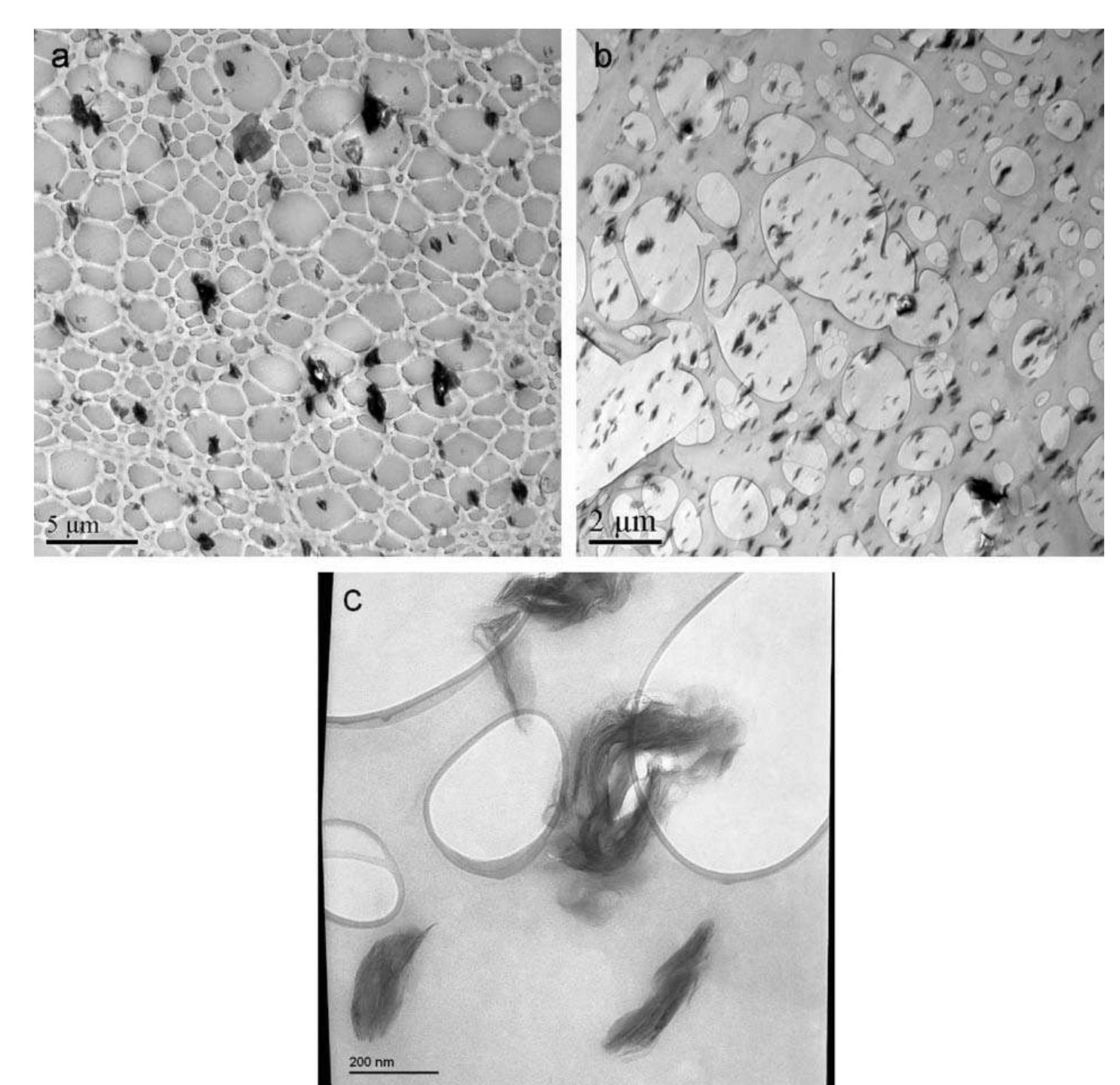
![Fig. 2. (a) An SEM image of aggregated reduced GO sheets. (b) A platelet having an upper bound thickness at a fold of ~2 nm. At the resolution limit of the FEG SEM used here, the following are relevant parameters of the microscope and of the specimen. Of the microscope: spot size and flux of e-beam (e-current, density, solid angle/aperture size), energy of the electrons in the primary beam, electromagnetic lens alignment, type of detectors used and their operating parameters. Of the sample: geometry (such as edges, roughness, thickness, and orientation with respect to e-beam), material (such as density, atomic number, electrical conductivity, and composition), substrate or matrix (what the specimen is affixed to or a part of). There is also the role of the operator expertise. Thus for (b) and discussion of a measured value for the fold thickness of about 2 nm, we suggest a confidence limit of roughly +1 nm. Surface area measurement of the reduced GO sheets via nitrogen gas absorption yielded a BET value of 466 m7/g (Fig. 3). This high specific surface area is partially an indi- cation of the degree of GO exfoliation prior to the reduc- tion. However, it is still lower than the theoretical specific surface area for completely exfoliated and isolated graph- ene sheets (~2,620 m7/g [20]), potentially due to the agglomeration of the graphene oxide sheets upon reduc- tion. While this agglomeration can result in the partial overlapping and coalescing of the reduced sheets and low- ers the surface area of the bulk materials, the crumpled Although GO is thermally unstable and starts to lose mass upon heating even below 100 °C, the major mass los: occurs at ~200°C, presumably due to pyrolysis of the labile oxygen-containing functional groups, yielding CO CO3;, and steam [27,44]. Hence, the thermal decompositior](https://figures.academia-assets.com/44579794/figure_003.jpg)
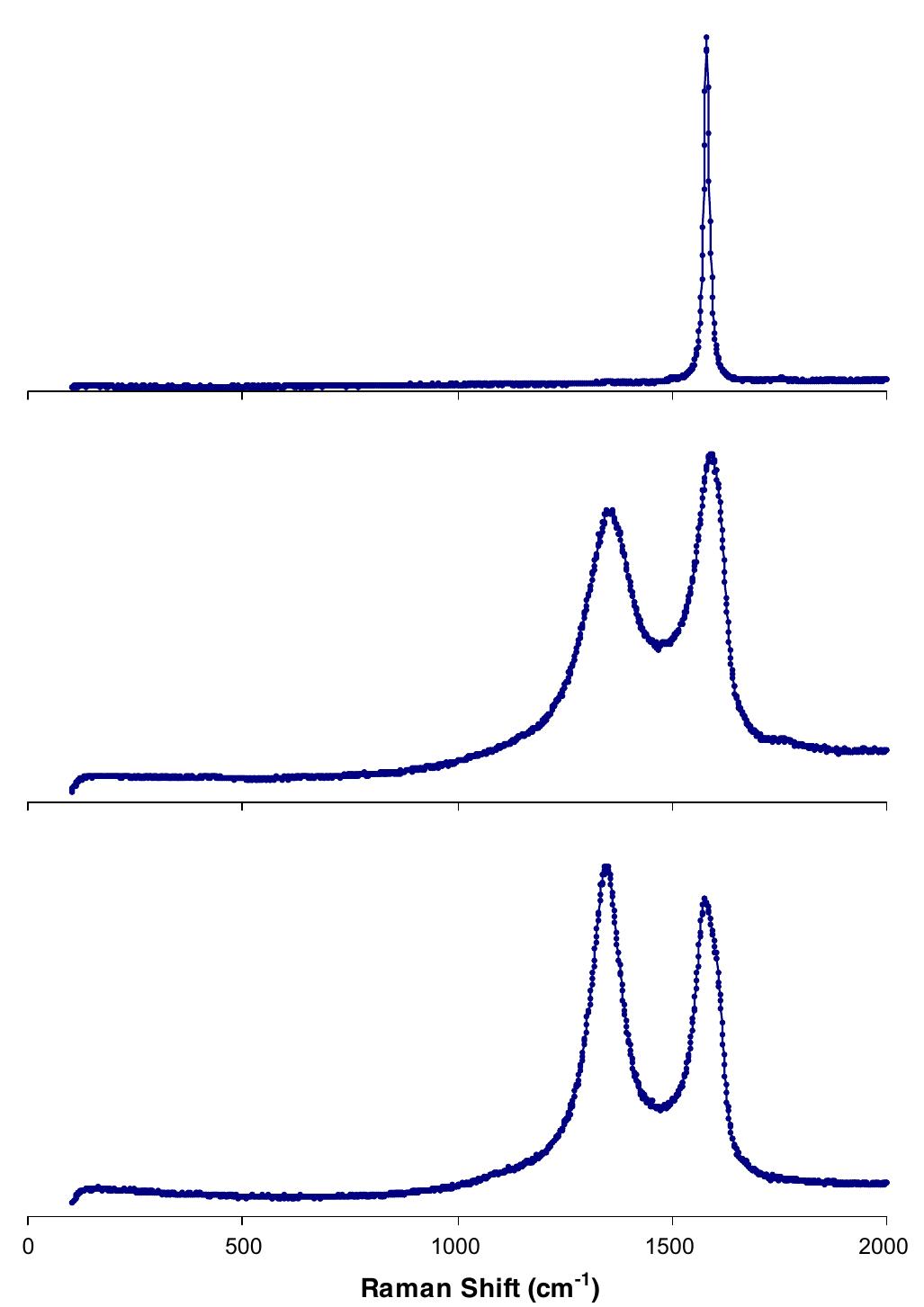
![Fig. 6. The Cls XPS spectra of: (a) GO, (b) reduced GO. Adapted from reference [18]. The significant structural changes occurring during the chemical processing from pristine graphite to GO, and then to the reduced GO, are also reflected in their Raman spec- tra (Fig. 7). The Raman spectrum of the pristine graphite, as expected, displays a prominent G peak as the only fea- ture at 1581 cm, corresponding to the first-order scatter- ing of the E2g mode [47]. In the Raman spectrum of GO, the G band is broadened and shifted to 1594 cm7!. In addi- tion, the D band at 1363 cm~' becomes prominent, indicat- ing the reduction in size of the in-plane sp* domains, possibly due to the extensive oxidation. The Raman spec- trum of the reduced GO also contains both G and D bands (at 1584 and 1352cm™', respectively); however, with an increased D/G intensity ratio compared to that in GO. This change suggests a decrease in the average size of the sp” domains upon reduction of the exfoliated GO [47], and can be explained if new graphitic domains were created that are smaller in size to the ones present in GO before reduction, but more numerous in number.](https://figures.academia-assets.com/44579794/figure_004.jpg)
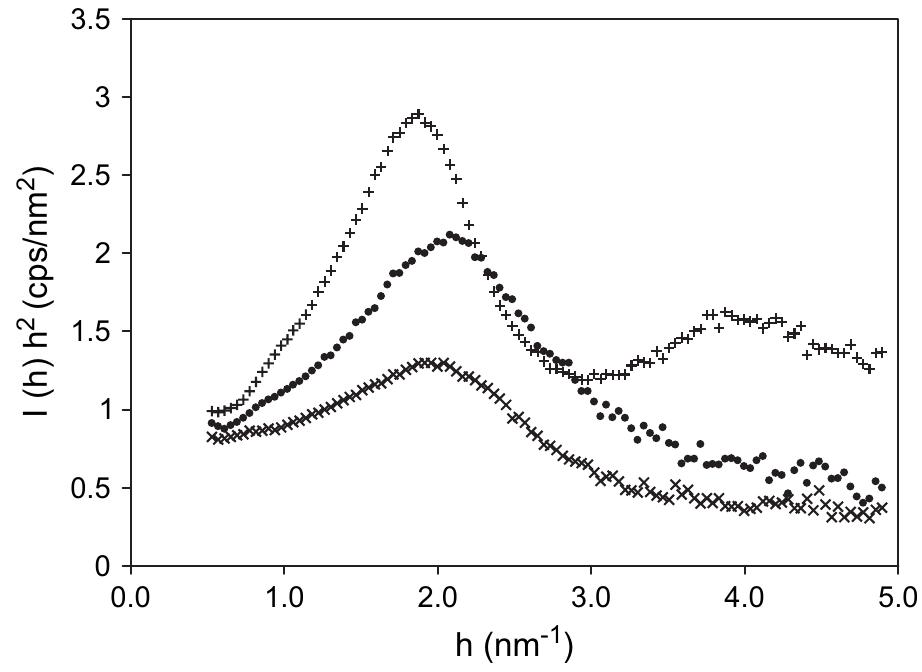
![Fig. 1. A non-contact mode AFM image of exfoliated GO sheets with three height profiles acquired in different locations During the hydrazine reduction of graphene oxide sheets dispersed in water, the brown-colored dispersion turns In our work, sufficiently dilute colloidal suspensions of GO prepared with the aid of ultrasound are clear, homog- enous, and stable indefinitely [18]. AFM images of GO](https://figures.academia-assets.com/44579794/figure_001.jpg)
![Fig. 3. Nitrogen adsorption and desorption isotherms for reduced exfoliated GO. The shape of the isotherm indicates that the material contains both micro and mesopores [53].](https://figures.academia-assets.com/44579794/figure_002.jpg)
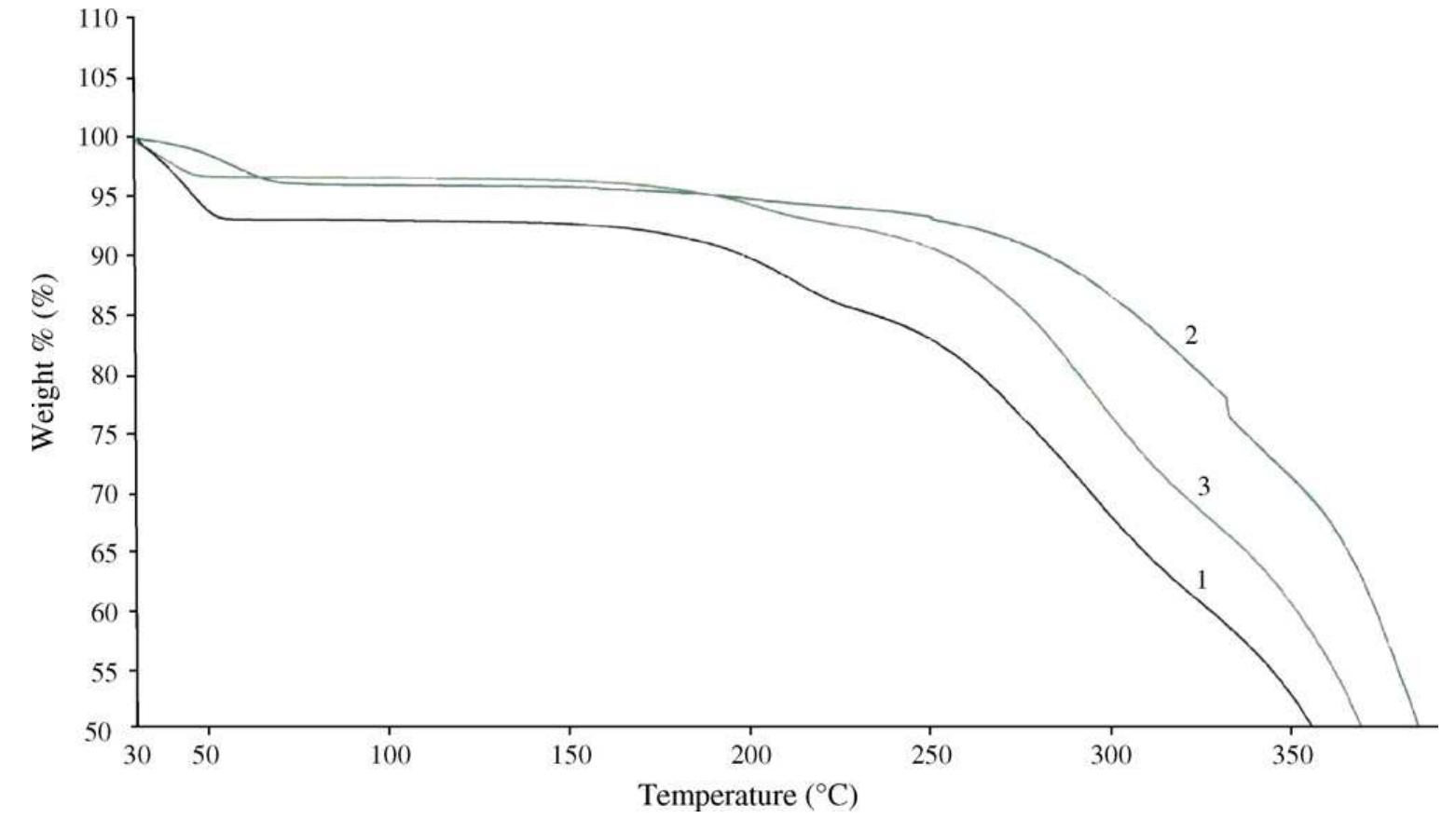
![The °C MAS NMR spectra of GO and the reduced GO indicate significant structural change induced by the reduc- tion. In the spectrum of GO (Fig. 5), the peaks at 57 and 68 ppm represent the '°C nuclei in the epoxide and hydro- xyl groups, respectively [24-27]. The resonance at 130 ppm belongs to the un-oxidized sp” carbons of the graphene net- work and that at 188 ppm presumably arises from the car- bonyl groups. In the ‘°C spectrum of the reduced GO, the peaks from the oxygenated and the carbonyl carbons are absent. The remaining prominent feature is the resonance at 117 ppm that is broadened by chemical shift distribu- tion and corresponds to variations of carbon atom environments. Fig. 4. Normalized TGA plots for GO and the reduced GO. The downward slopes are due to normal instrument drift.](https://figures.academia-assets.com/44579794/figure_005.jpg)
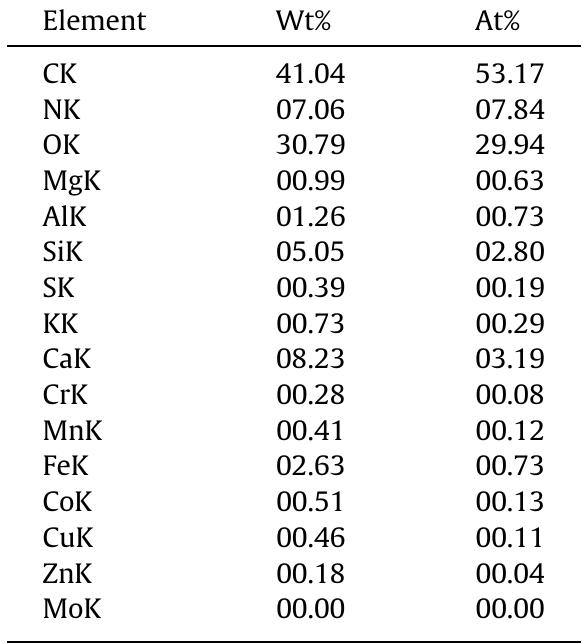
![General composition of pressmud [5]. Table 1](https://figures.academia-assets.com/44051706/table_001.jpg)
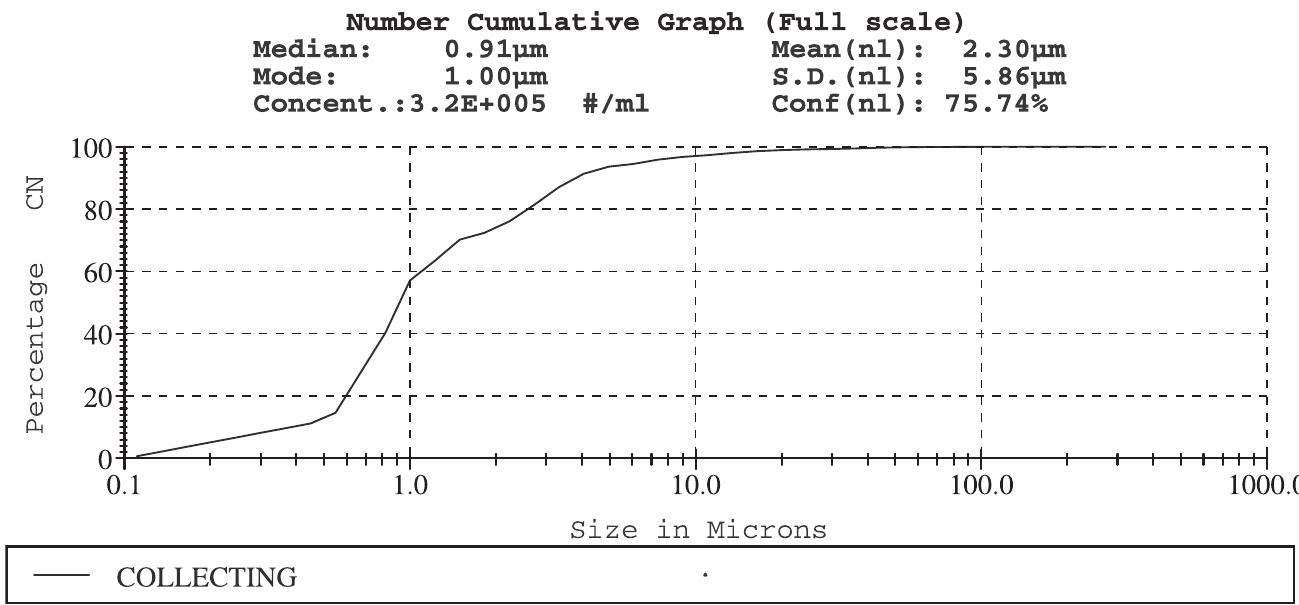

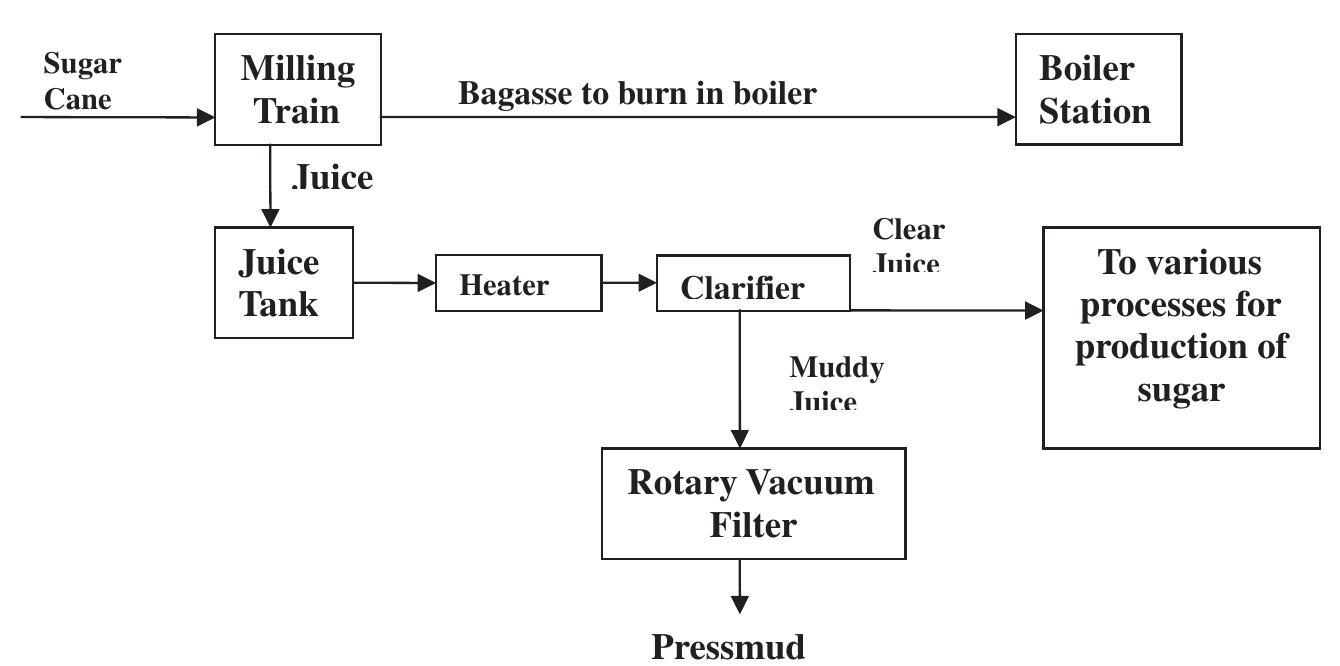
![A typical nutrient contents of pressmud [6]. Table 2](https://figures.academia-assets.com/44051706/table_002.jpg)
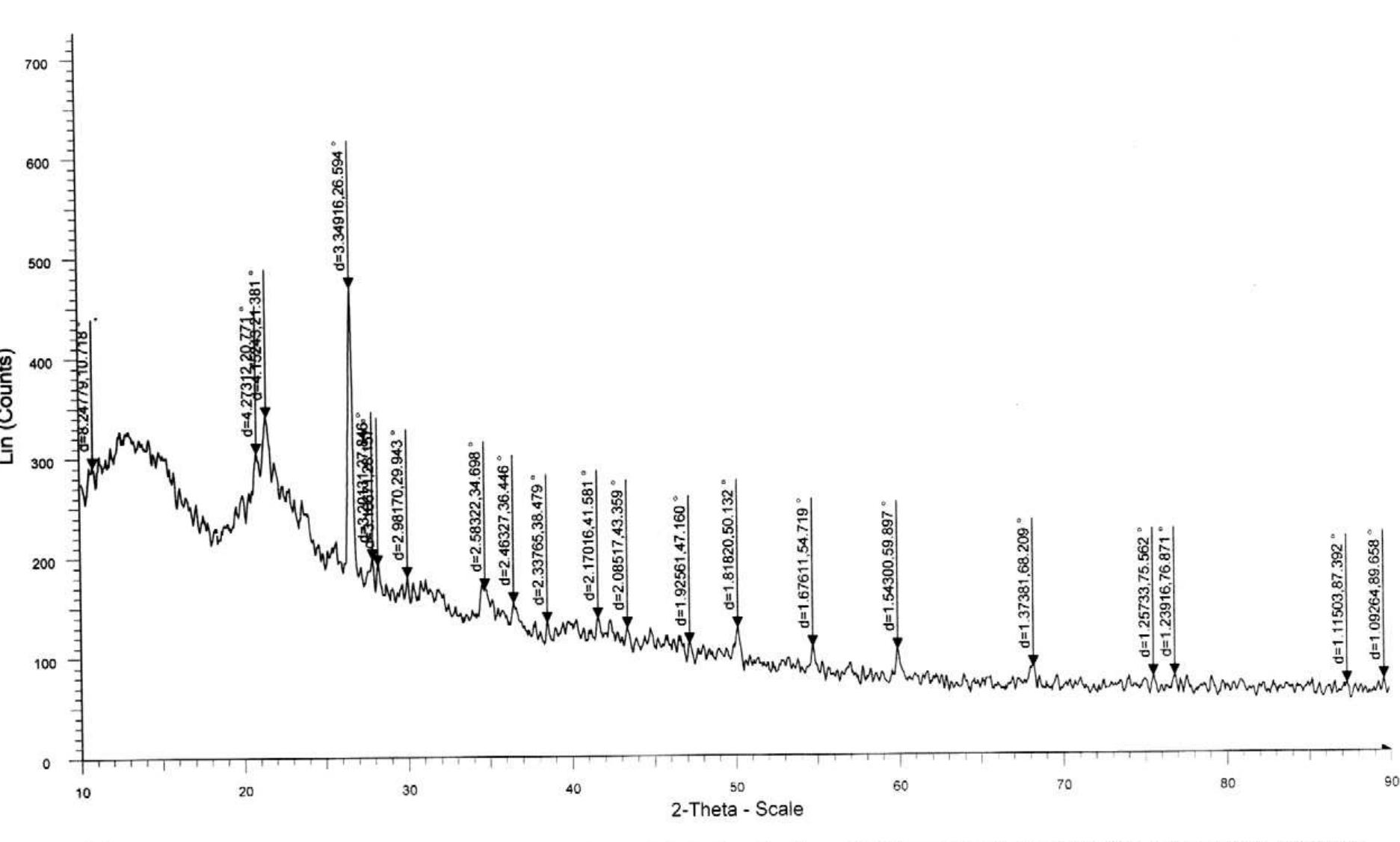










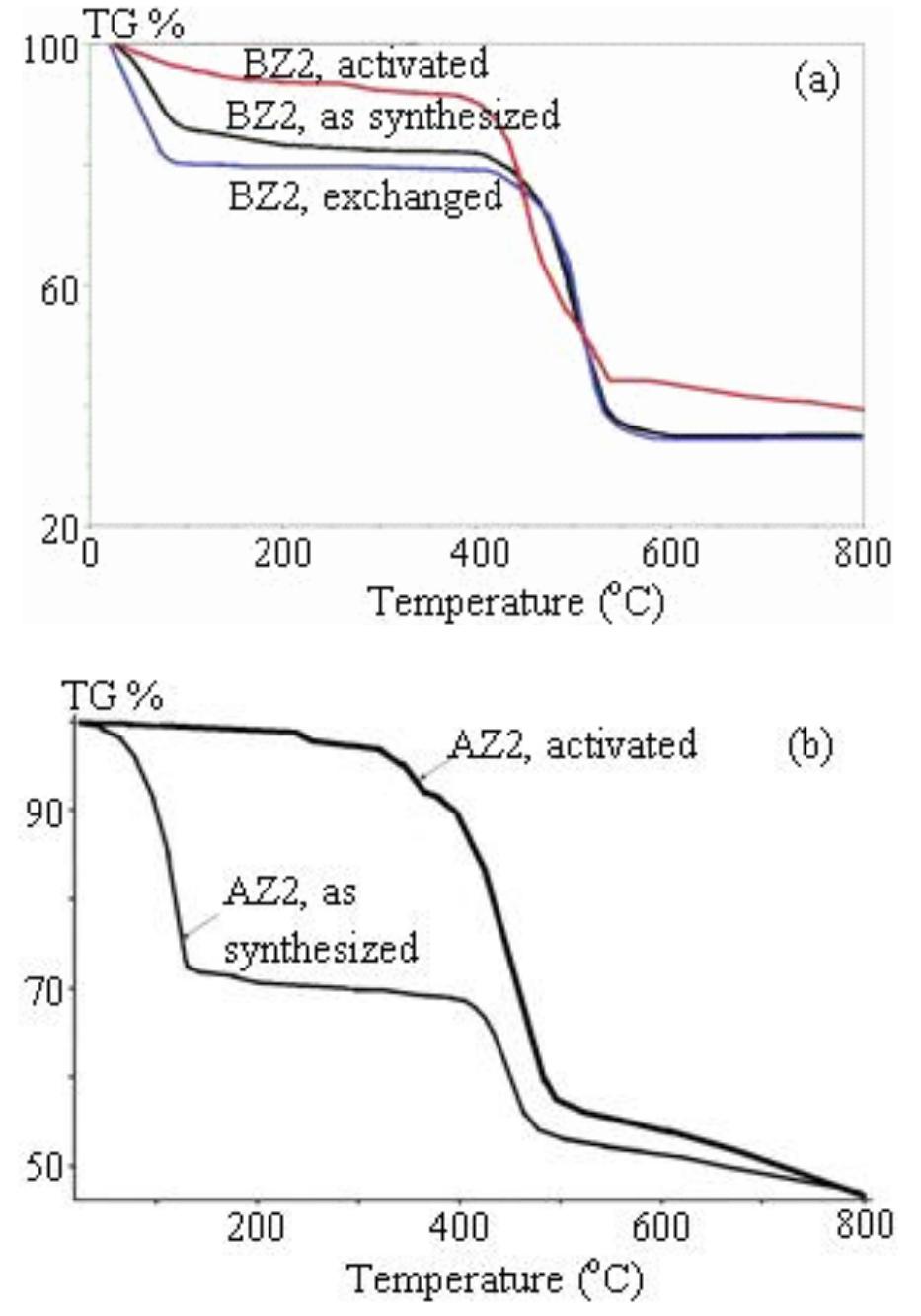
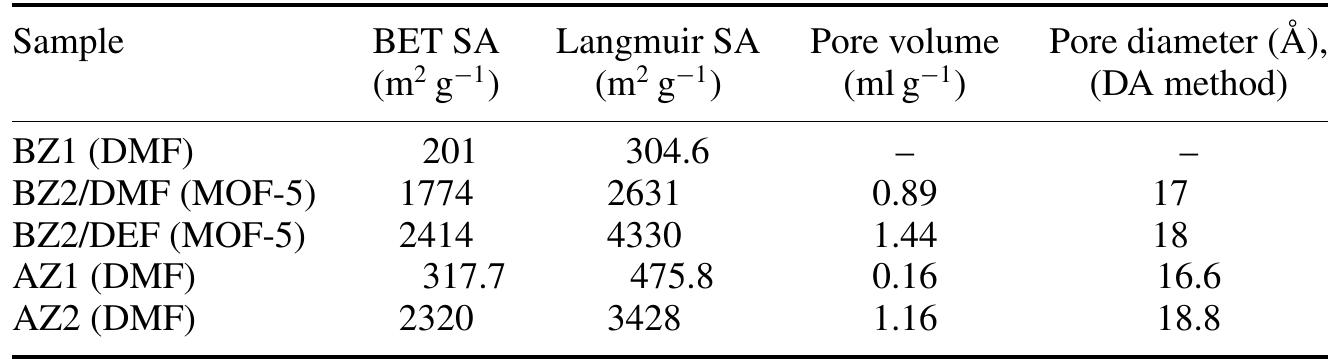
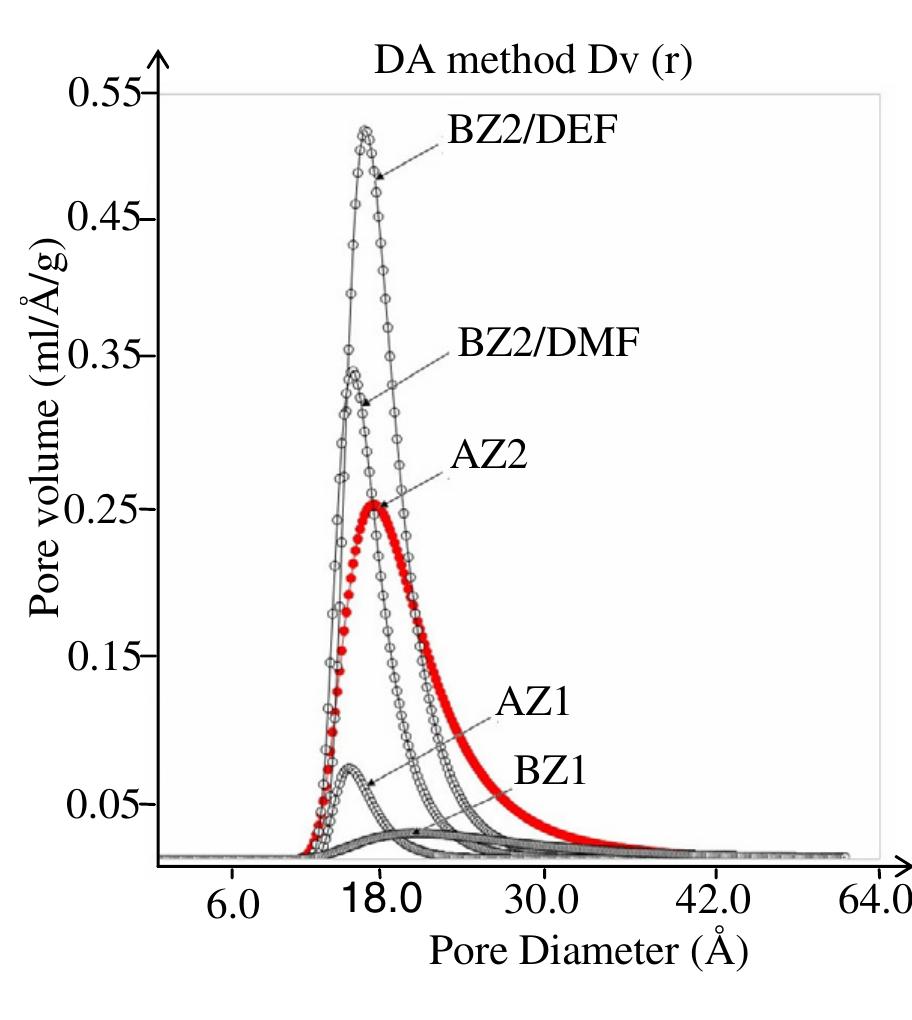
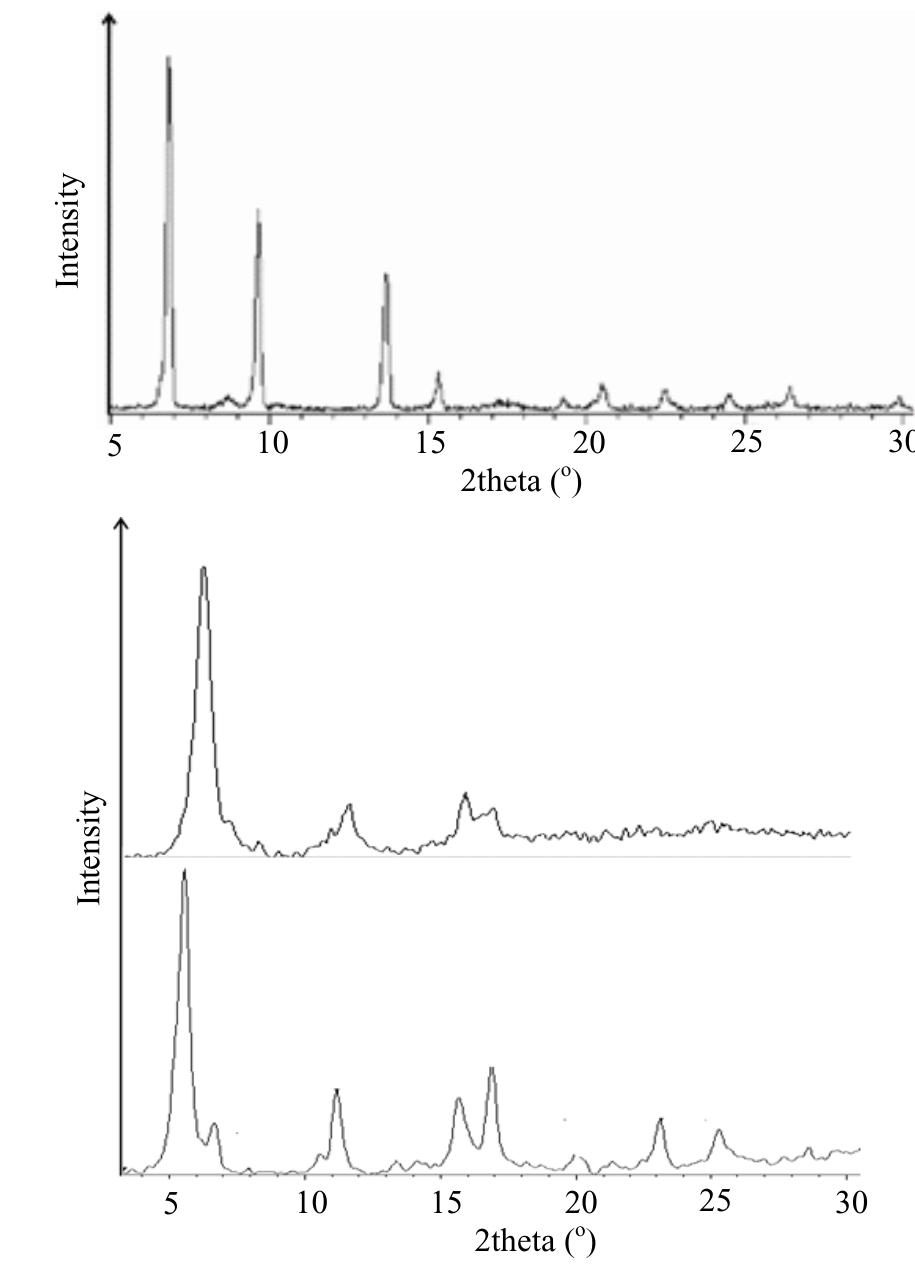
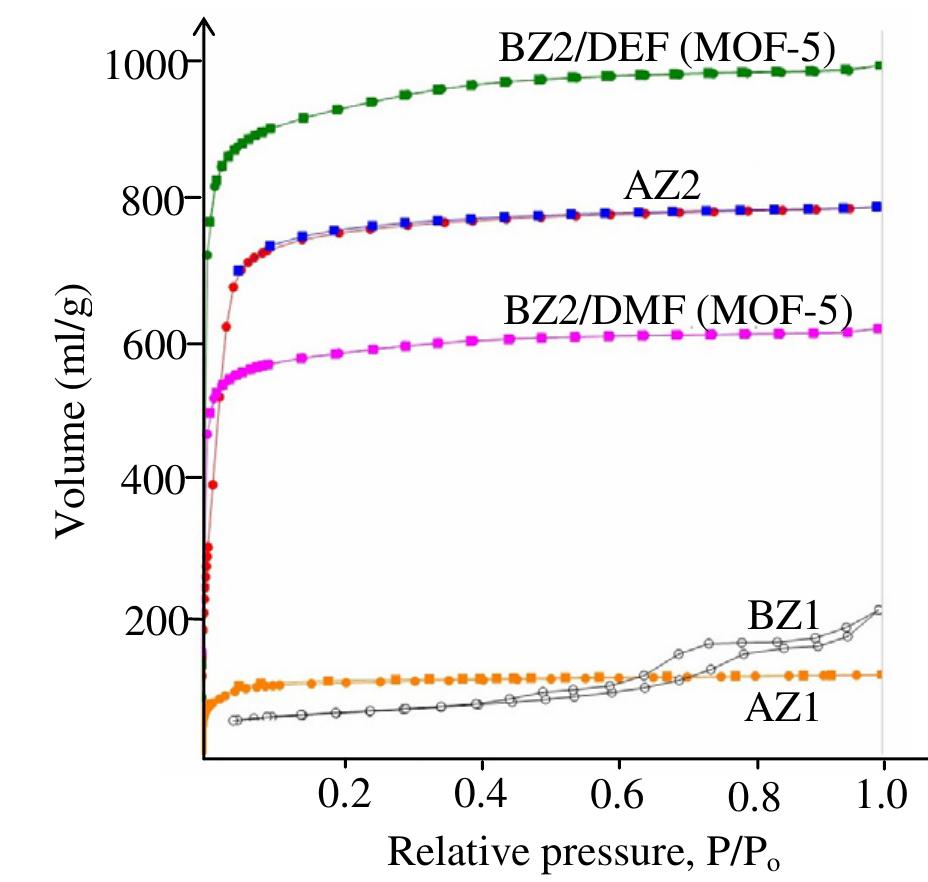
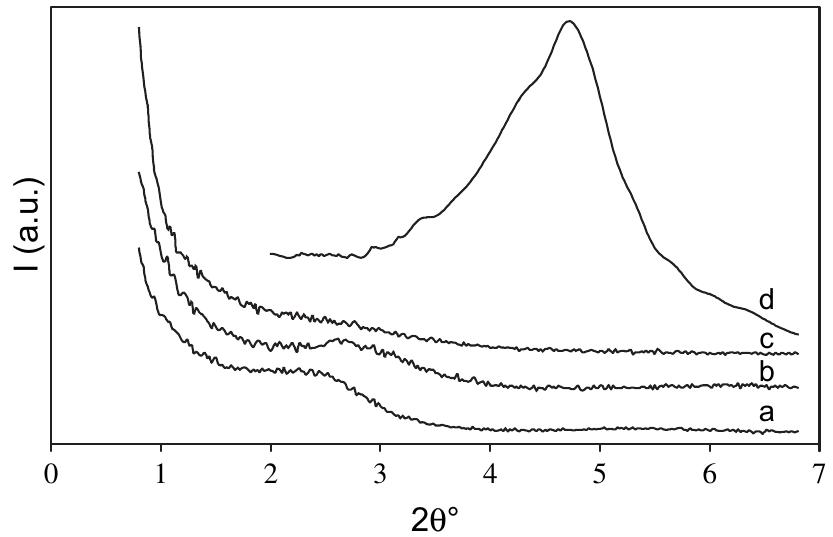
![Figure 1. Microscope images of crystalline materials synthesized in DMF at 80°C, 12h, pH 4.18, [H»BDC] : Zn?* = 1: 1, [H;BDC] = 0.1 M (a, BZ1), in DMF at 100°C, 24h, pH 4.82, [H,BDC] : Zn?* = 1 : 4, [H] BDC] = 0.01 M (b, BZ2/DMP), in DEF at 100°C, 24h, pH 4.64, [H)BDC] : Zn?* = 1 : 4, [HyBDC] = 0.01 M (c, BZ2/DEP). 2.2.2. Synthesis of MOFs from azobenzene-1,4-dicarboxylic acid and zinc nitrate. The mixtures with mole ratios of H,ABD to zinc nitrate (H,ABD: Zn) varying from 1:1 to 1:5 were dissolved in 10 ml DMF. In each experiment with a certain H»ABD : Zn mole ratio, the concentration of HxABD was varied from 0.01 M to 0.001 M. The reaction mixture was put in a 20 ml reaction vial, which was capped and heated in an isothermal oven from 80°C to 100°C for 12—72 h, and then cooled to room temperature. The red sheet-shaped crystals were collected by decanting from mother liquor. The washing and solvent exchange procedures were the same as described above in section 2.2.1. Finally, the crystals were dried under vacuum at 150°C for 10h. XA-Tay diffraction data were recorded on a Do Advance Bruker powder diffractometer with Cu-Ka radiation (A = 1.542 A). The thermal behavior of synthesized materials was recorded on a TGA Q500 analyzer with a heating rate of 10°C min! in nitrogen. Nitrogen physisorption measurements were carried out on a Quantachrome Autosorb-1C system. Prior to the measurement, the sample was outgassed at 150—200°C for 10h.](https://figures.academia-assets.com/46371533/figure_001.jpg)
![Figure 2. Microscope images of crystalline materials synthesized in DMF at 90°C, [H>ABD]: Zn** = 1 : 1, [H,ABD] =0.01M (a, AZ1), at 85°C, [H,ABD]: Zn** = 2: 5, [H; ABD] = 0.004M (b, AZ2). 3.1.2. MOFs from azobenzene-1,4-dicarboxylic acid and zinc nitrate. The products of the reaction of zinc nitrate and H,ABD in DMF were coded as AZ1 and AZ2. Compound AZI was formed at equivalent HyABD: Zn mole ratio and 0.010M concentration of H2ABD (figure 2(a)). When this concentration was reduced to a very low value of 0.004M and H,ABD : Zn mole ratio was decreased to 2 : 5, the product AZ2 was obtained.](https://figures.academia-assets.com/46371533/figure_002.jpg)
![(aromatic peaks)[r, s, q] while C-NMR (Fig. 4) spectra of pure PUR-1 shows the spectral peaks at 149-152 ppm (>C=O urethane and free —NCO)[q, n], 127-132 ppm (aromatic ring carbons)[r, s, t, u, v, w], 65-69 ppm (-CH,—O and —CH-O-)[a’, p] and 18.5 ppm (CH; attached to aro- matic ring)[x]. These spectral bands are the characteristic bands of TDI based polyurethanes [13, 17]. Closer exami- nation of 'H-NMR spectra of HERA and PUR-1 reveals that pure PUR-1 shows suppressed characteristic peaks of -OH, —CH-OH and CH,—OH compared to HERA (sect. 2.1) at 5.26 ppm [-CH(OH)], 4.85 ppm and (—CH,—OH), 3.59 ppm [CH(OH)], 3.56 and 3.49 ppm (-CH2—OH), while](https://figures.academia-assets.com/49819660/figure_002.jpg)
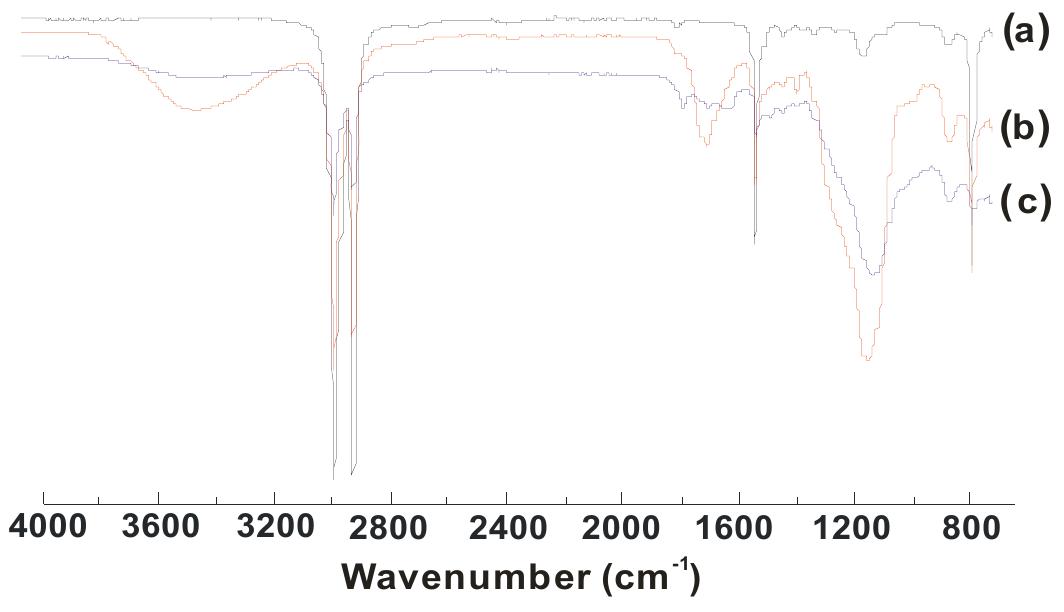
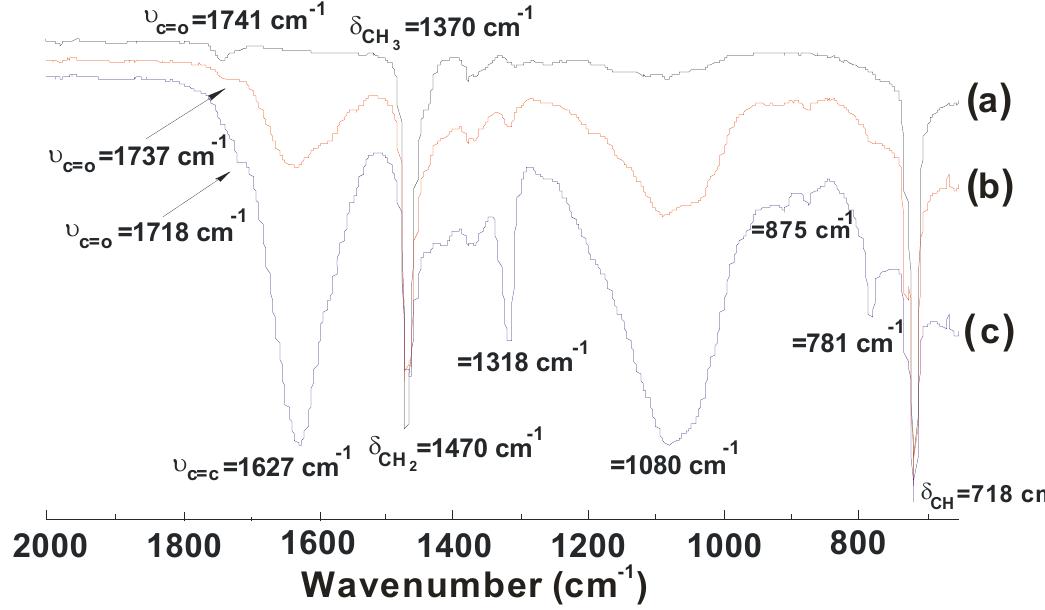
![1,241, 1,164 and 1,097 cm!) of RC oil disappeared with the appearance of new bands at 1,622, 1,078.9, 1,052.9 cm~' for amide carbonyl, primary and secondary alcoholic groups (C—O stretching), respectively, as well as the presence of a broad band at 3,369 cm! instead of 3,422.1 cm! confirming the amidation of RC oil. In PUR spectra, the hydroxyl band of HERA (3,369.3 cm‘) diminished and was centered at 3,290.9 cm~'. The presence of new bands at 3,339 cm! (N-H stretching of urethane linkages), 1,716 cm~' (urethane carbonyl stretching), 1,228 cm”! (NCOO- stretching), 2,273 (free NCO stretch- ing), and 697 (N-H of urethane) [13, 16, 17] as well as bands at 3,069, 1,605.5, and 744.5 for aromatic ring of TDI confirm the urethanation of HERA. 'H-NMR (Fig. 3) spectra of pure PUR-1 shows the characteristic peaks at 4.01-4.43 ppm {m, —CH O C (=O)NH- and —CH, O C (=O)NH-, urethane} [n, p], 2.2 ppm (s, —CH3; of TDDJ[t], and 6.9-7.38 ppm requirement of polyurethanes, i.e., to prepare high or low molecular weight polyurethane. In this case, at each ratio, we have an excess of hydroxyl groups (Table 1), which based on general principles of polyaddition reactions lead to low molecular weight PUR, which may find applications as coating and adhesive materials [14]. It is worth mentioning that the urethanation reaction was carried out by using a “one-shot” technique. In this method, all the ingredients (HERA and TDI) are mixed simulta- neously and the resulting mixture is allowed to polymerize directly. The advantage of using this method is that no intermediate prepolymer preparation is necessary [15]. FTIR spectra (Fig. 2): It is observed in the spectra of HERA that the characteristic bands of ester carbonyl (at 1,745,](https://figures.academia-assets.com/49819660/figure_001.jpg)
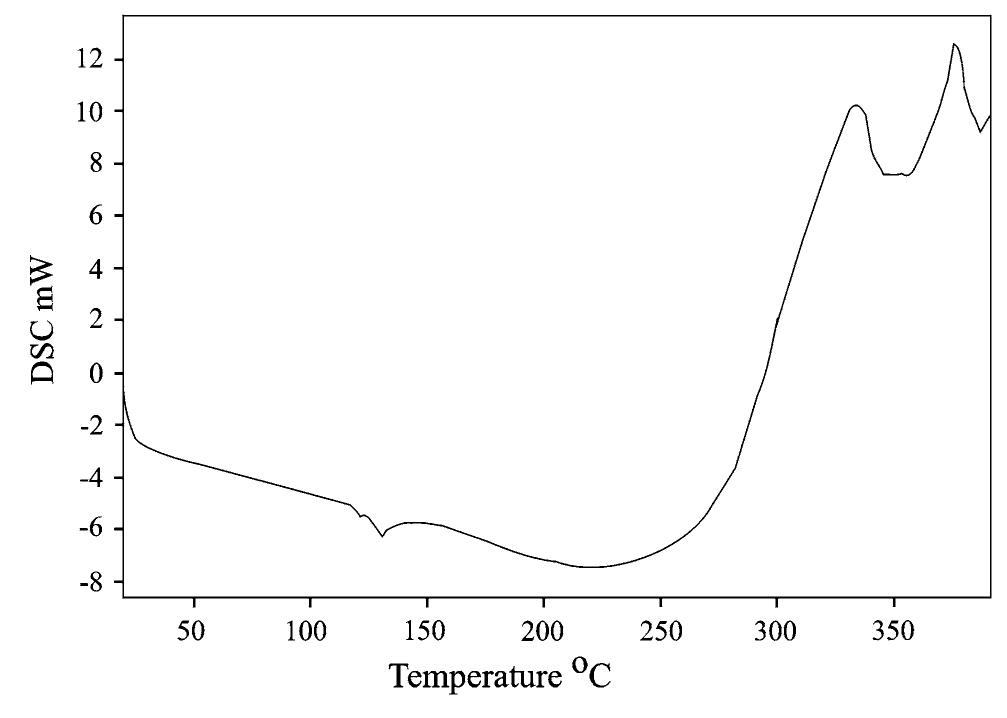
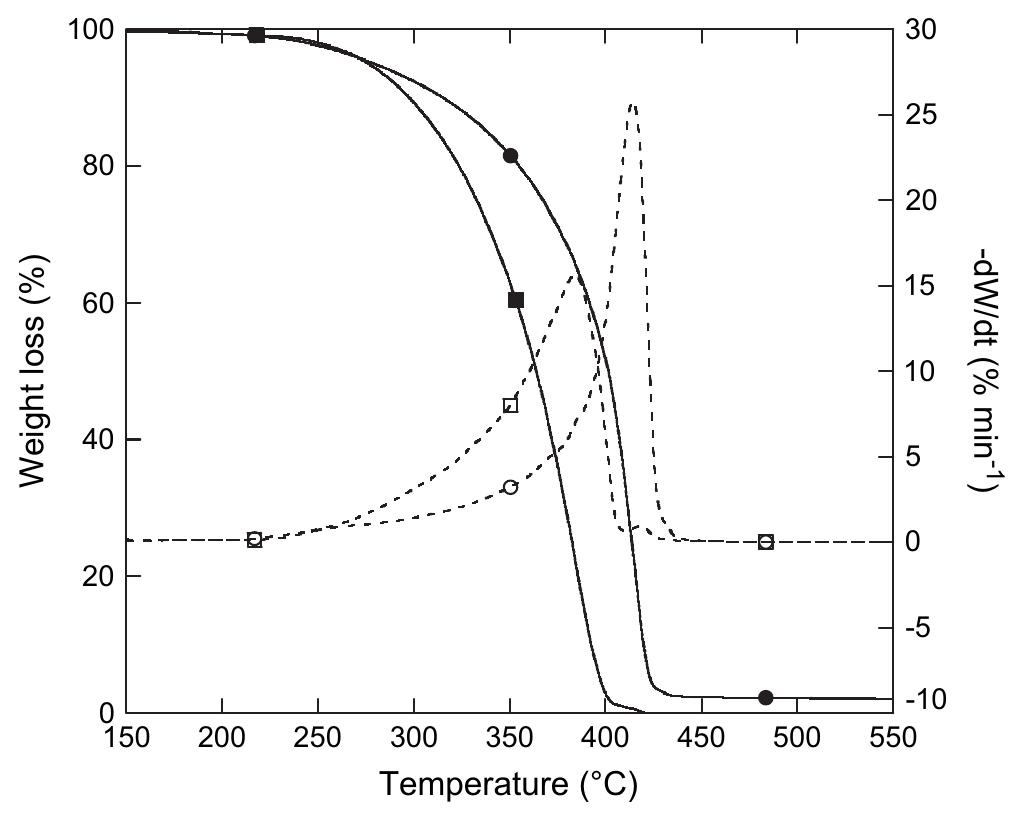
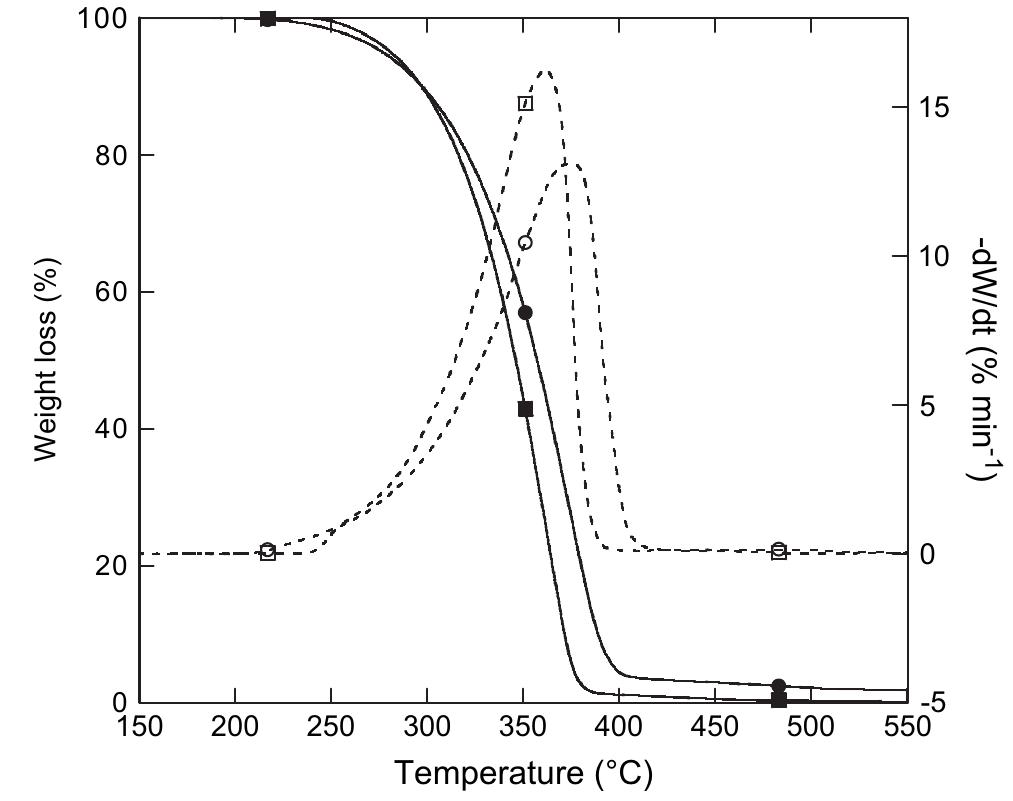
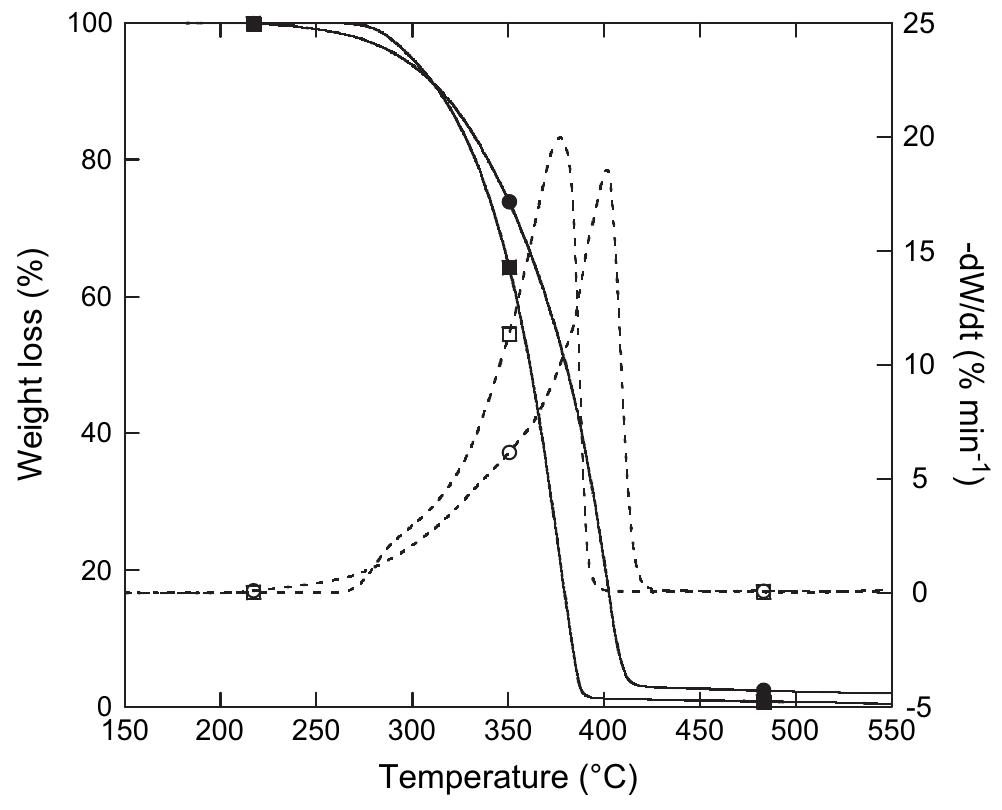
![The thermal stability of PUR-1 is shown in Fig. 5. It reveals that the degradation of PUR-1 was observed in the range of 150-390 °C. This may be correlated to the decomposition of urethane bonds followed by the volatilization of the decomposition products. It is reported that polyurethanes generally exhibit relatively low thermal stability due to the presence of labile urethane groups. The onset of this bond dissociation occurs around <300 °C through the dissocia- tion of isocyanate and alcohol, the formation of primary amine and olefins, or the formation of secondary amines that result in the loss of CO, from the urethane bond, depending upon the type of isocyanate and polyol employed [18]. The decomposition of the urethane bond in PUR-1 takes place at quite high temperatures due to any allo- phanate and biuret linkages formed during the curing reaction, they reopen quite readily on heating at a relatively high temperature as they are most thermolabile [18]. The degradation in the range of 390-410 °C may be correlated to the decomposition of the amide bond, aromatic ring and aliphatic alkyl chain of the fatty acid followed by the vol- atilization of the decomposition products, respectively. The polyol used in this study is based on HERA. So, the main chain cleavage may result in the formation of 16-undeca- noic acid and heptanal as evident by the thermal degrada- tion of ricinoleic acid [19]. Derivative TGA curves (DTG) of PUR-1 shows almost single step thermal degradation, which matches with TGA of the same. This step incurs about 85 wt% loss correlated to a cross-linked, homogenous](https://figures.academia-assets.com/49819660/figure_004.jpg)
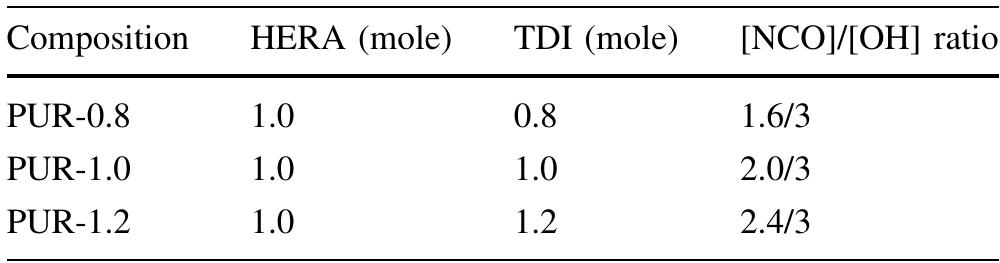
![Table 2, Physico-chemical tests of HERA and different compositions of PUR TDI attributed to the inclusion of increased urethane con- tent in the polymer backbone. The solubility test reveals that all PUR compositions (before curing) are completely soluble (100%) in EMK, chloroform, carbon tetrachloride, DMF (Dimethylformam- ide), DMSO (Dimethy] sulfoxide), THF (Tetrahydrofuran) and diethyl ether while they have 20% solubility in methyl alcohol, ethyl alcohol, and insoluble in water. The higher solubility of these resins in polar and non-polar solvents except H,O and alcohol is due to the presence of long fatty acid hydrocarbon chains and polar groups of the resin.](https://figures.academia-assets.com/49819660/table_002.jpg)
![The data for physico-chemical properties of HERA and different compositions of PUR are given in Table 2. It reveals that the percent hydroxy] content of PUR decreases gradually from PUR-0.8 to PUR-1.2, that is, with increases in the amount of TDI. The trend can be attributed to the consumption of hydroxyl groups of HERA during uretha- nation reaction with TDI. The specific gravity and refrac- tive index of PUR increase with the increased content of](https://figures.academia-assets.com/49819660/figure_003.jpg)
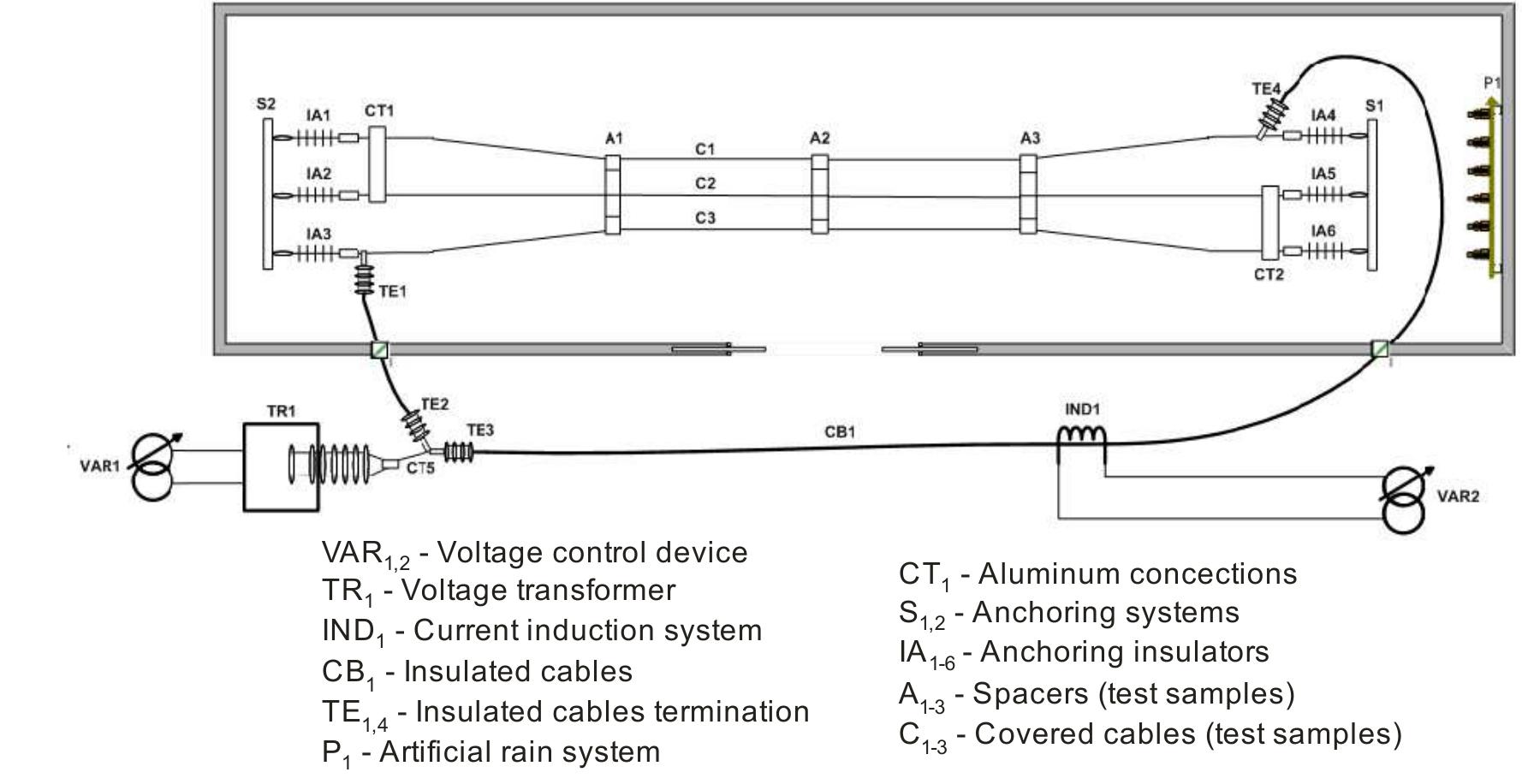
![Figure 3. Common damages developed along a covered conductor tested according to Brazilian recommendations [20].](https://figures.academia-assets.com/92297172/figure_003.jpg)
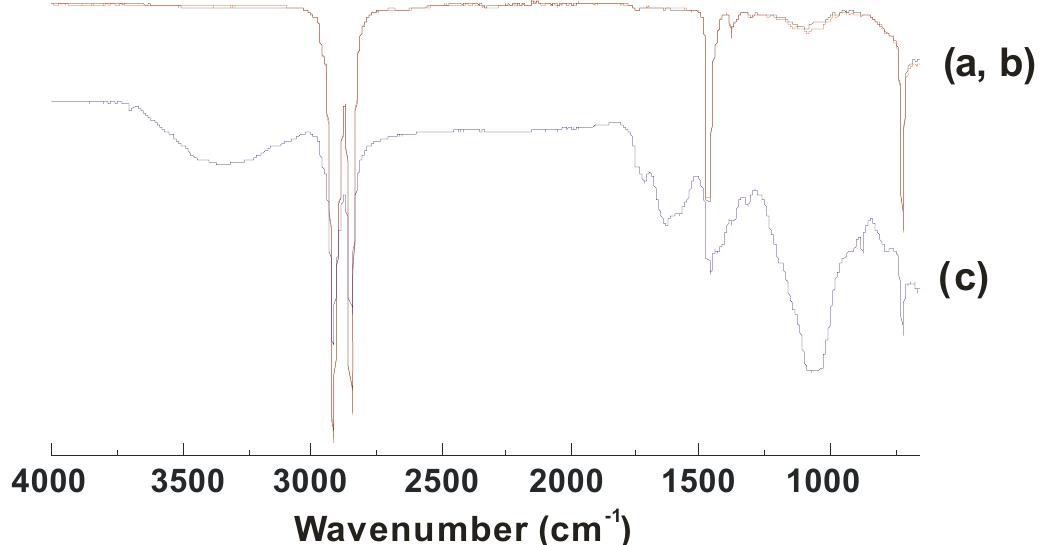
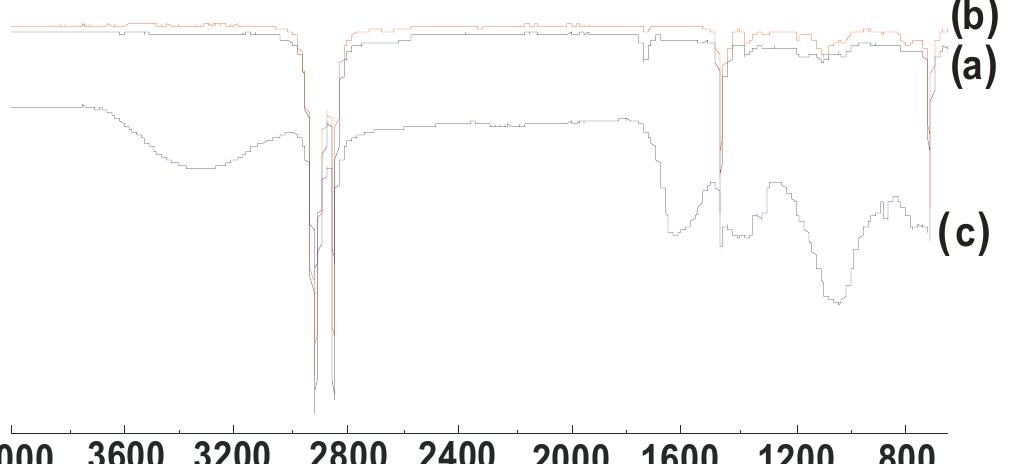


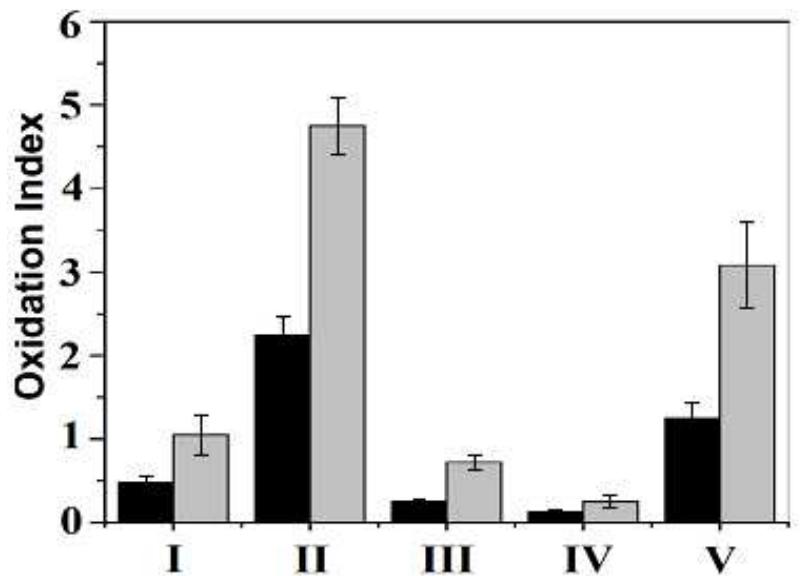




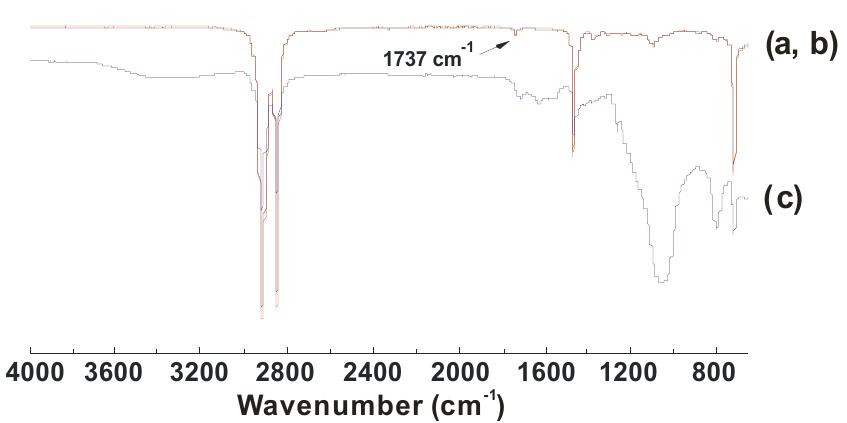
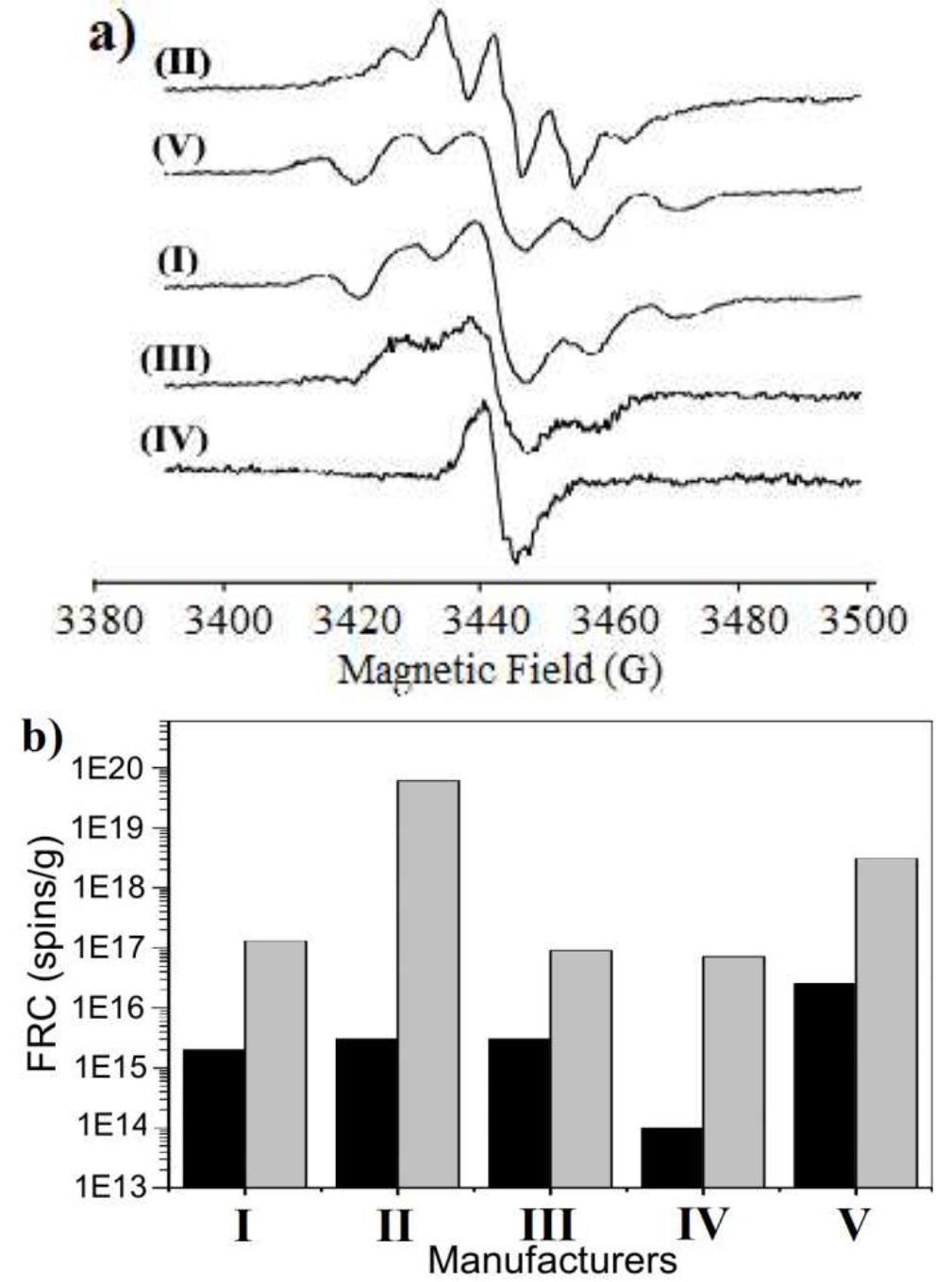

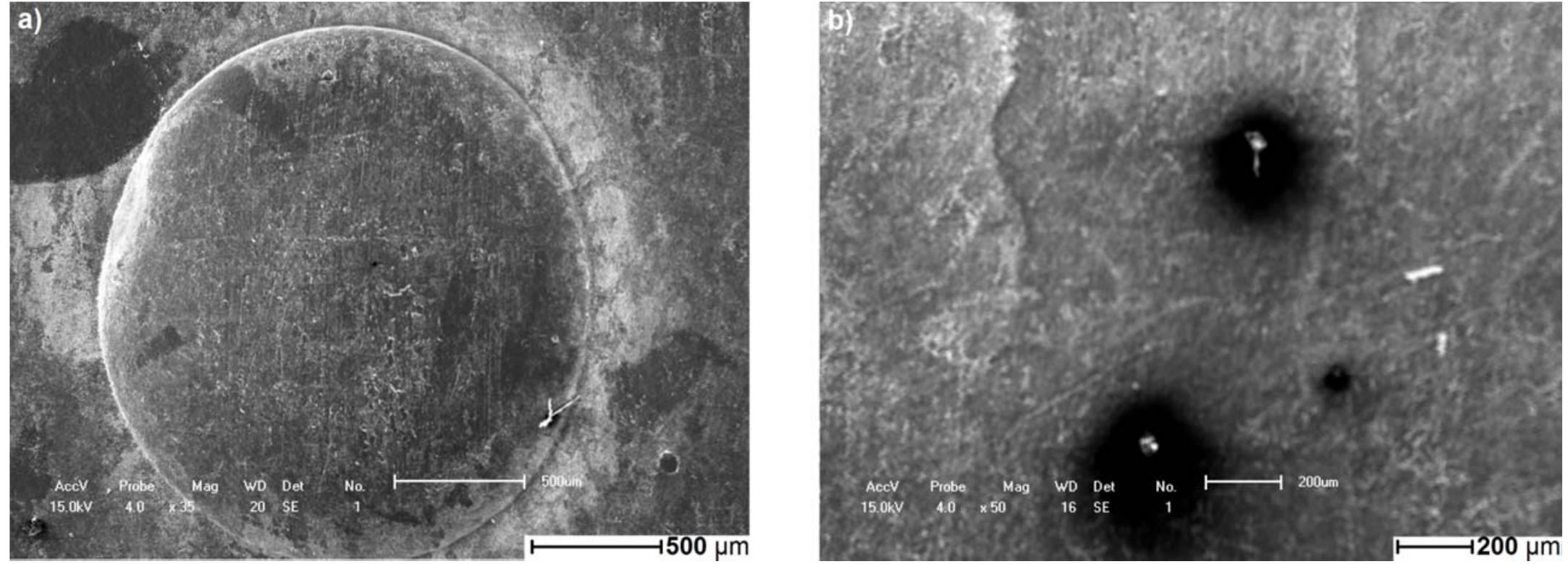
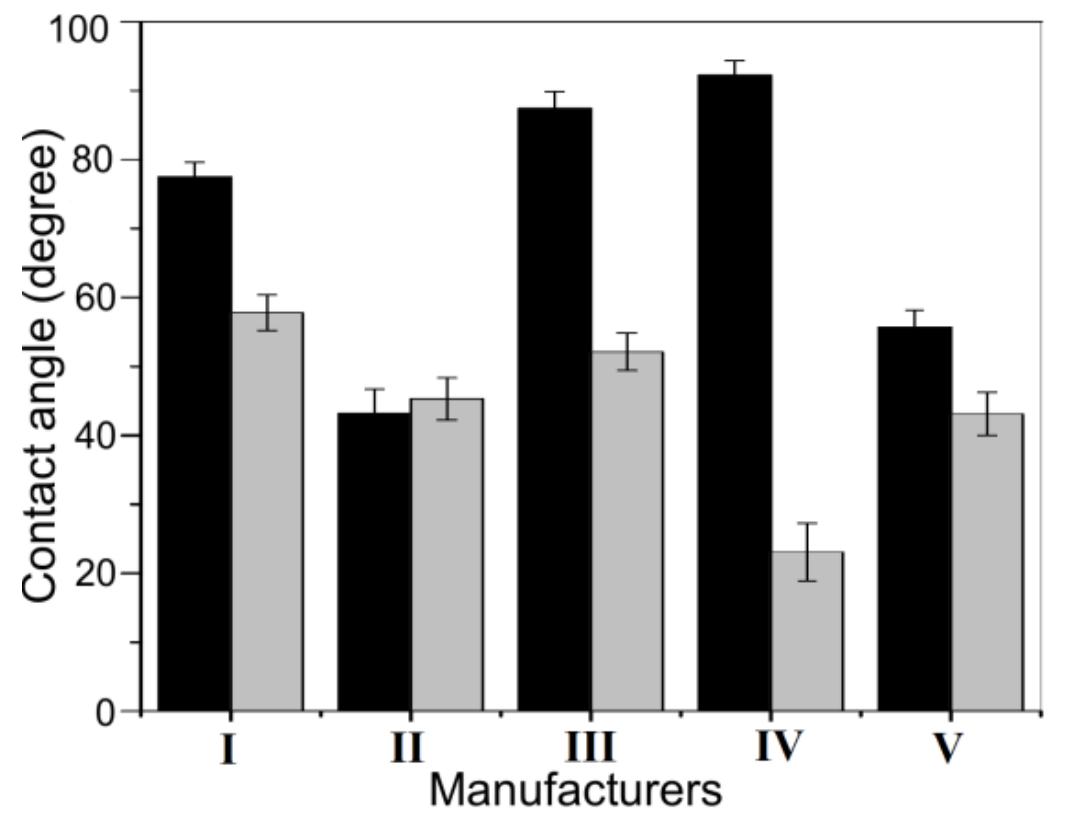

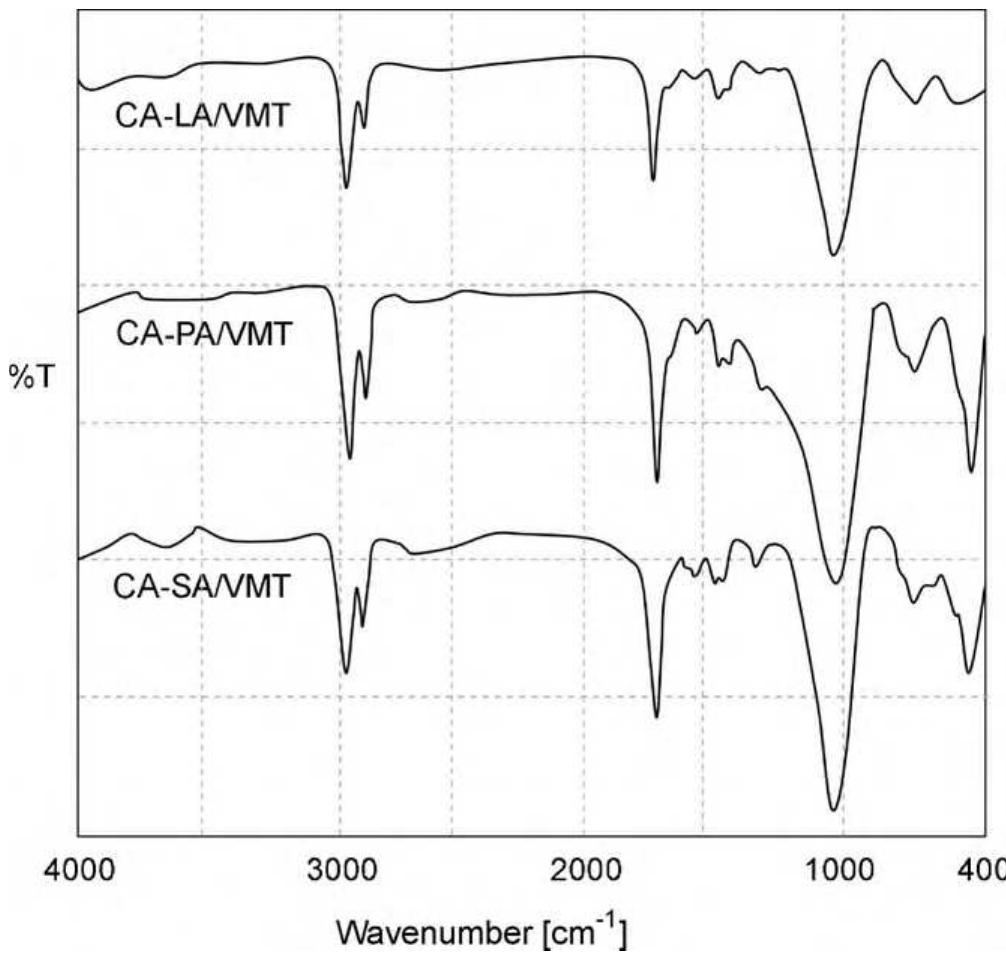

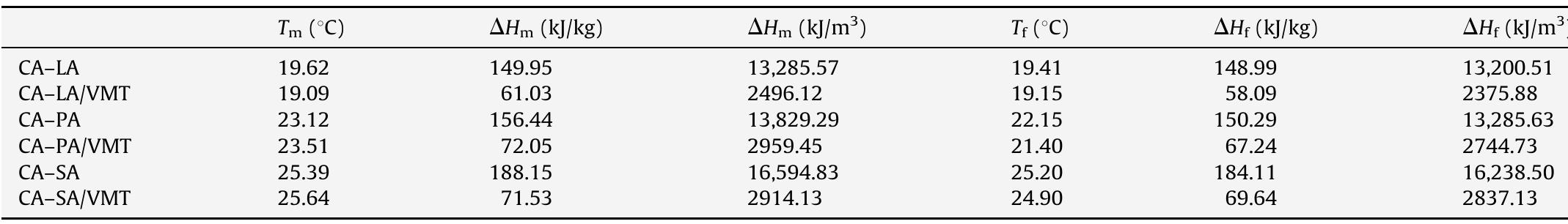
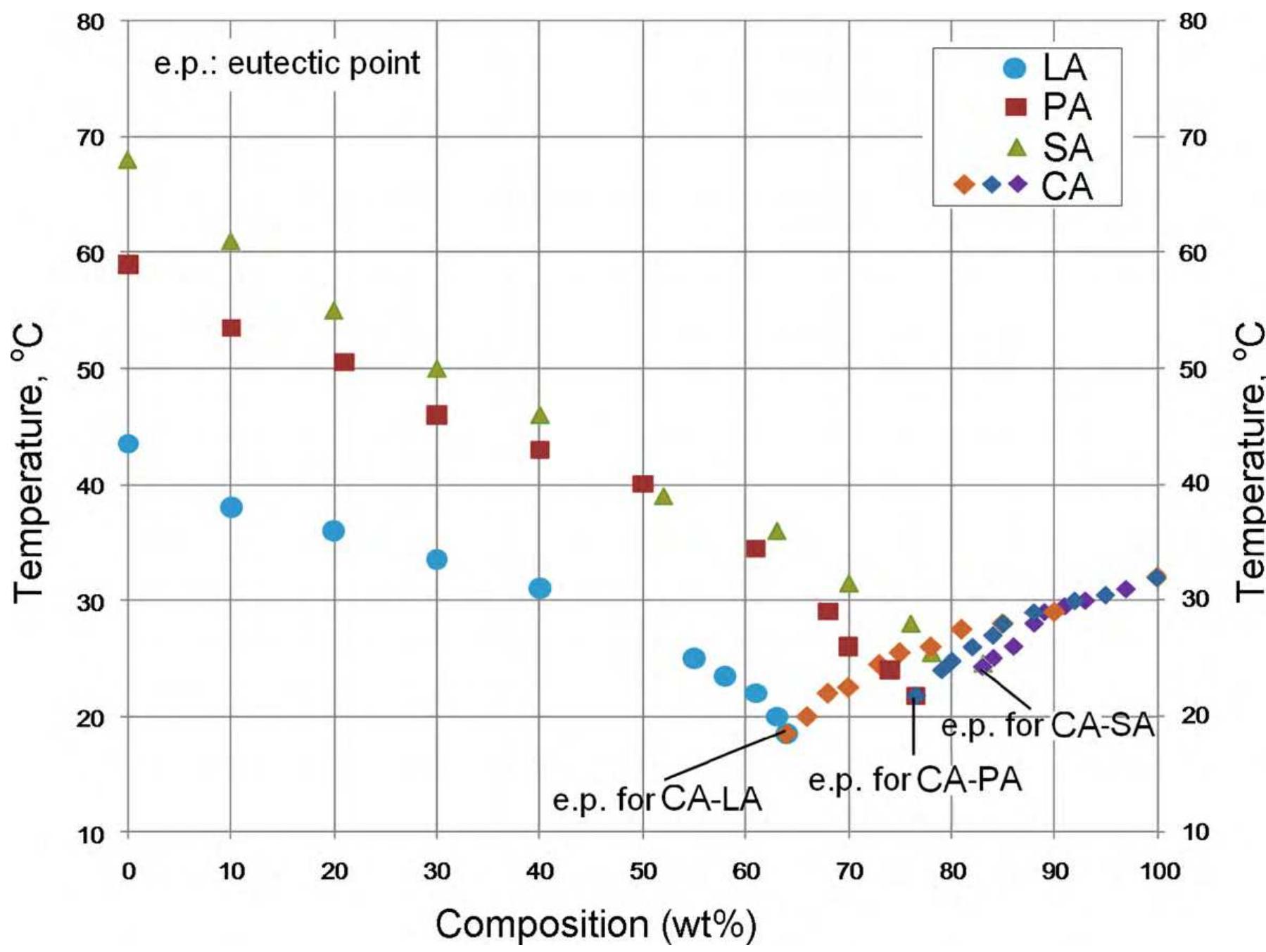
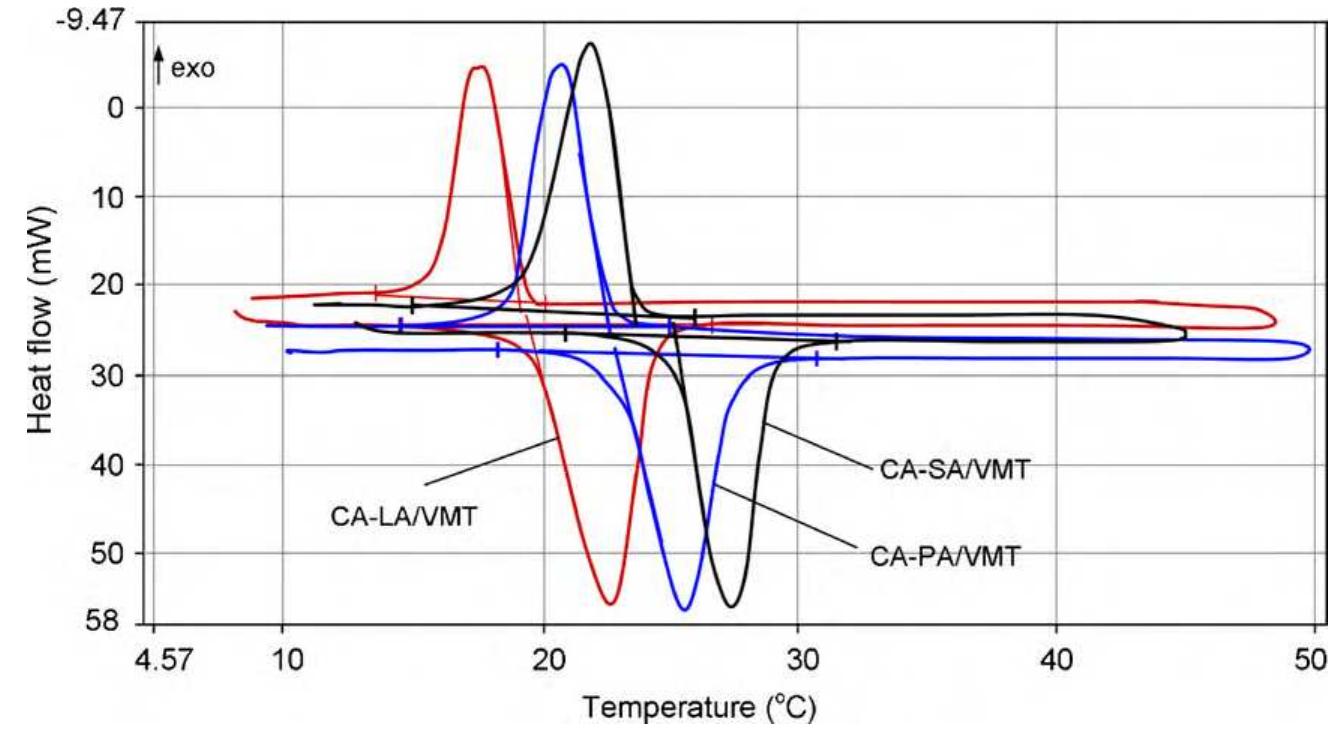
![Fig. 3. (a) FTIR spectrum of the form-stable composite PCMs and (b) DSC curves of the form-stable composite PCMs. temperatures of eutectic mixtures in the composites, the phase change temperatures of composites are very close to pure eutectic mixtures. The decrease in phase change temperatures is due to weak attractive interaction between fatty acid molecules and inner surface wall of the porous material. These results are in agreement with that reported by Radhakrishnan and Gubbins [26]; Radhak- rishnan et al. [27]; Zhang et al. [15]; Karaipekli and Sari [11]. In On the other hand, the latent heats of melting (AH,,) were found to be 61.03 kJ/kg, 72.05 kJ/kg and 71.53 kJ/kg for CA-LA/ VMT, CA-PA/VMT, and CA-SA/VMT, respectively as the latent](https://figures.academia-assets.com/49935873/figure_003.jpg)



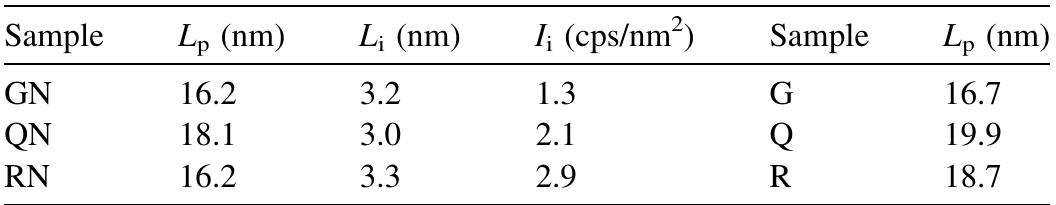


![The nitrogen content of cationized cotton fabric equal 0.0891%; [ammonium citrate], 6%; [SHP], 6%; AC/SHP molar ratio, 1; wet pick-up, 100%; drying, 100°C/3 min; curin 180°C/90s. Control sample is the cationized sample which have nitrogen content of 0.0891% then crosslinked with 6% ammonium citrate. Effect of concentration of SME on the performance properties of treated cotton fabrics. Table 2](https://figures.academia-assets.com/50160225/table_001.jpg)


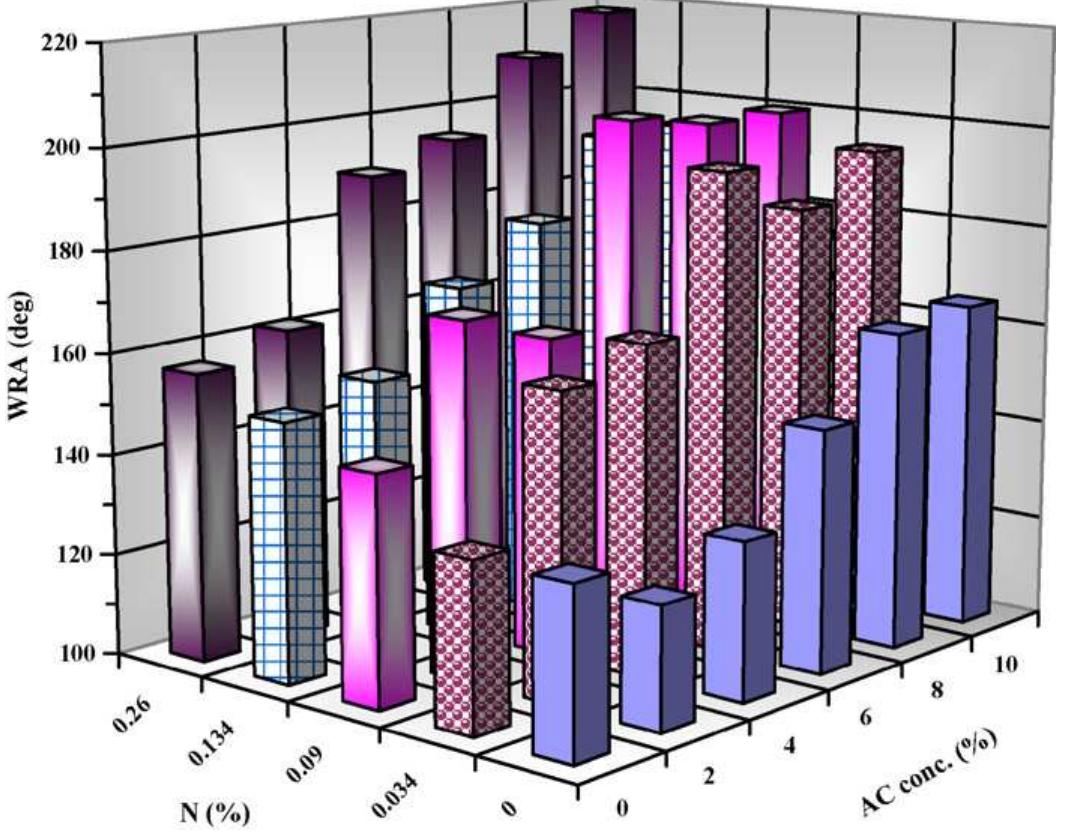

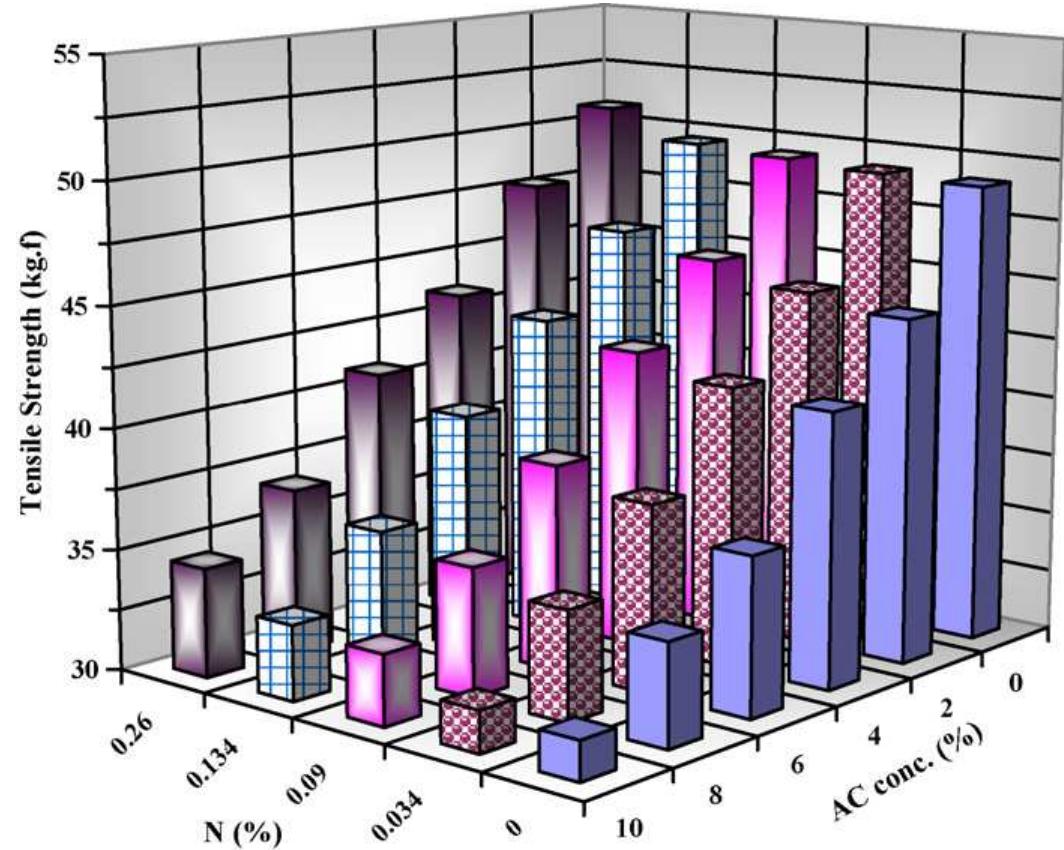

![‘he nitrogen content of cationized cotton fabric equal 0.0891%; [ammonium citrate], 6%; [SHP], 6%; AC/SHP molar ratio, 1; wet pick-up, 100%; drying, 100°C/3 min; curing 80°C/90s. Comparative study on the effect of ammonium citrate vis a vis citric acid. Table 1](https://figures.academia-assets.com/50160225/table_002.jpg)