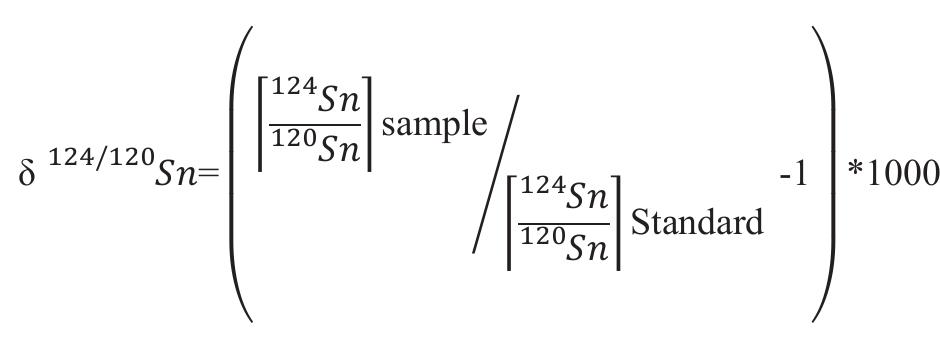Key research themes
1. How can mass spectrometric and nuclear reaction techniques enhance precision in stable isotope analysis of trace elements and isotopes?
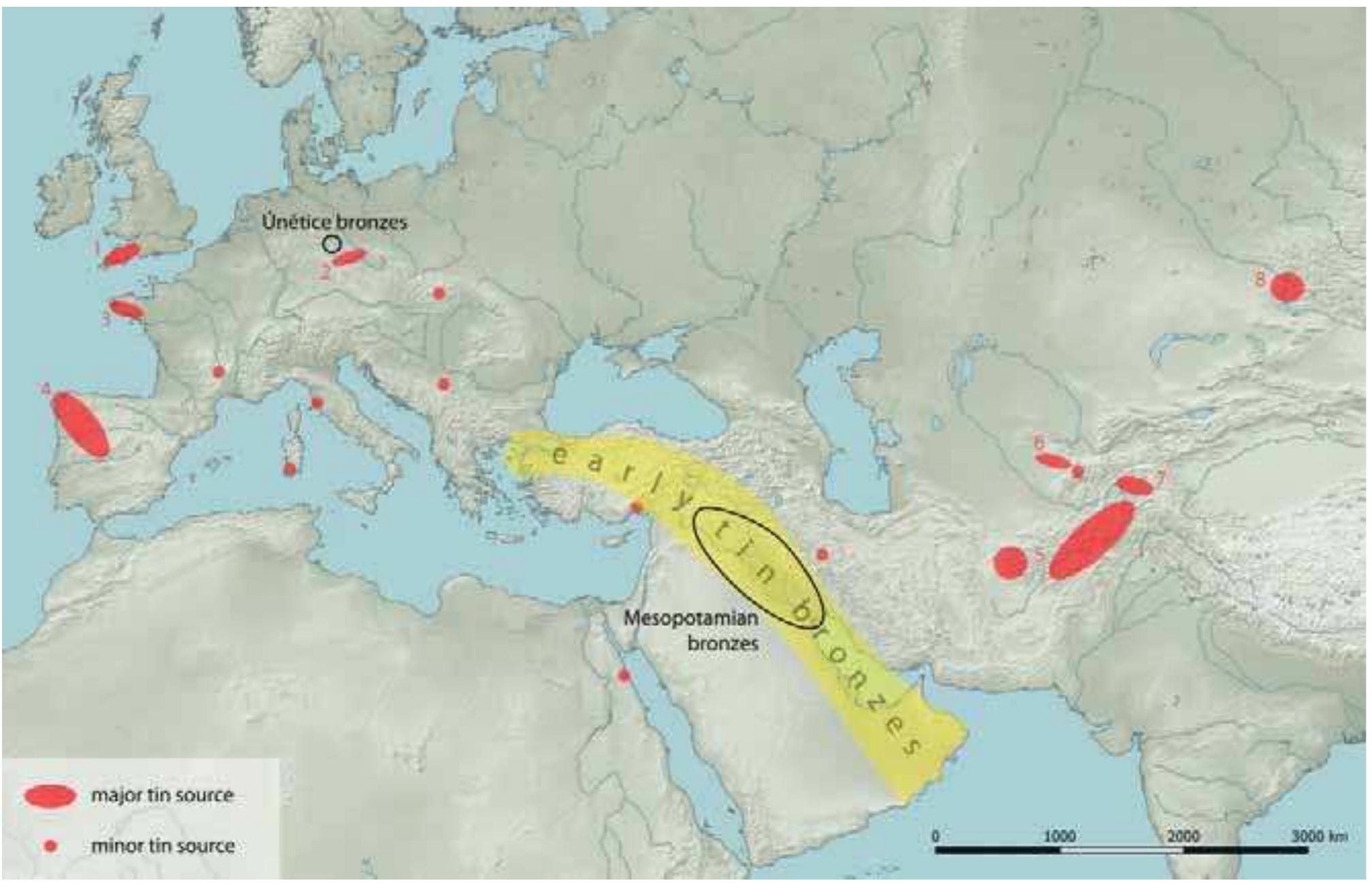
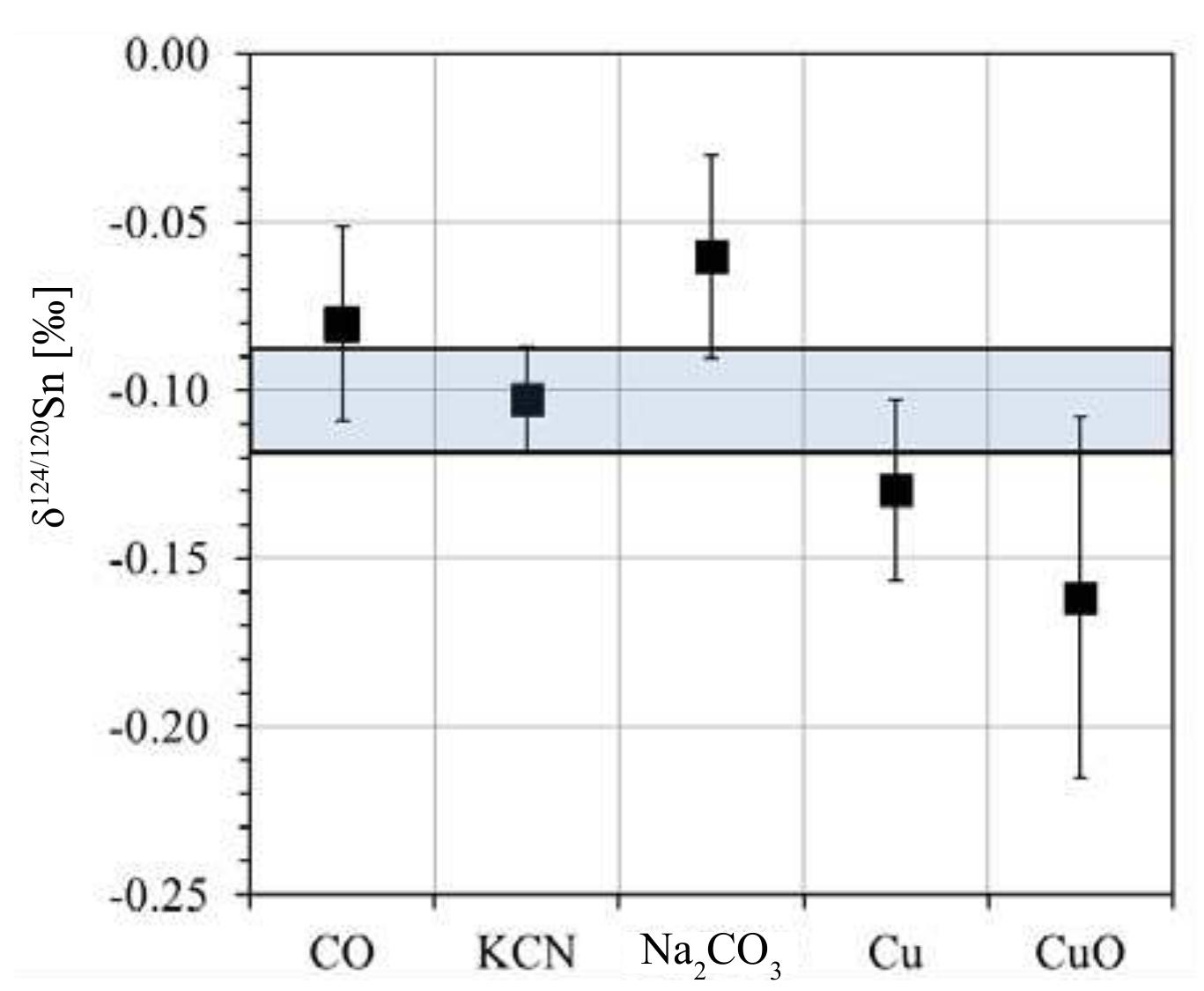
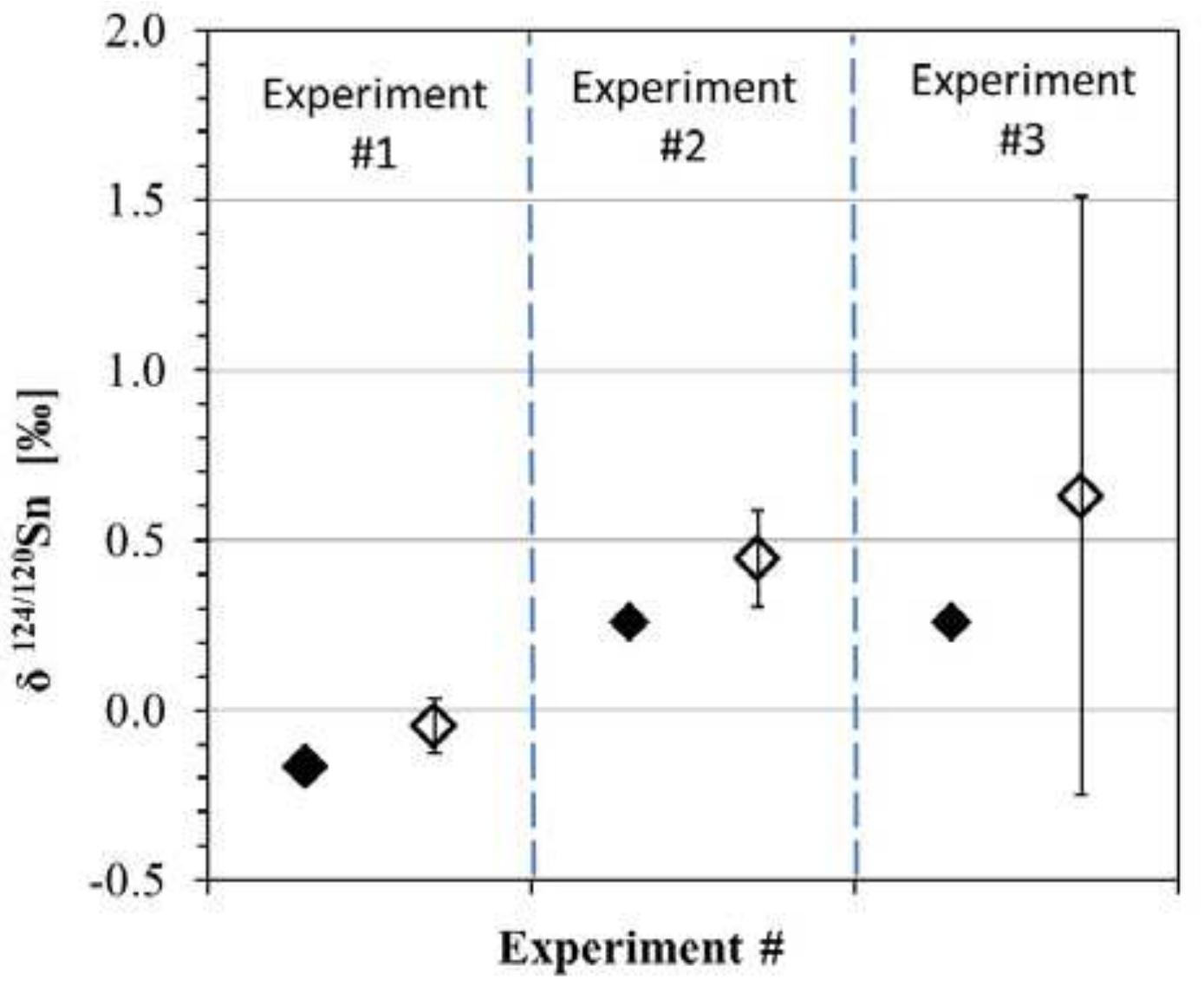
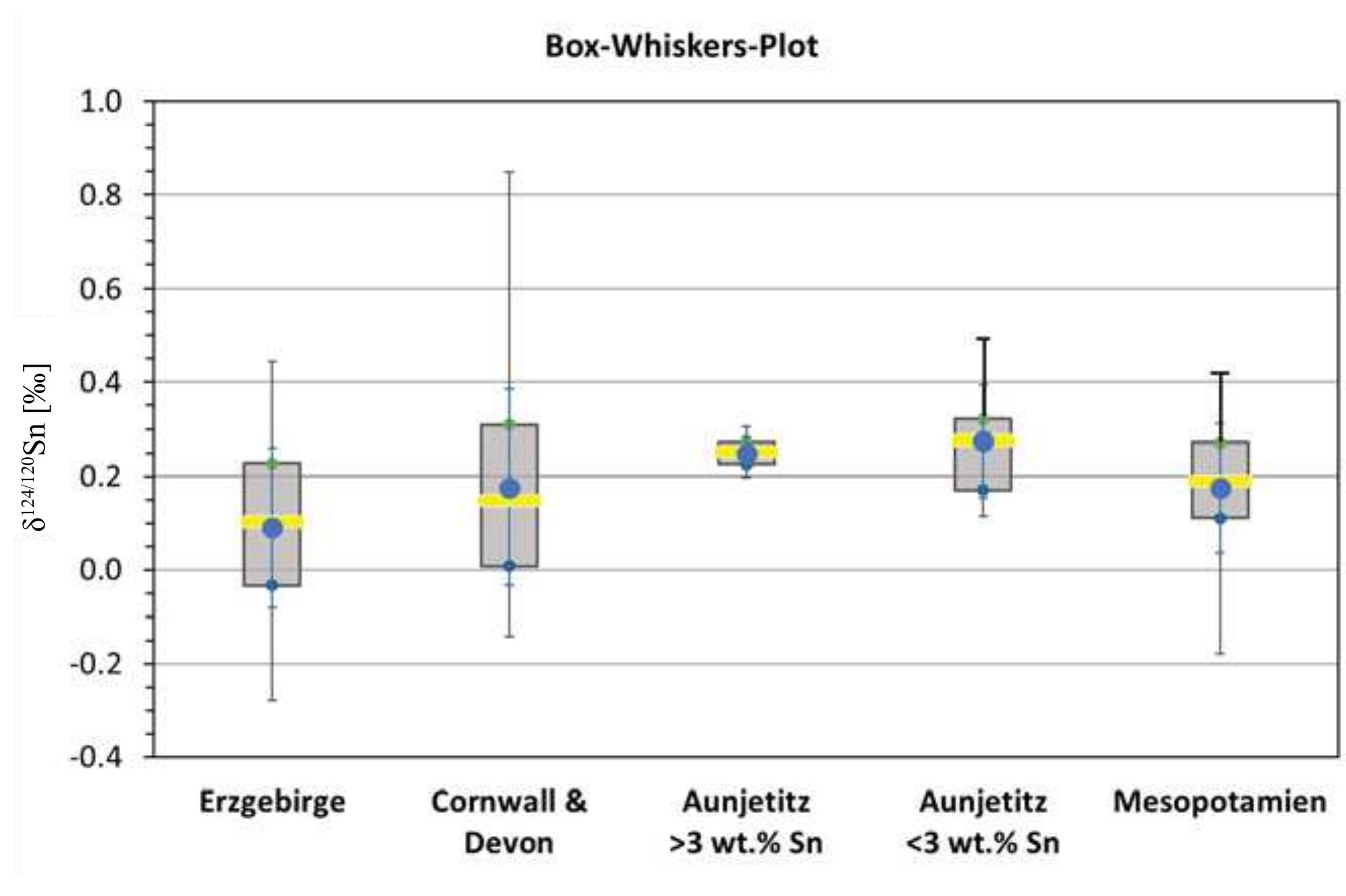
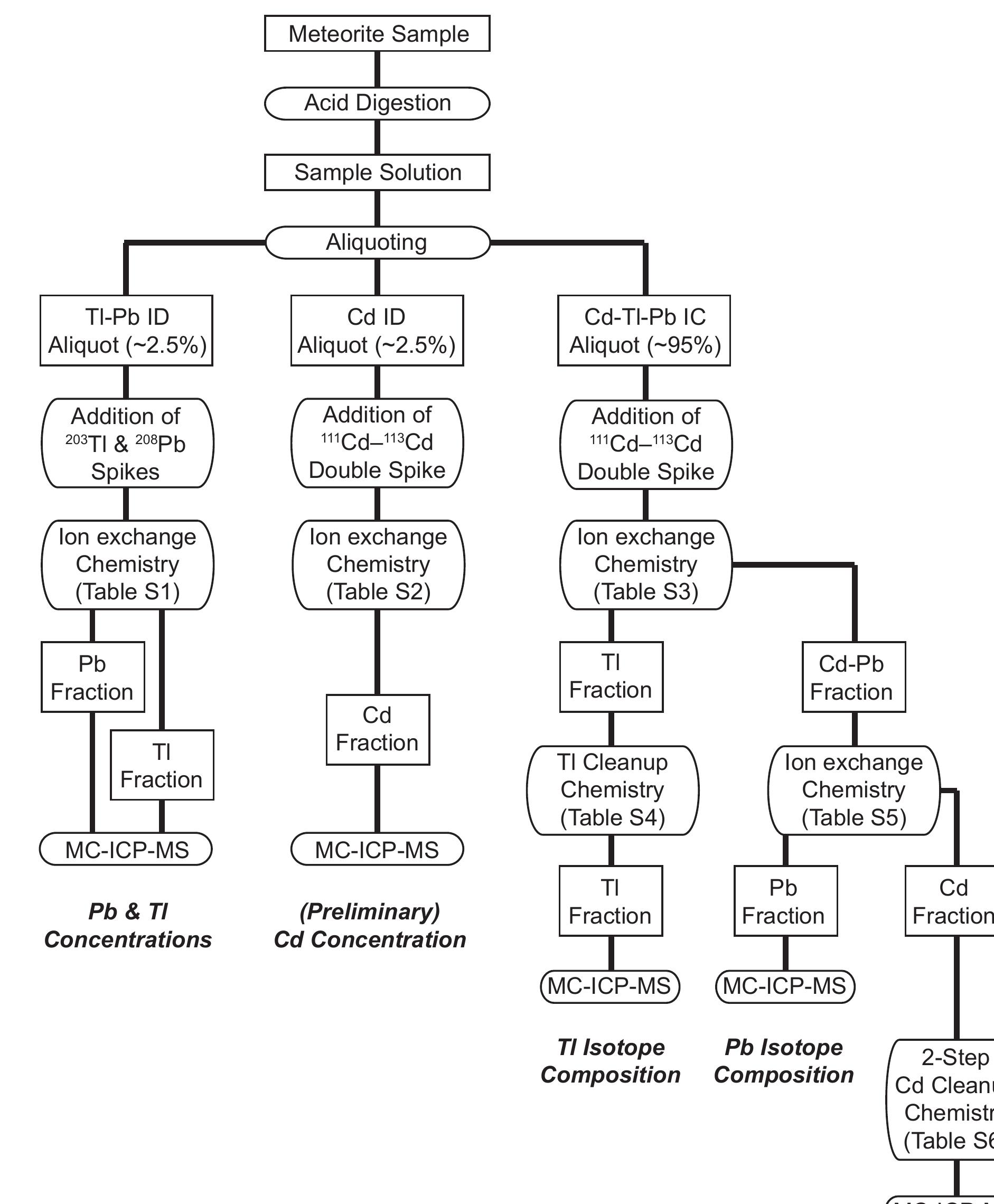
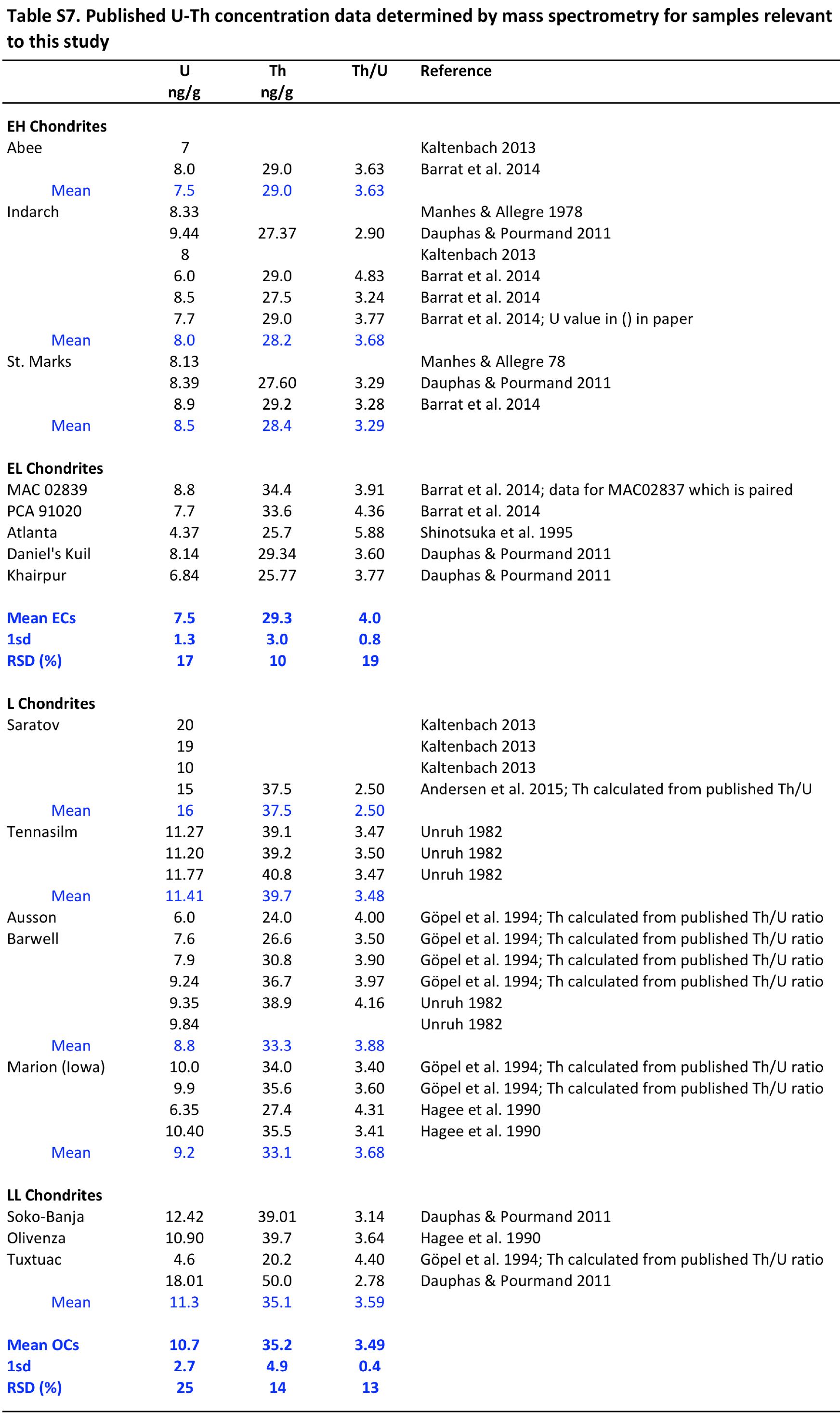
Precise and accurate measurement of stable isotopes at micro to ultra-trace levels is critical for advancing fields ranging from geochemistry to nuclear physics and environmental sciences. This research theme focuses on development, optimization, and validation of analytical methodologies—primarily nuclear reaction-based activation analysis and various mass spectrometry techniques including MC-ICP-MS and laser-ablation coupled mass spectrometry—to improve sensitivity, specificity, and reproducibility in stable isotope determinations, especially when sample quantities are limited or matrix complexity is high. Enhancing methodological rigor allows isotope ratios to be used more effectively as tracers in complex systems and expands the scope of isotope applications.
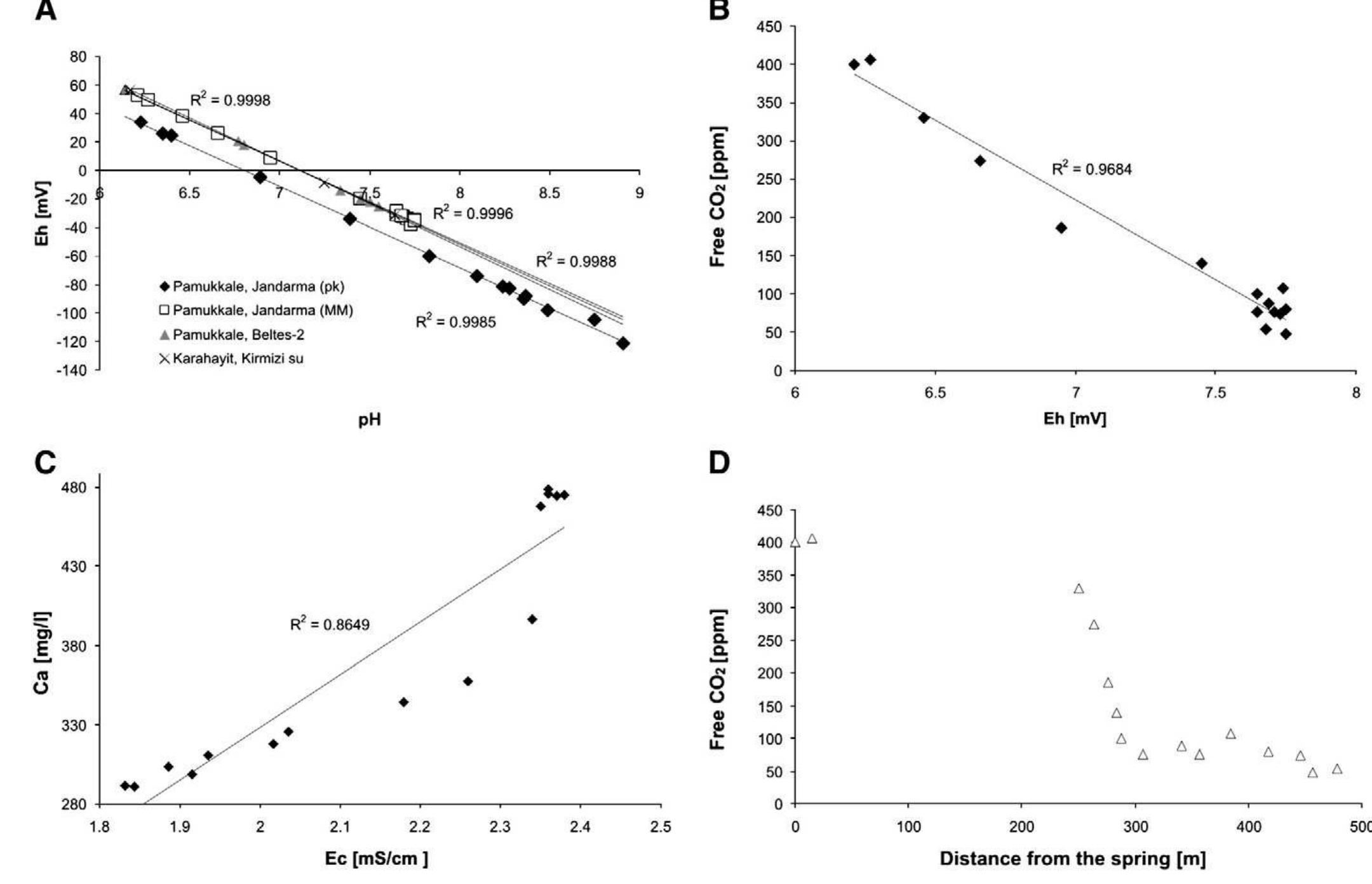
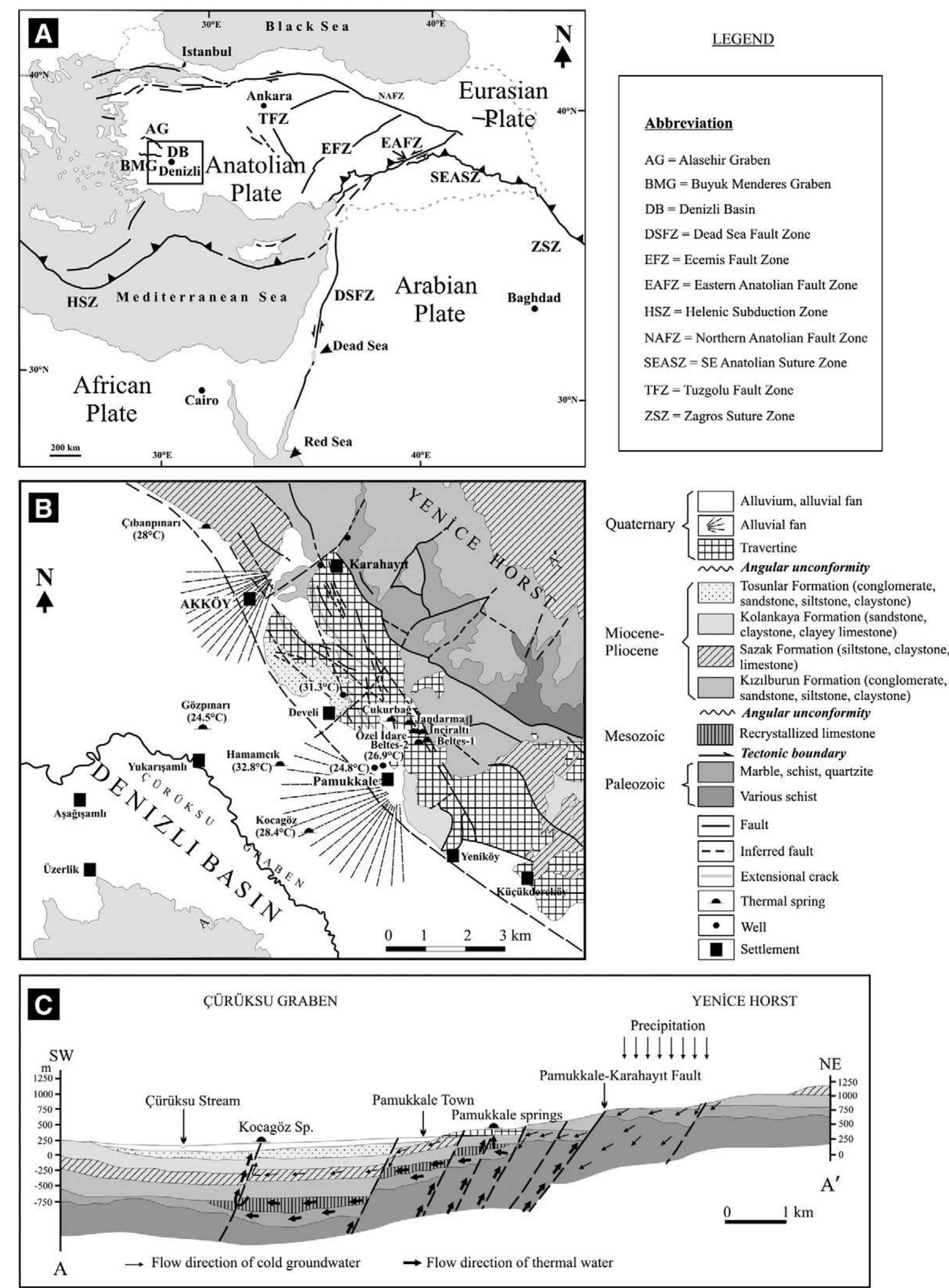
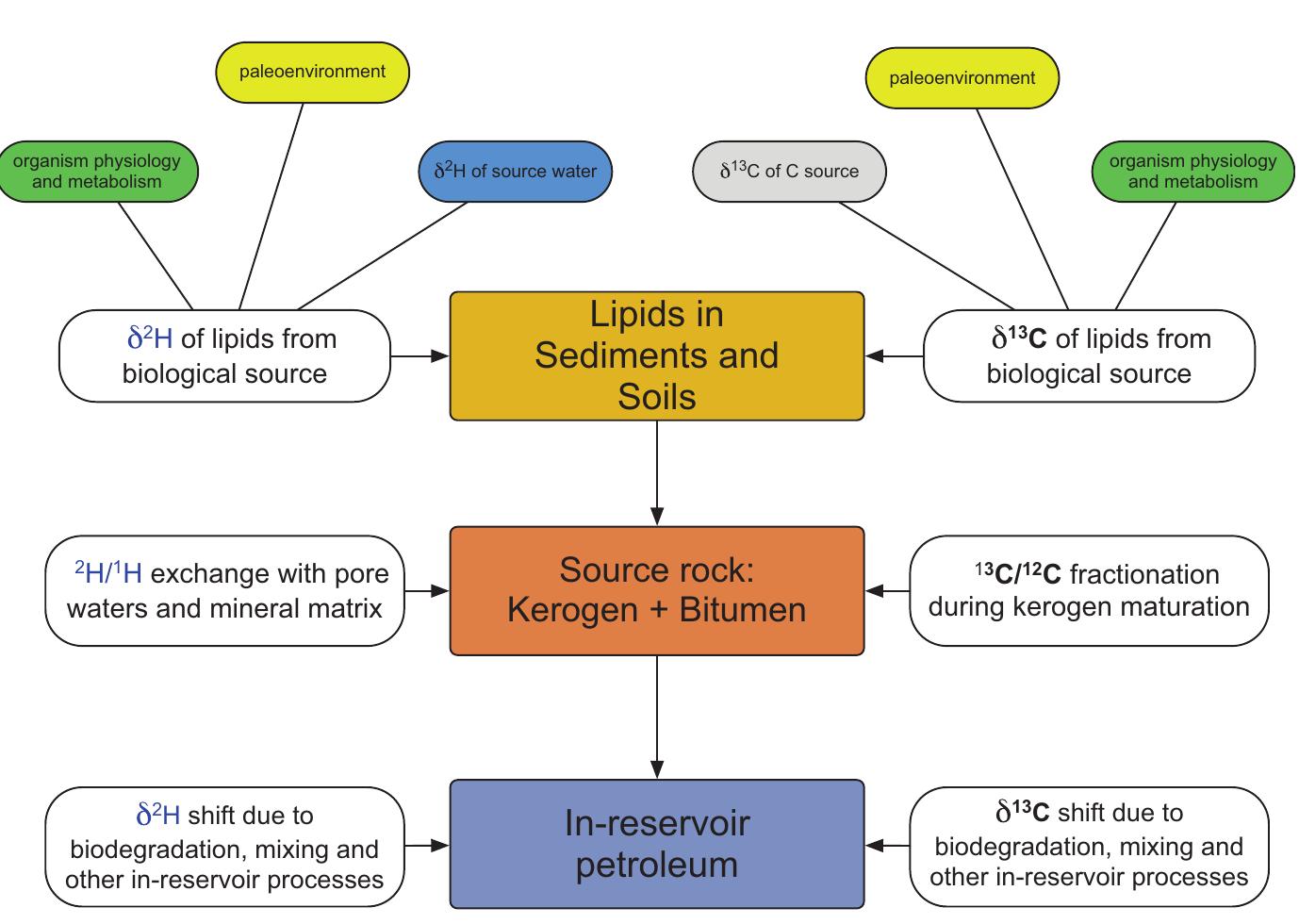
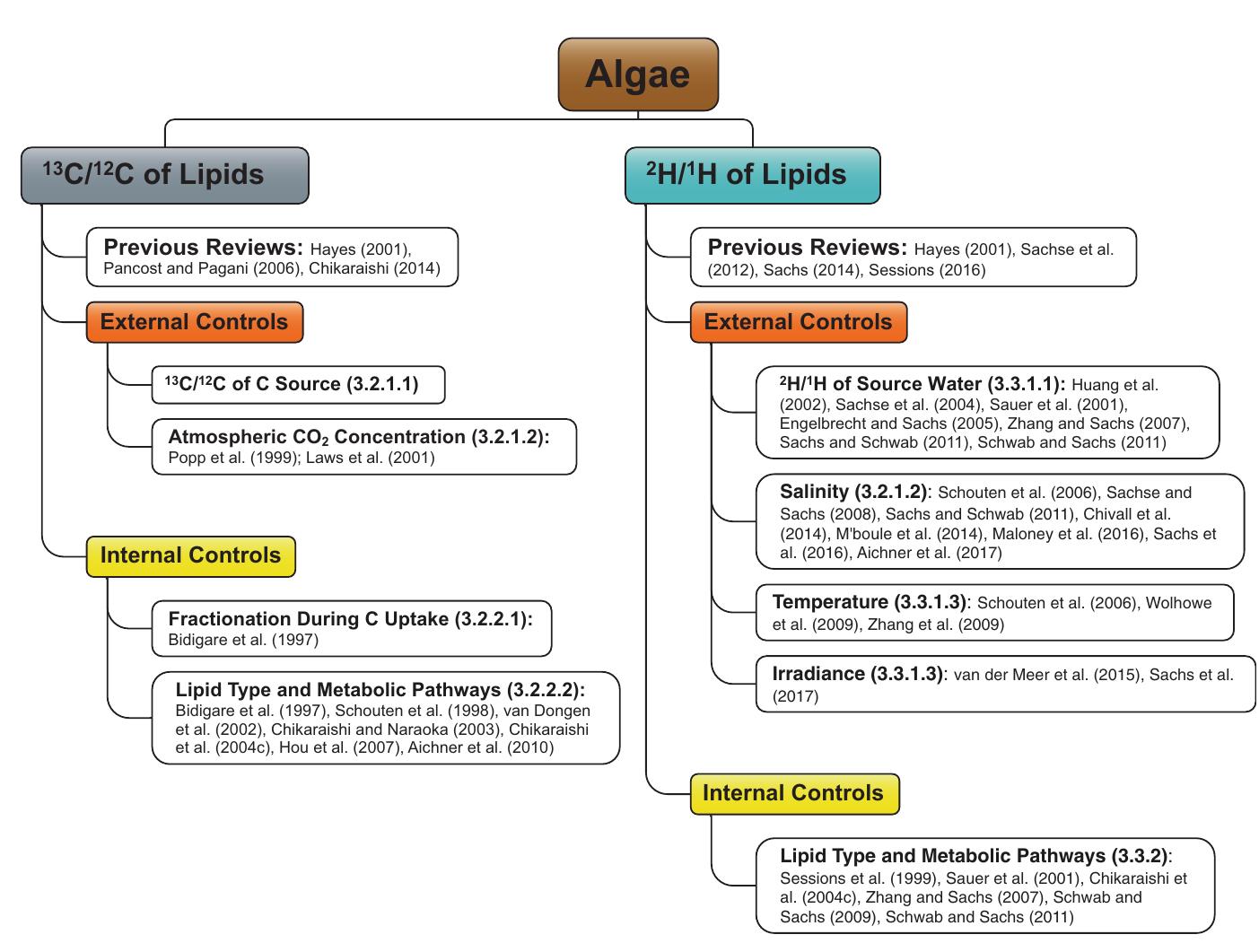
2. What are the causes and implications of kinetic and equilibrium isotope fractionation effects in natural and anthropogenic carbonate systems and biological contexts?
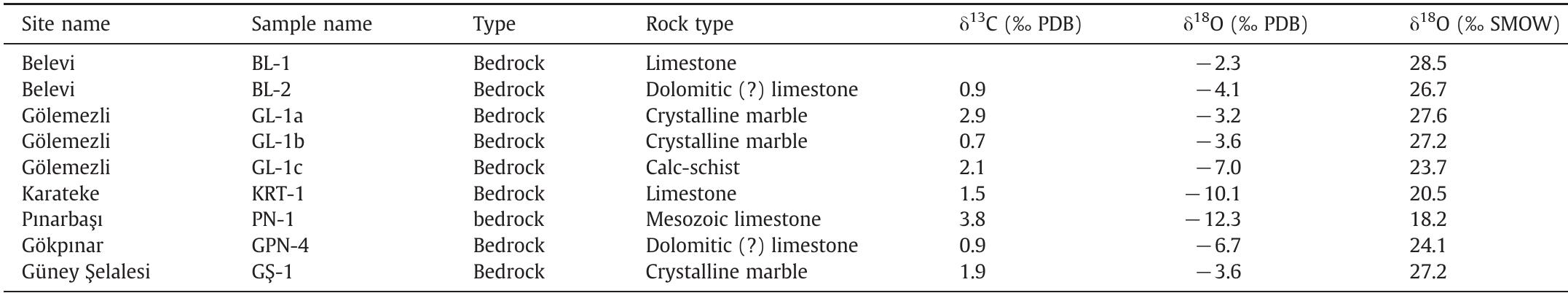
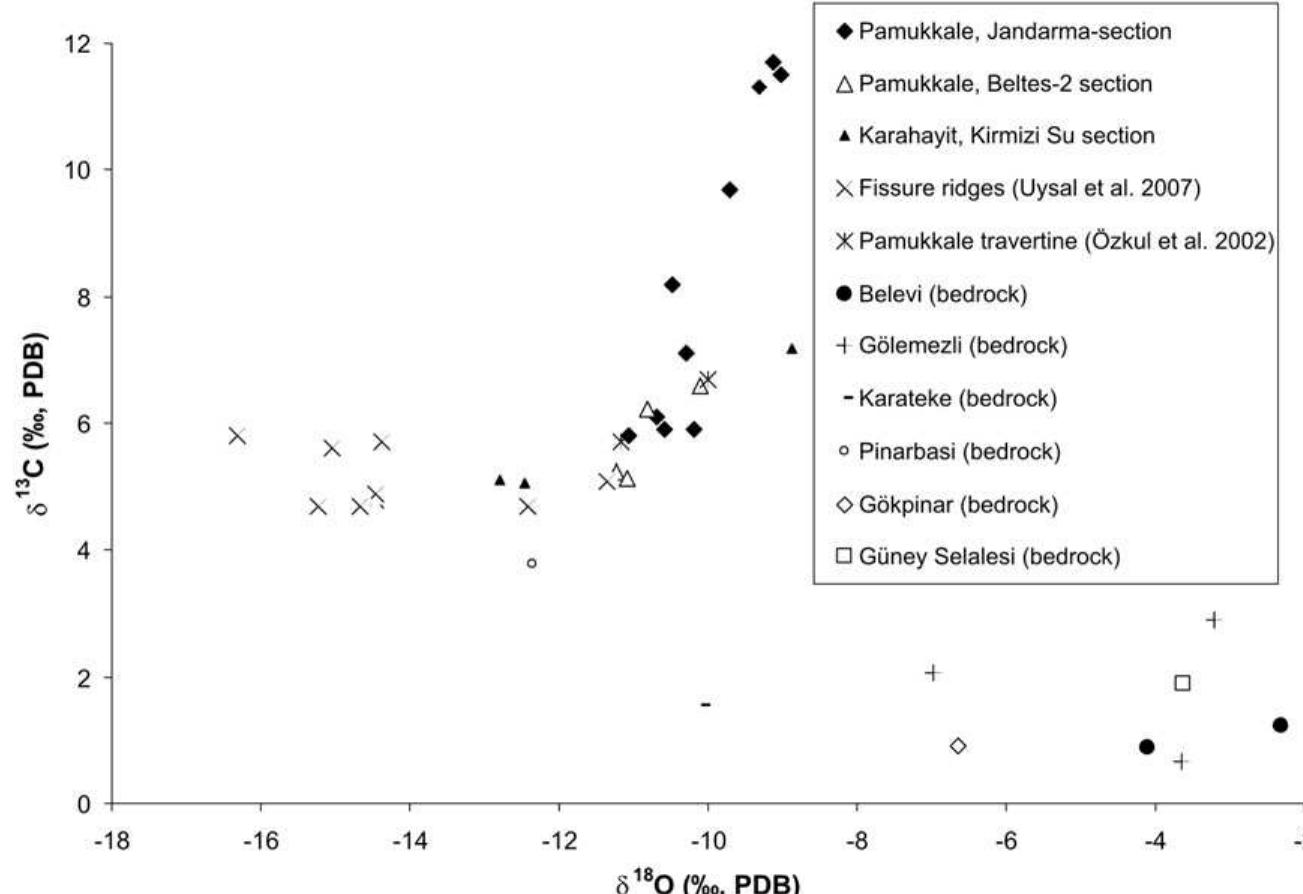
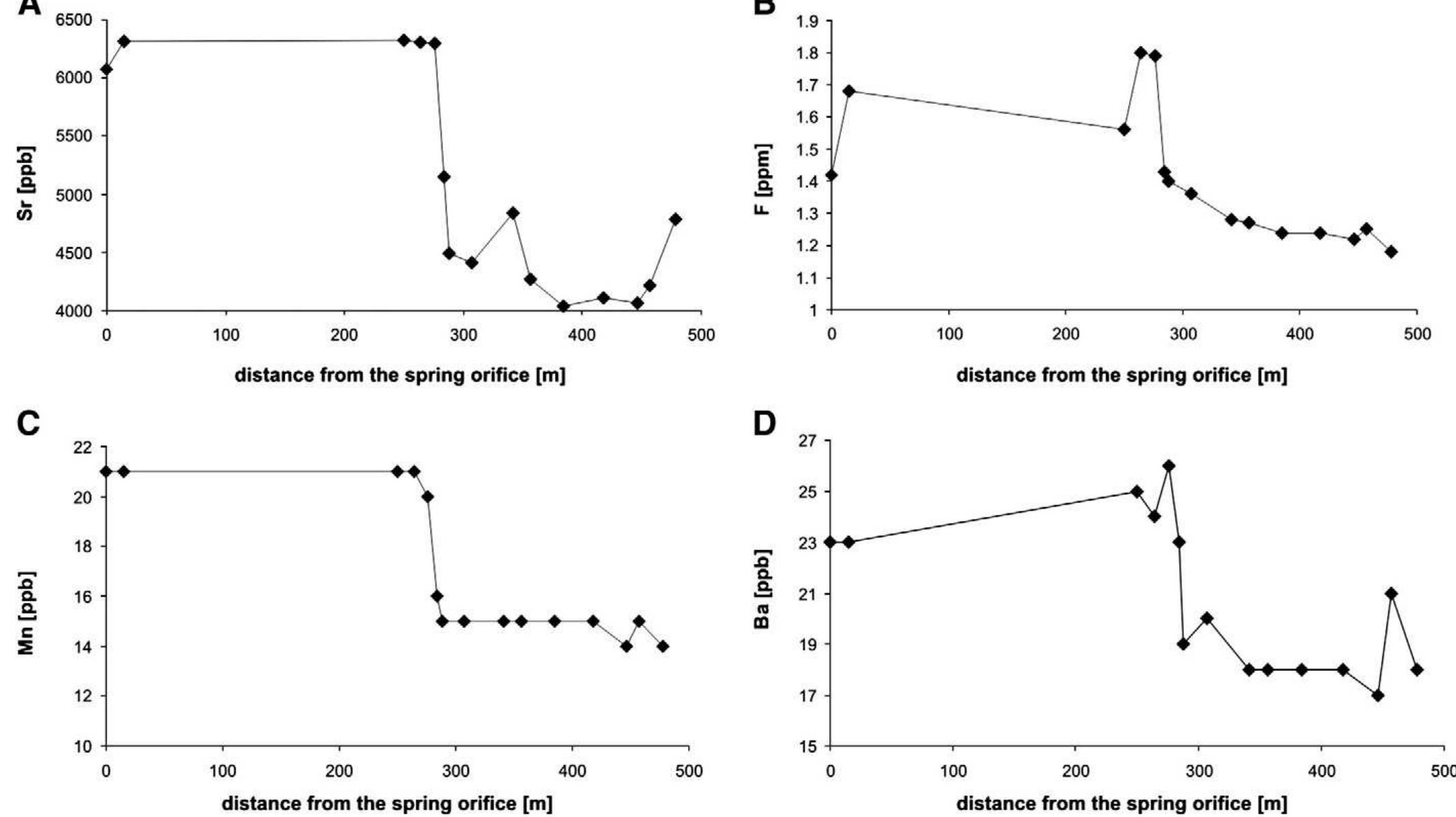
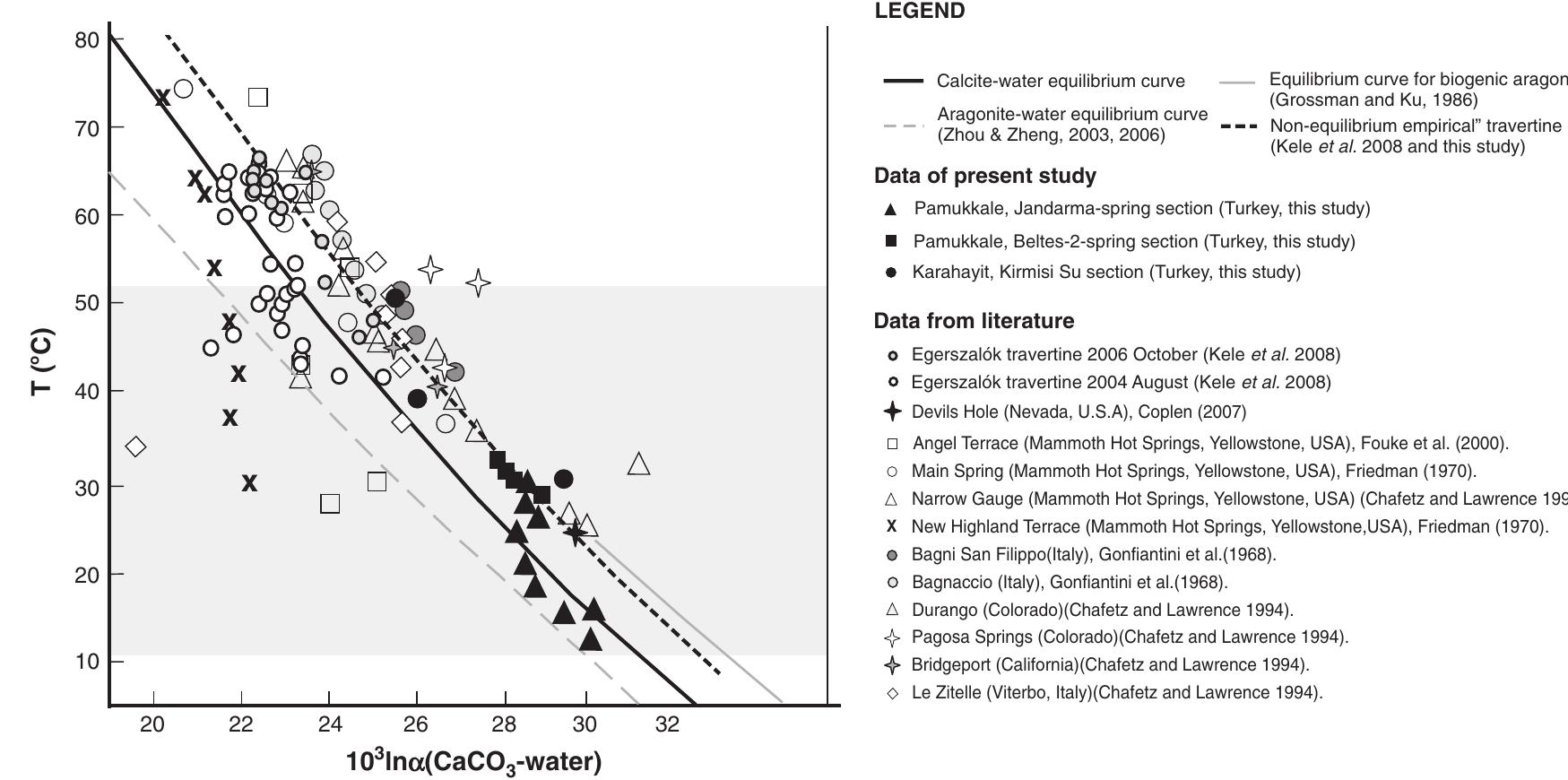
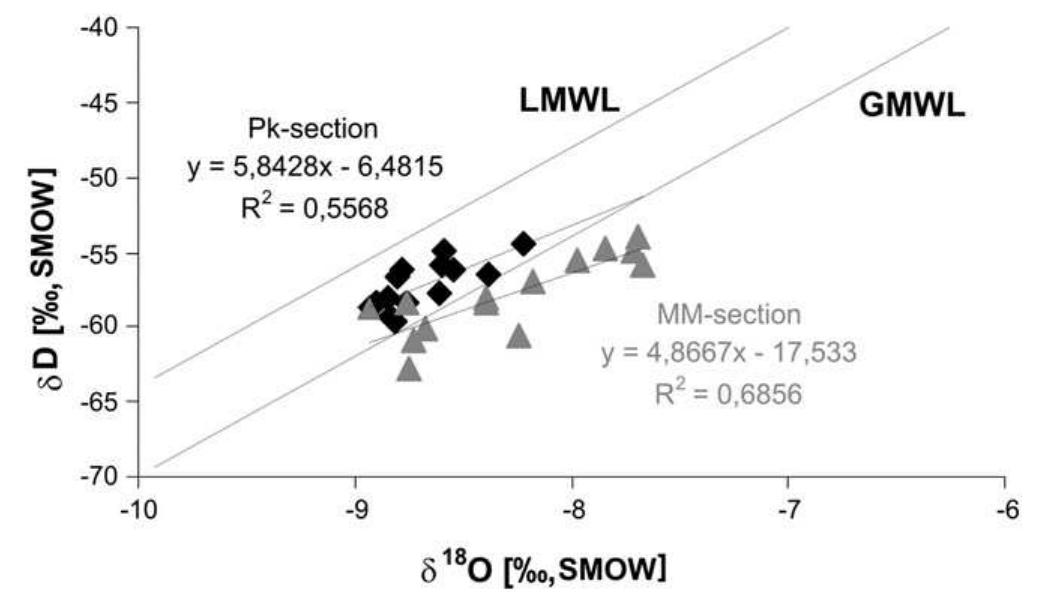
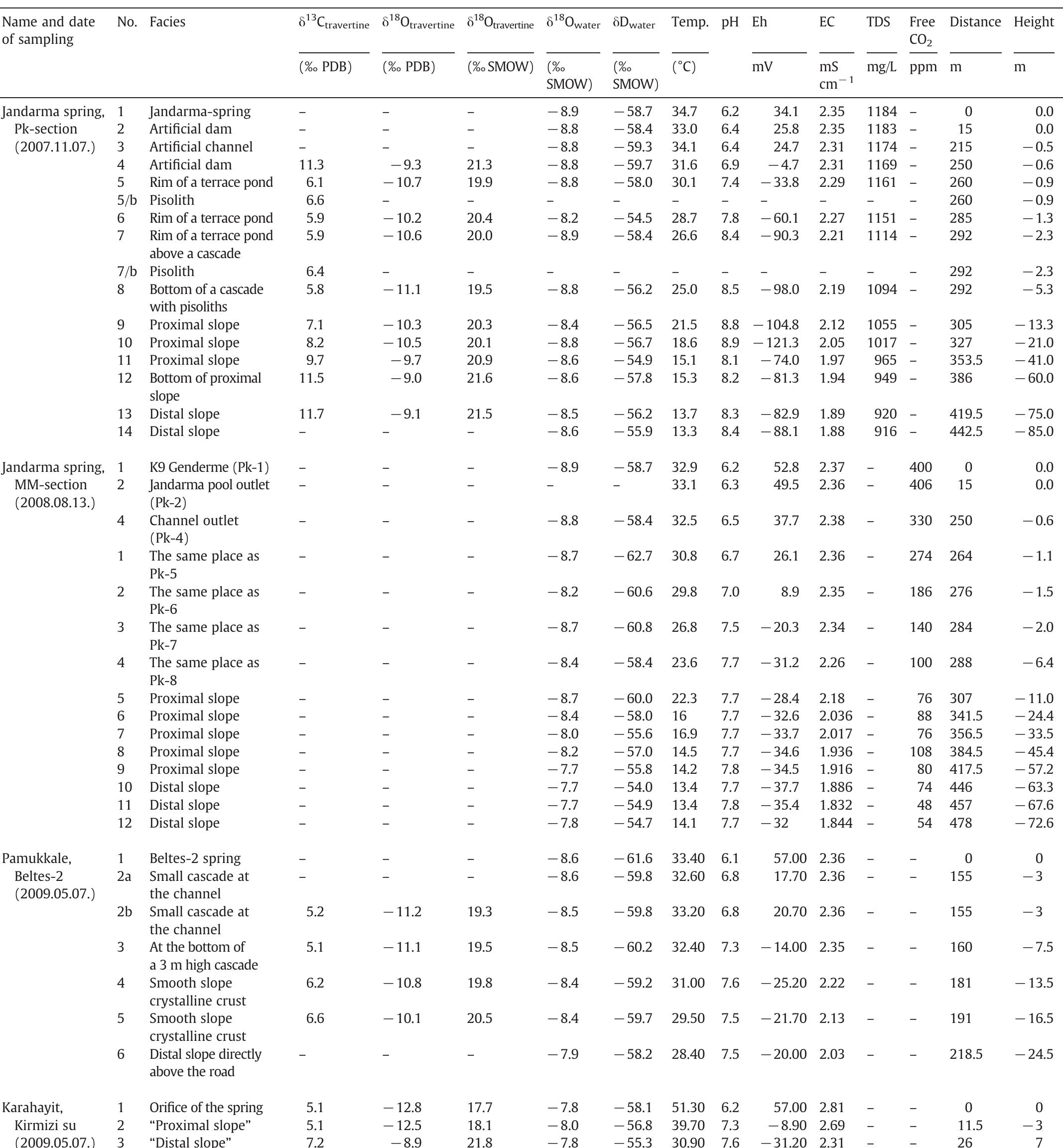
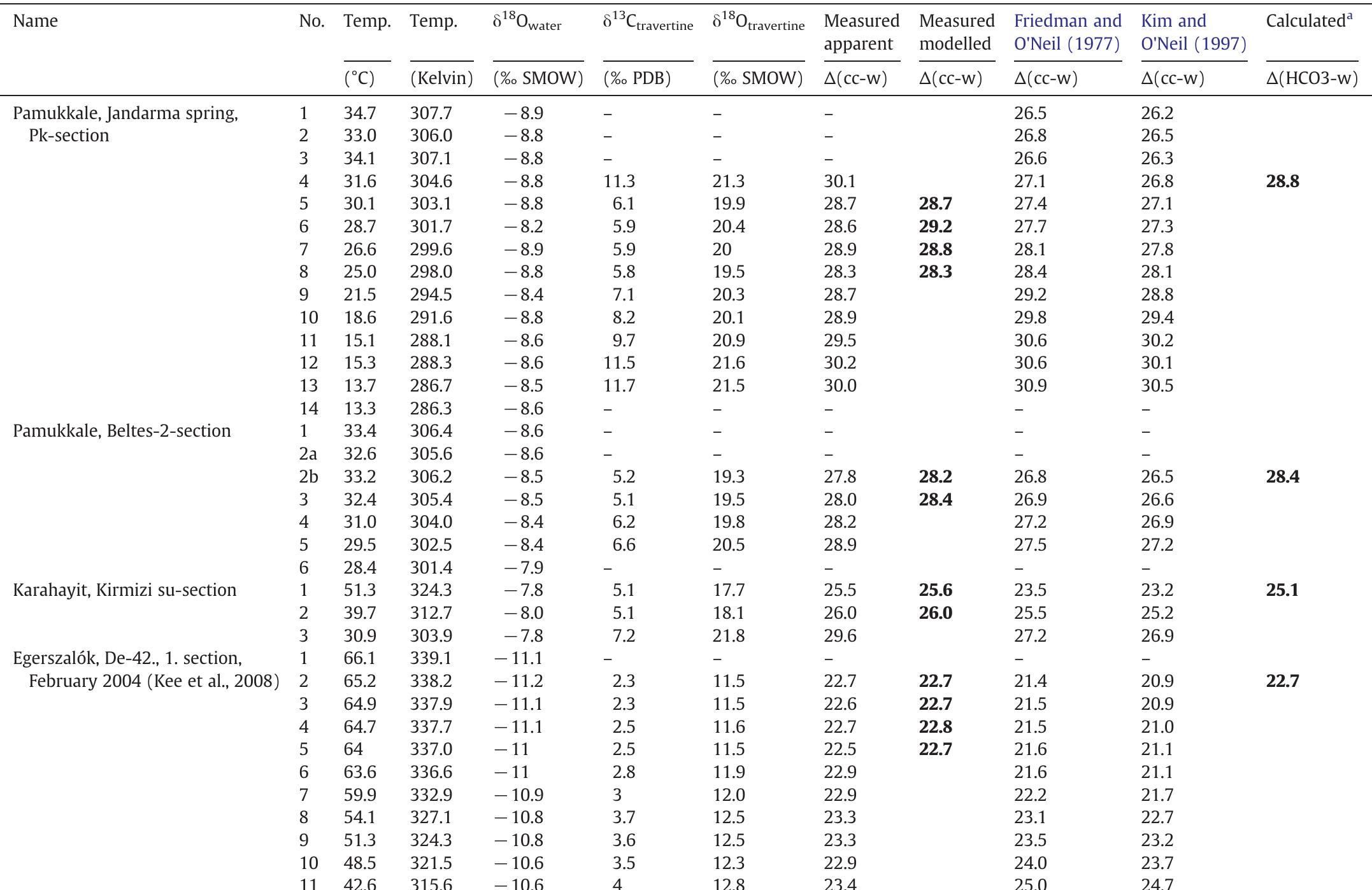
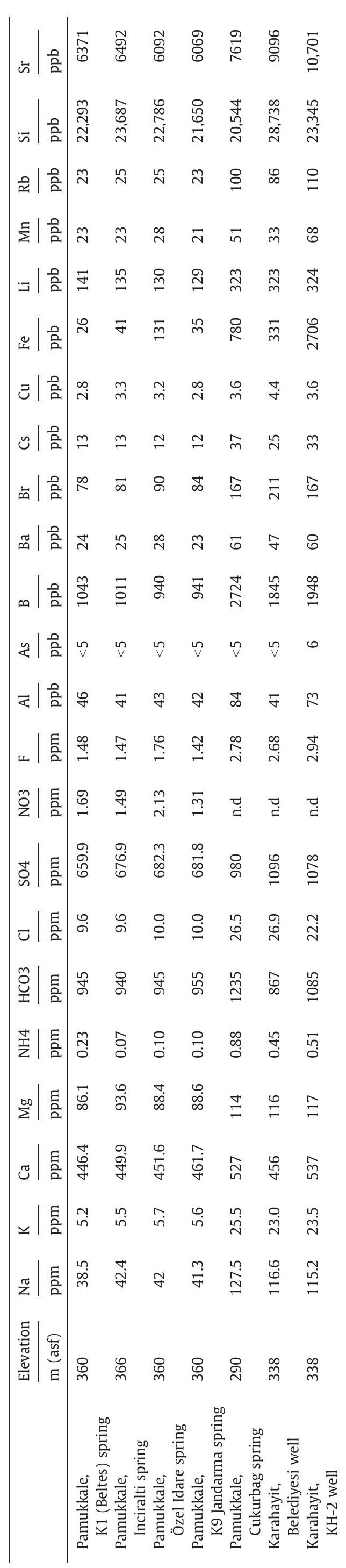
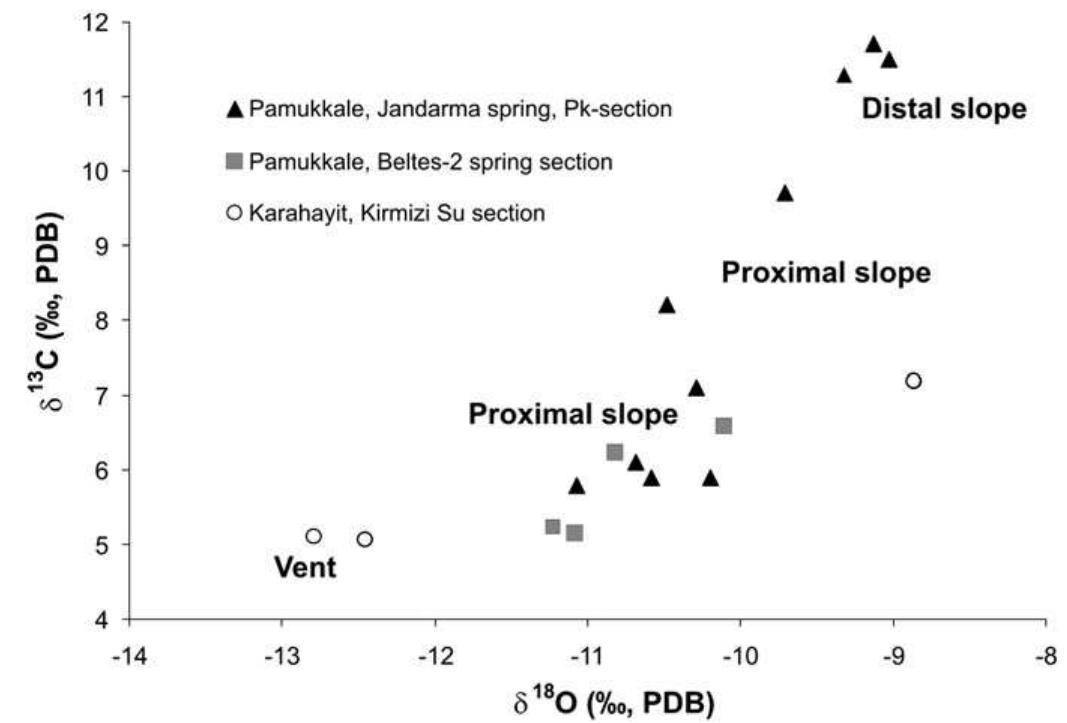
Stable isotope fractionation in carbonates and biological materials is influenced by a complex interplay of kinetic, equilibrium, and environmental factors. This theme encompasses investigations into the mechanistic origins, environmental dependencies, and applied implications of isotope fractionations—especially for oxygen, carbon, nitrogen, and sulfur isotopes—in contexts ranging from carbonate precipitation under high-pH anthropogenic conditions to isotope signals reflecting human diseases. Understanding these processes is critical for reconstructing paleoenvironments, diagnosing pathologies, and interpreting isotopic records influenced by both enzymatic and abiotic reactions.
3. How can stable isotope applications be optimized through software standardization and uncertainty management in the measurement and calibration of isotope ratios?
Achieving reproducible, accurate stable isotope data requires standardized computational frameworks for data reduction, analysis, and uncertainty evaluation, particularly in complex or time-resolved isotopic measurements. Additionally, precise uncertainty propagation and adherence to international normalization standards underpin reliable calibration of measurements against reference materials. This theme focuses on software solutions for harmonizing isotope data workflows, best practices in reference material use, calibration curve construction, and uncertainty quantification to ensure data comparability across laboratories and improve confidence in isotopic interpretations across scientific fields.