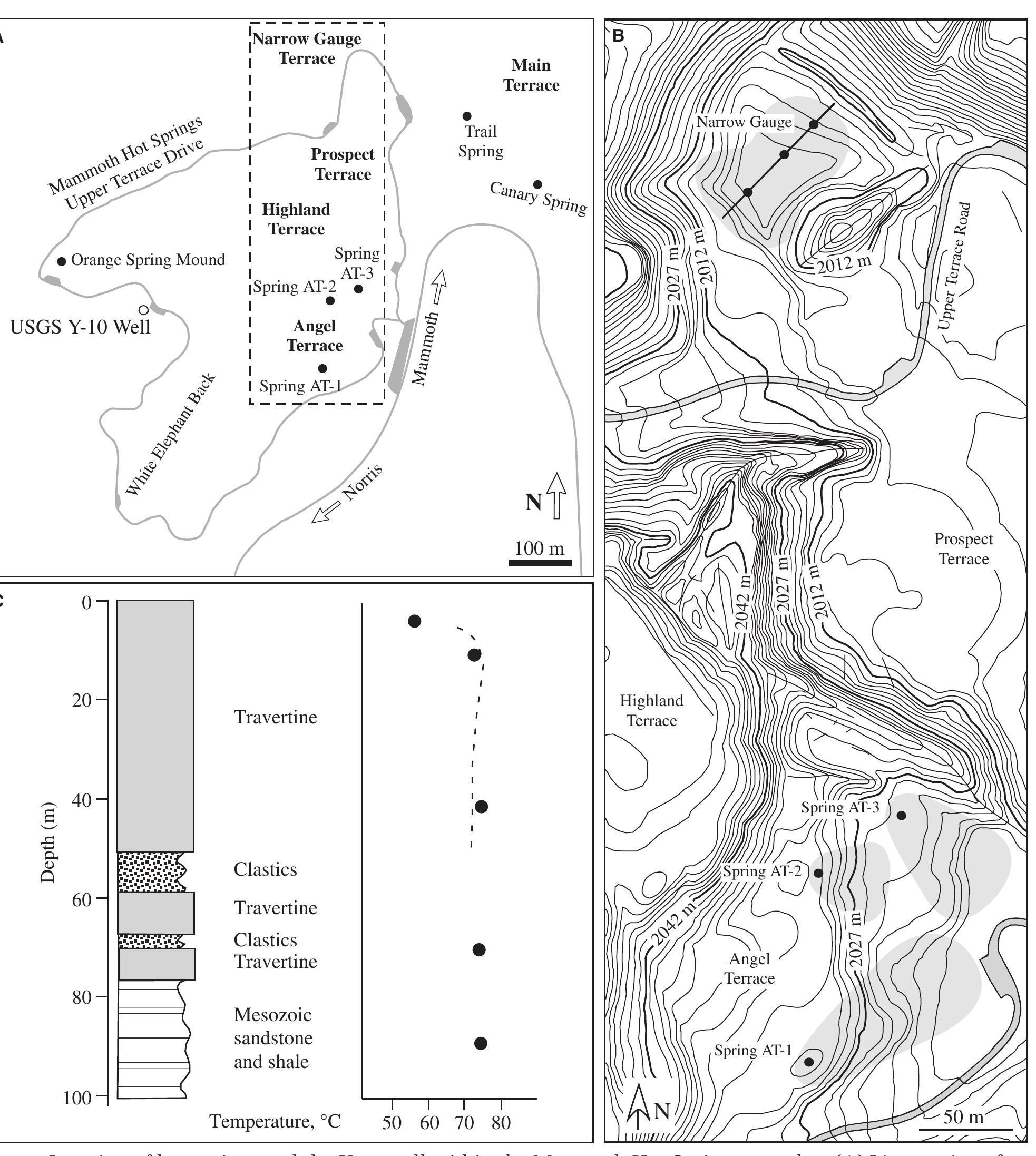
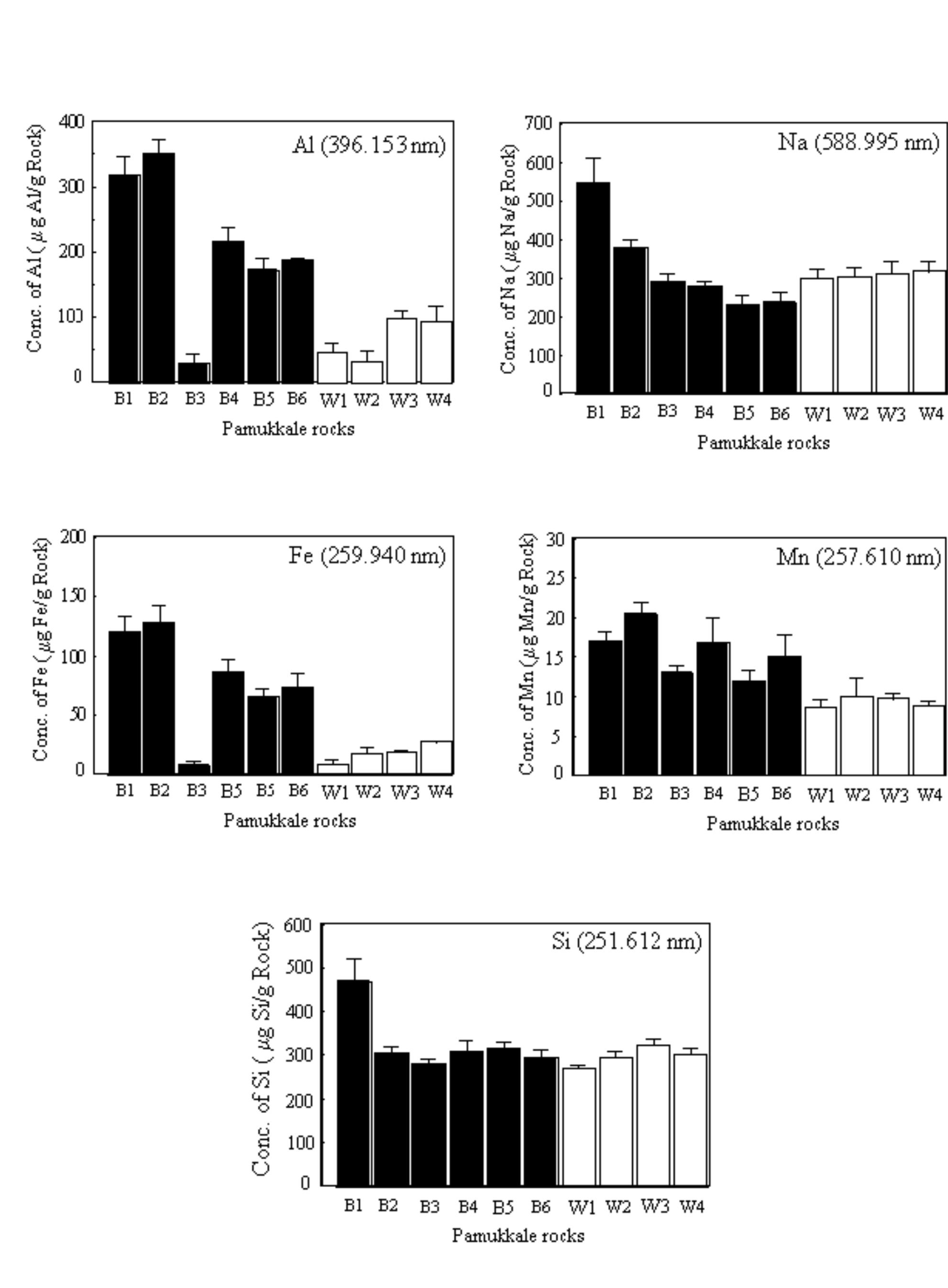
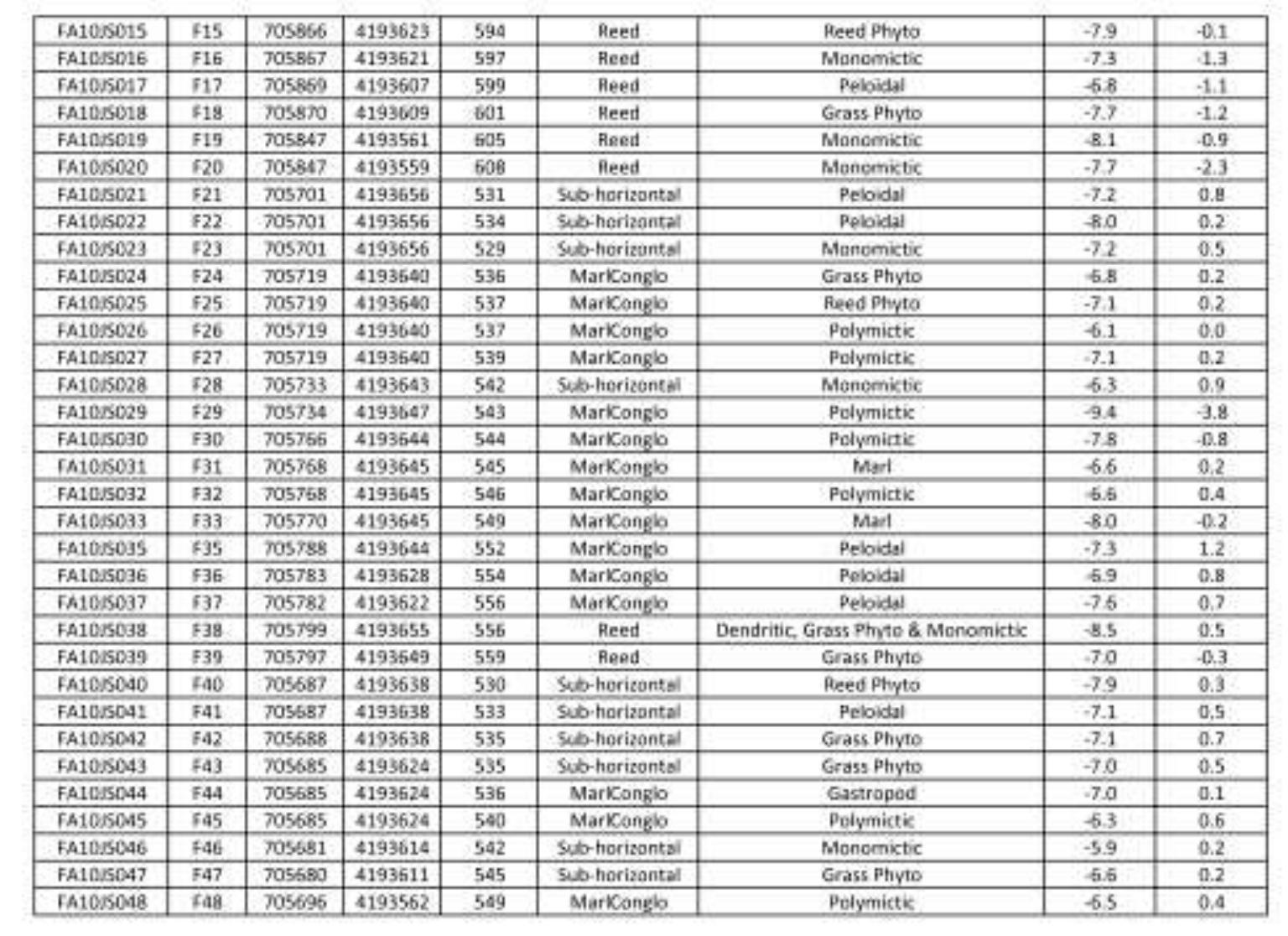
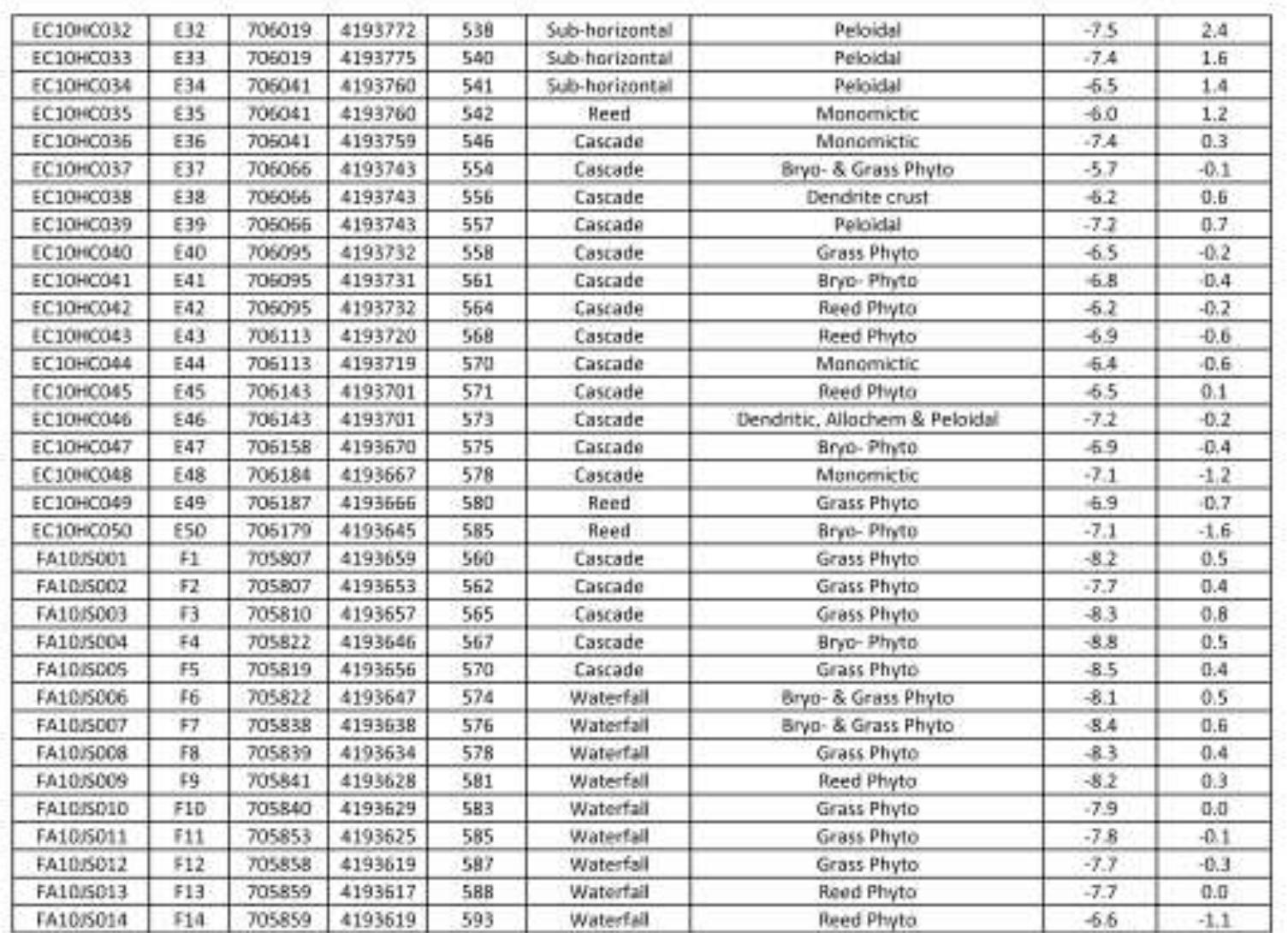
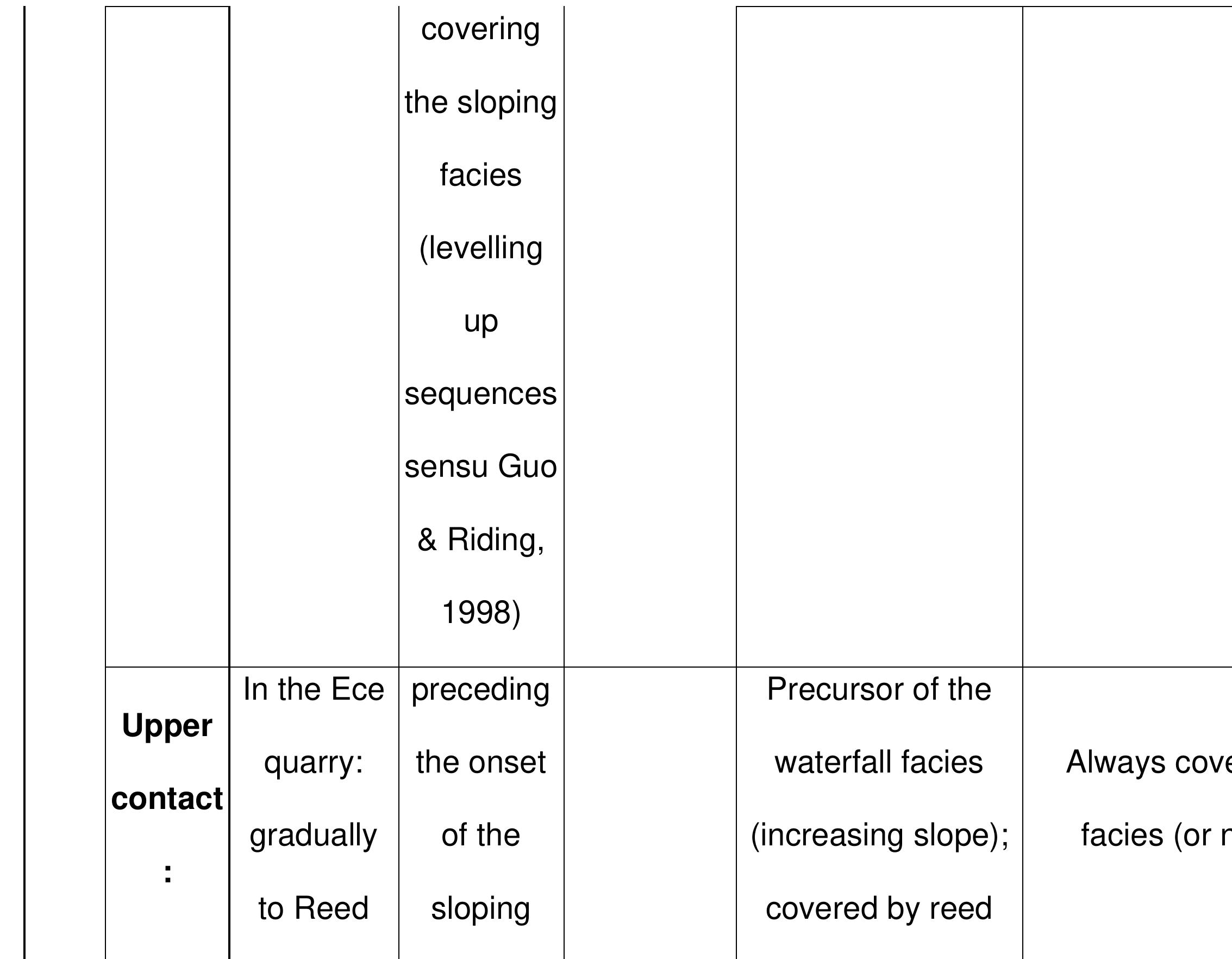
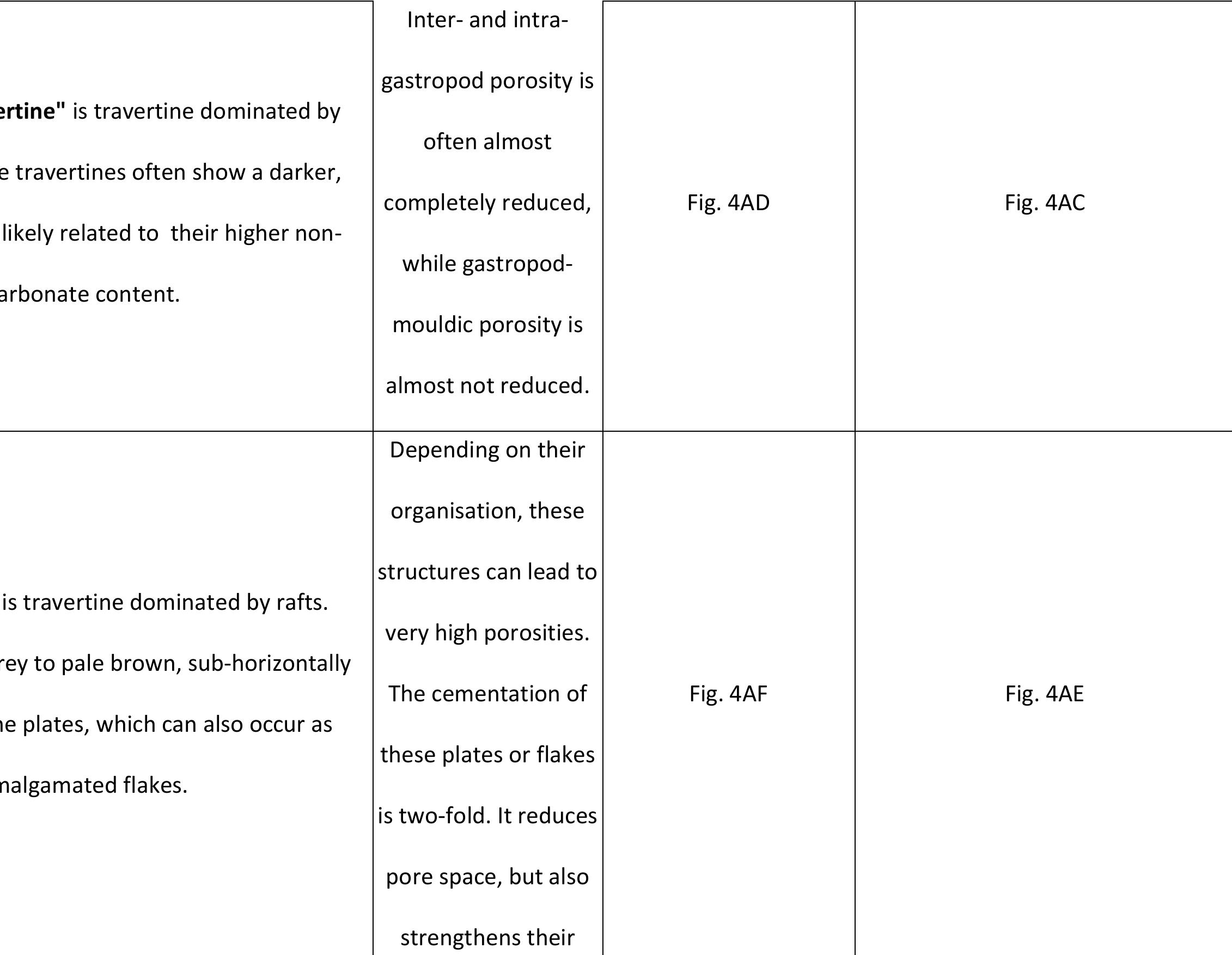
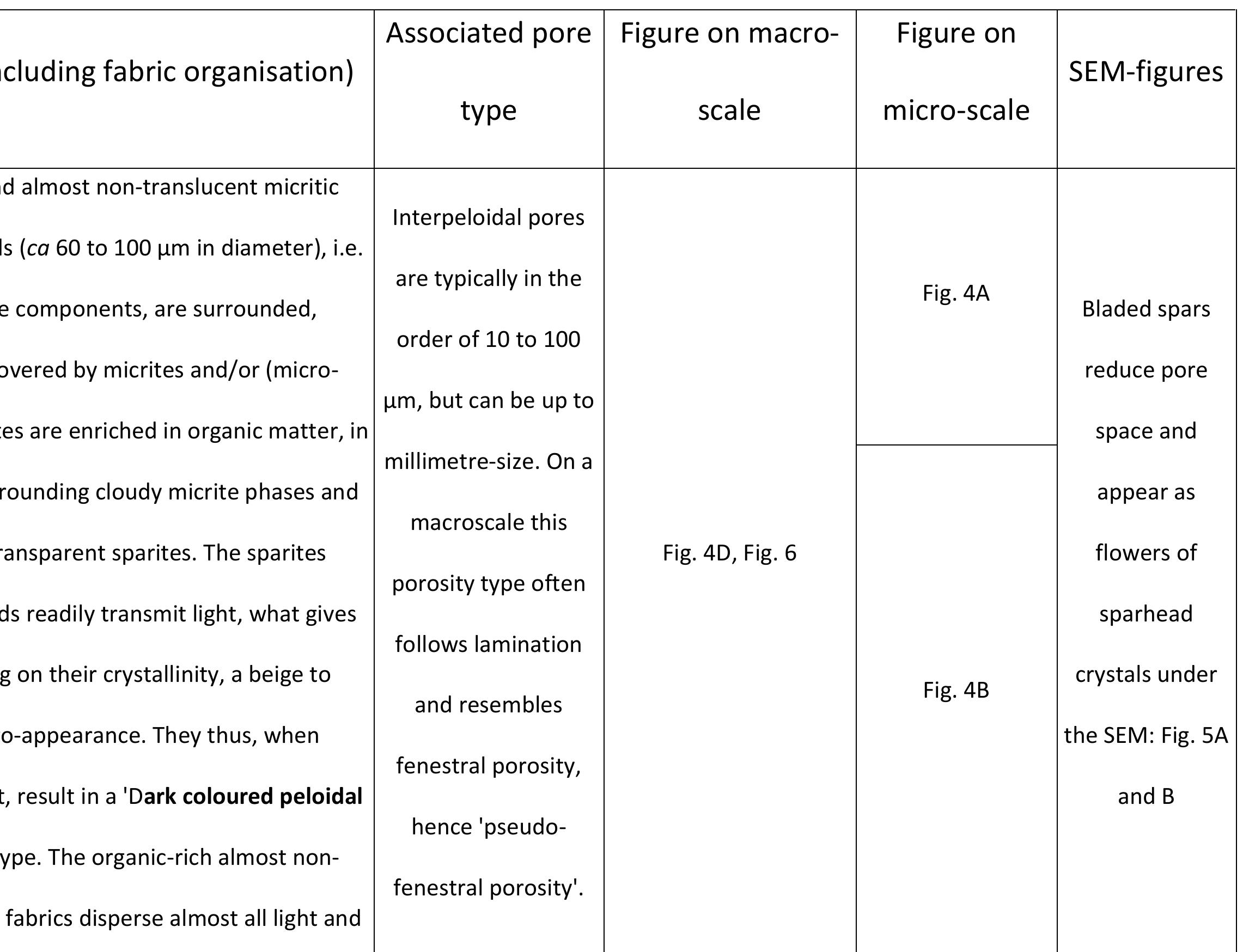
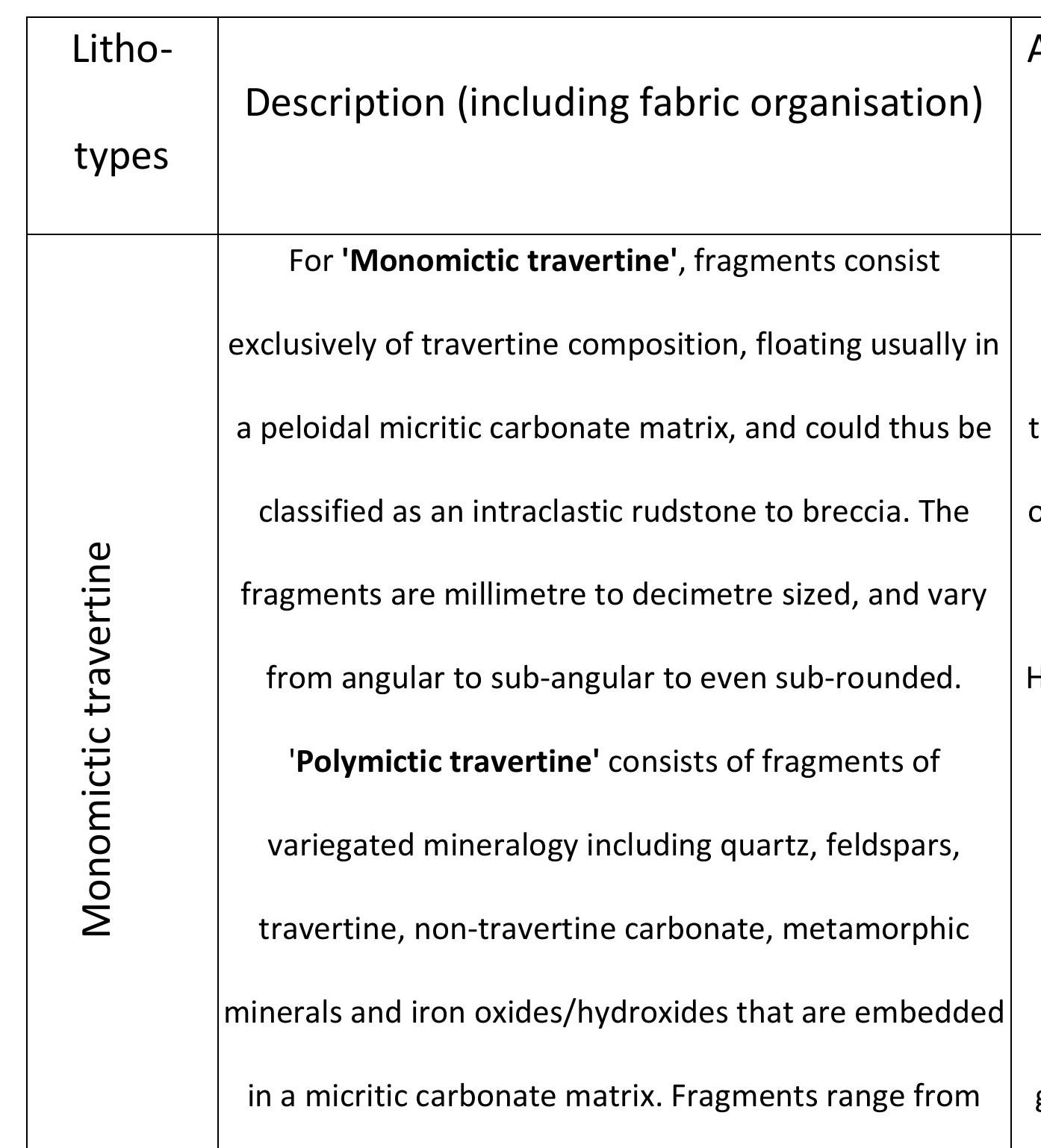
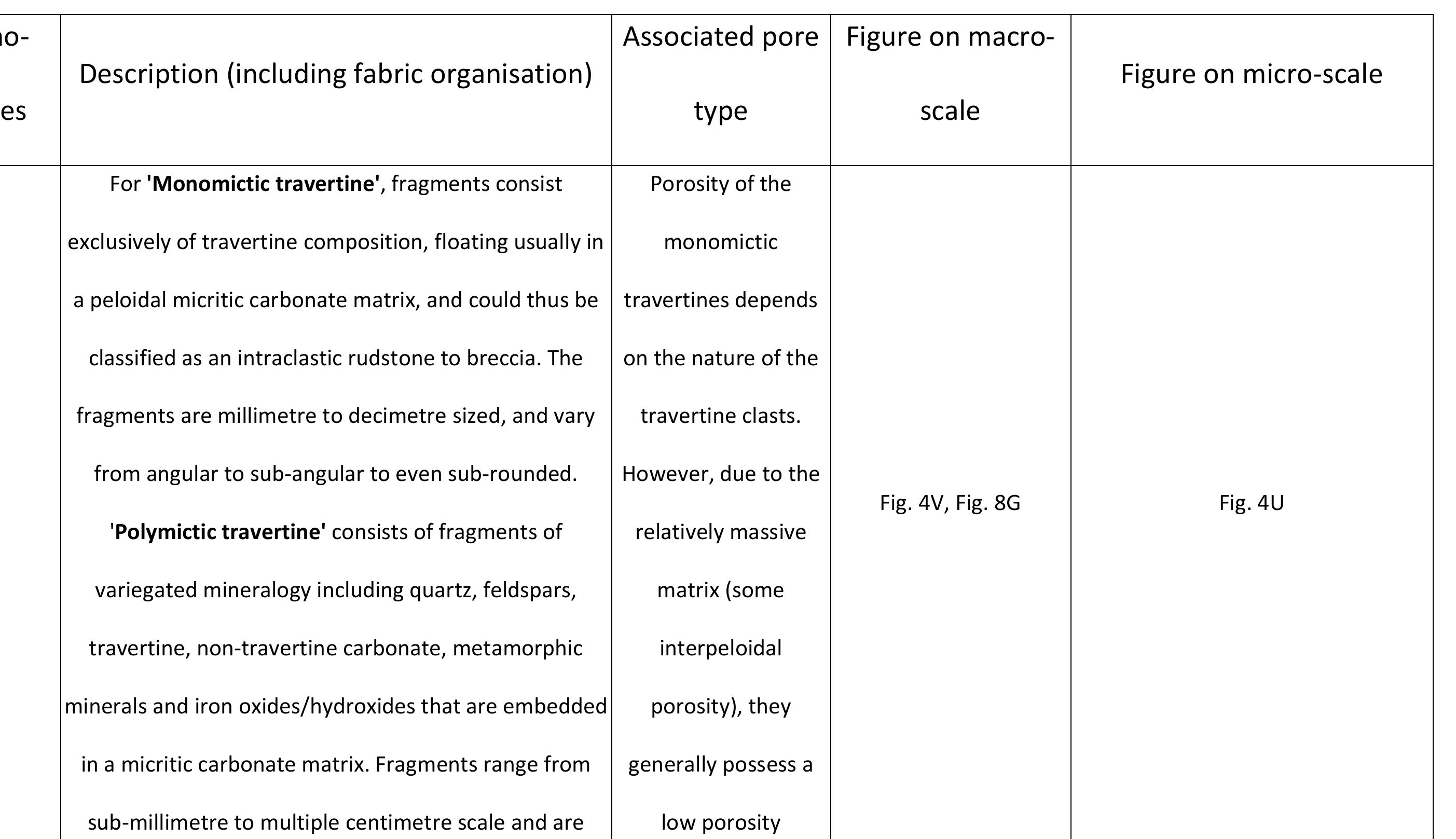
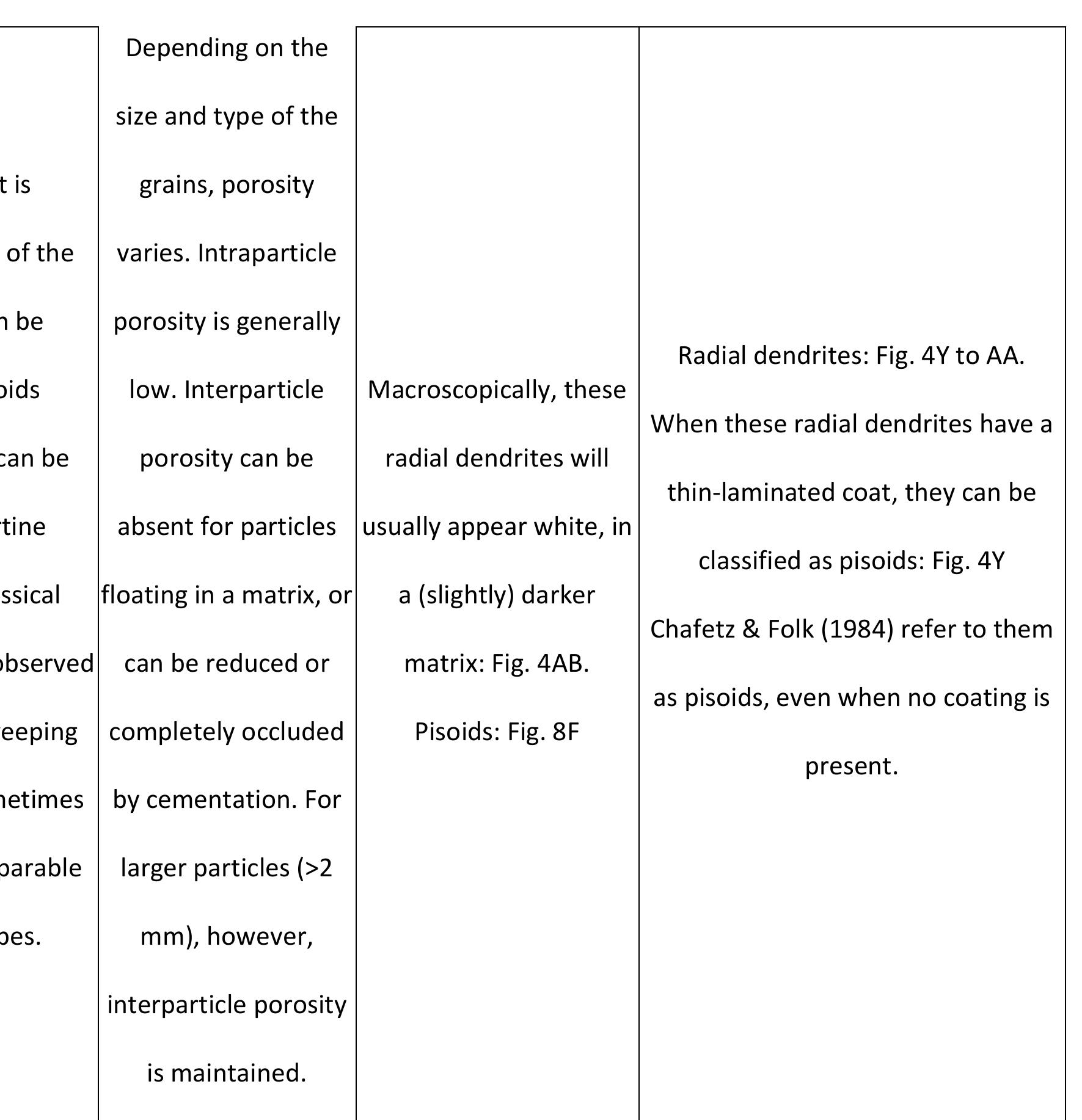
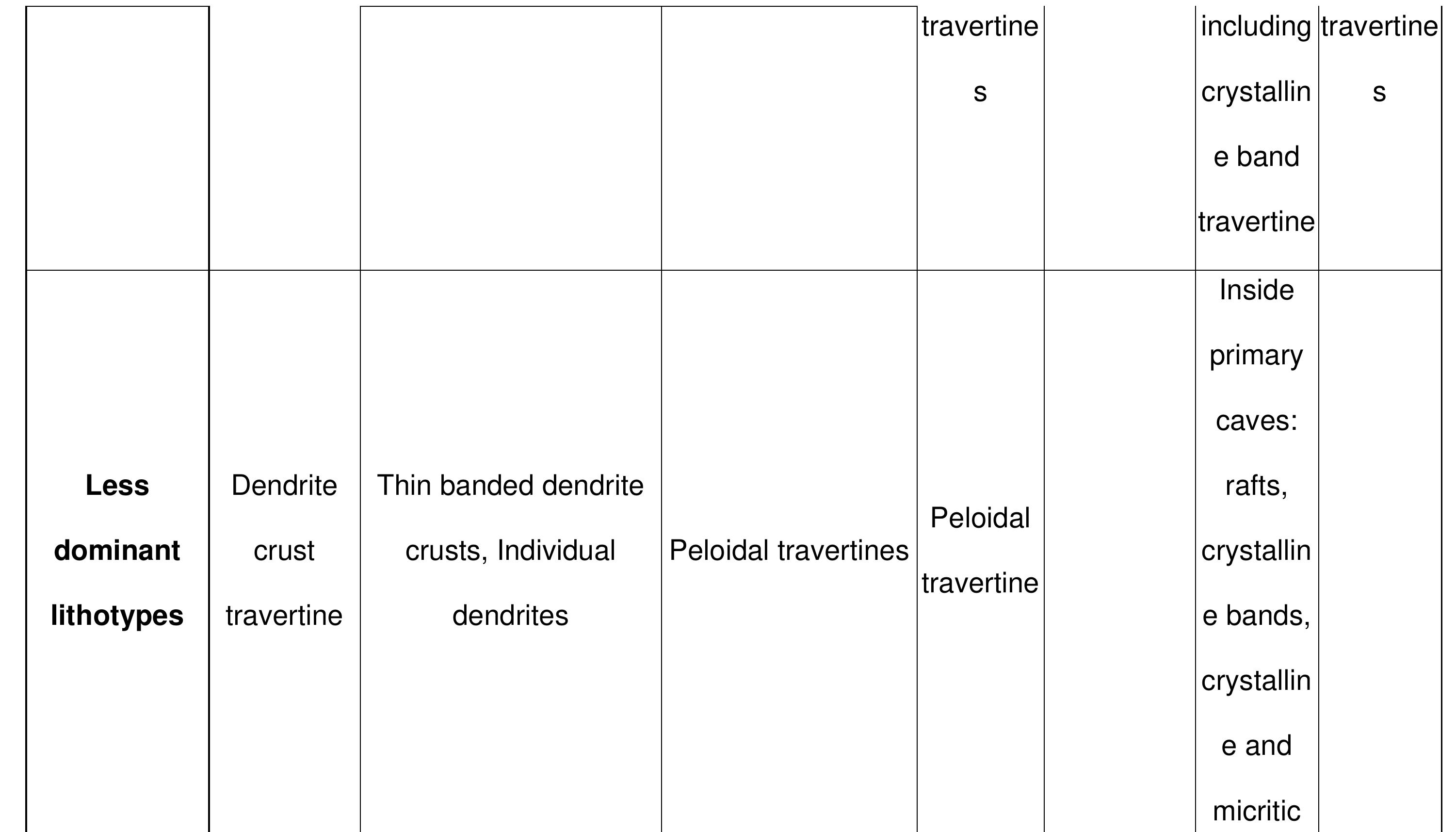
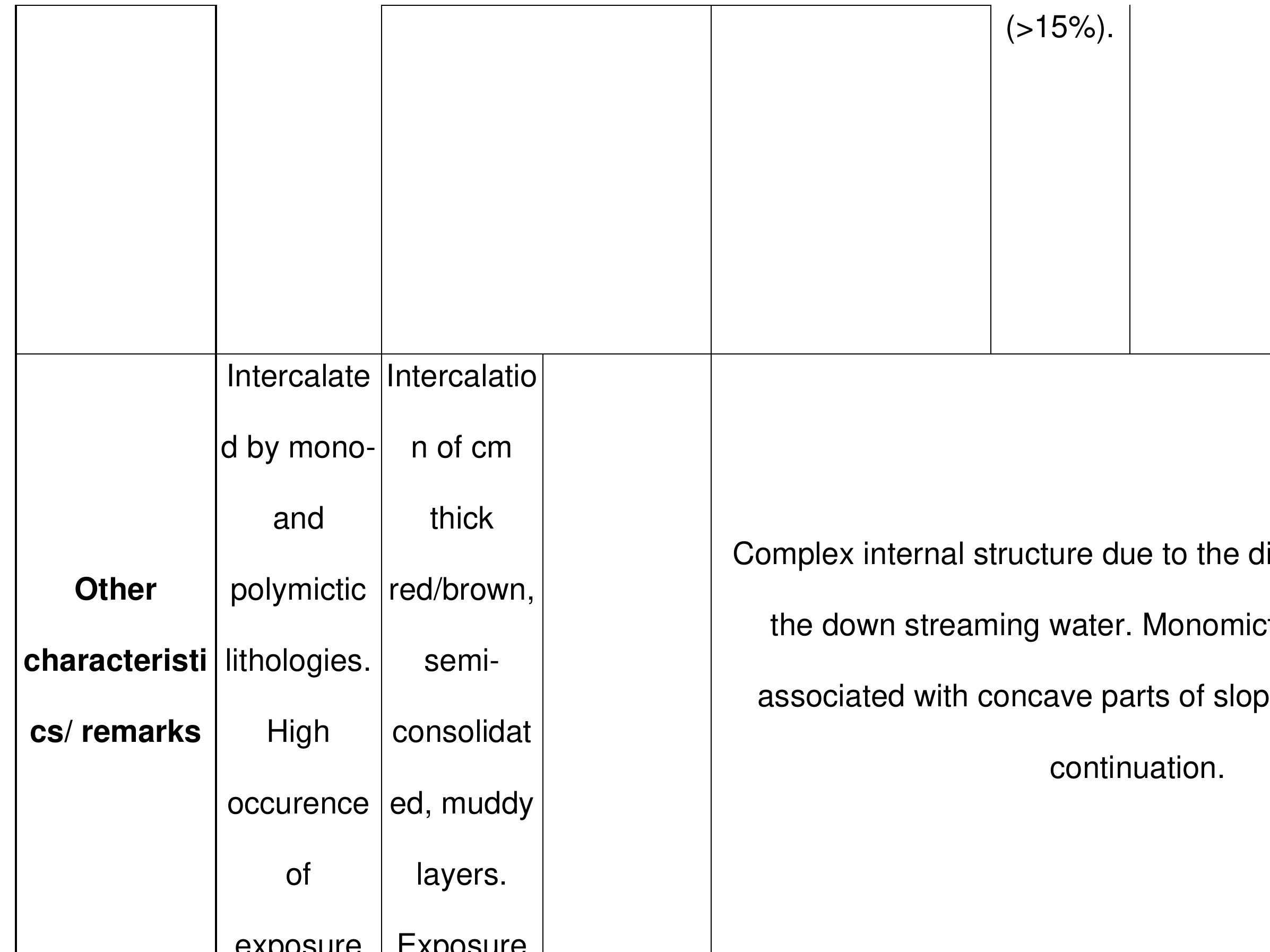
Key research themes
1. How can mass spectrometric and nuclear reaction techniques enhance precision in stable isotope analysis of trace elements and isotopes?
Precise and accurate measurement of stable isotopes at micro to ultra-trace levels is critical for advancing fields ranging from geochemistry to nuclear physics and environmental sciences. This research theme focuses on development, optimization, and validation of analytical methodologies—primarily nuclear reaction-based activation analysis and various mass spectrometry techniques including MC-ICP-MS and laser-ablation coupled mass spectrometry—to improve sensitivity, specificity, and reproducibility in stable isotope determinations, especially when sample quantities are limited or matrix complexity is high. Enhancing methodological rigor allows isotope ratios to be used more effectively as tracers in complex systems and expands the scope of isotope applications.
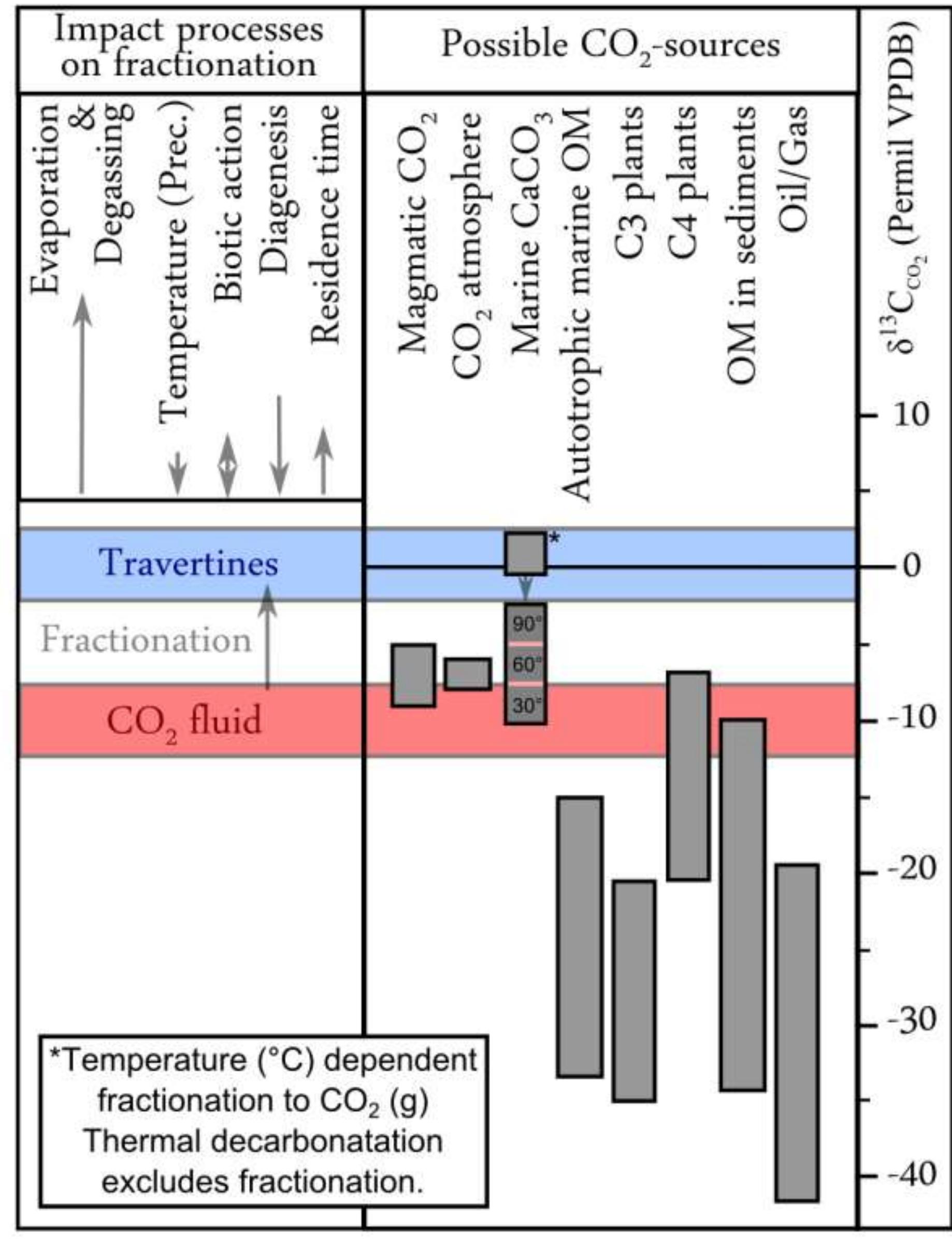
2. What are the causes and implications of kinetic and equilibrium isotope fractionation effects in natural and anthropogenic carbonate systems and biological contexts?
Stable isotope fractionation in carbonates and biological materials is influenced by a complex interplay of kinetic, equilibrium, and environmental factors. This theme encompasses investigations into the mechanistic origins, environmental dependencies, and applied implications of isotope fractionations—especially for oxygen, carbon, nitrogen, and sulfur isotopes—in contexts ranging from carbonate precipitation under high-pH anthropogenic conditions to isotope signals reflecting human diseases. Understanding these processes is critical for reconstructing paleoenvironments, diagnosing pathologies, and interpreting isotopic records influenced by both enzymatic and abiotic reactions.
3. How can stable isotope applications be optimized through software standardization and uncertainty management in the measurement and calibration of isotope ratios?
Achieving reproducible, accurate stable isotope data requires standardized computational frameworks for data reduction, analysis, and uncertainty evaluation, particularly in complex or time-resolved isotopic measurements. Additionally, precise uncertainty propagation and adherence to international normalization standards underpin reliable calibration of measurements against reference materials. This theme focuses on software solutions for harmonizing isotope data workflows, best practices in reference material use, calibration curve construction, and uncertainty quantification to ensure data comparability across laboratories and improve confidence in isotopic interpretations across scientific fields.



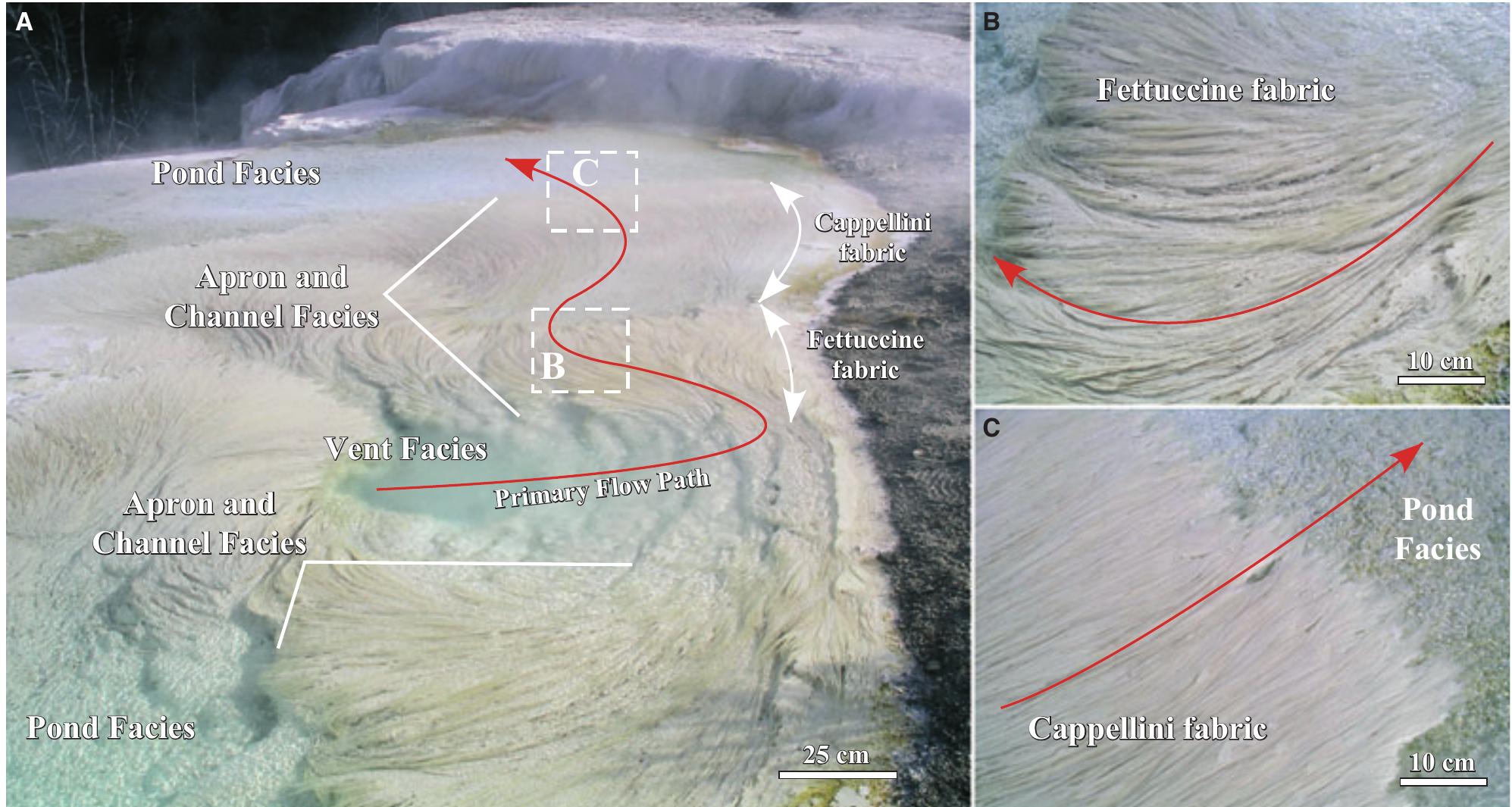
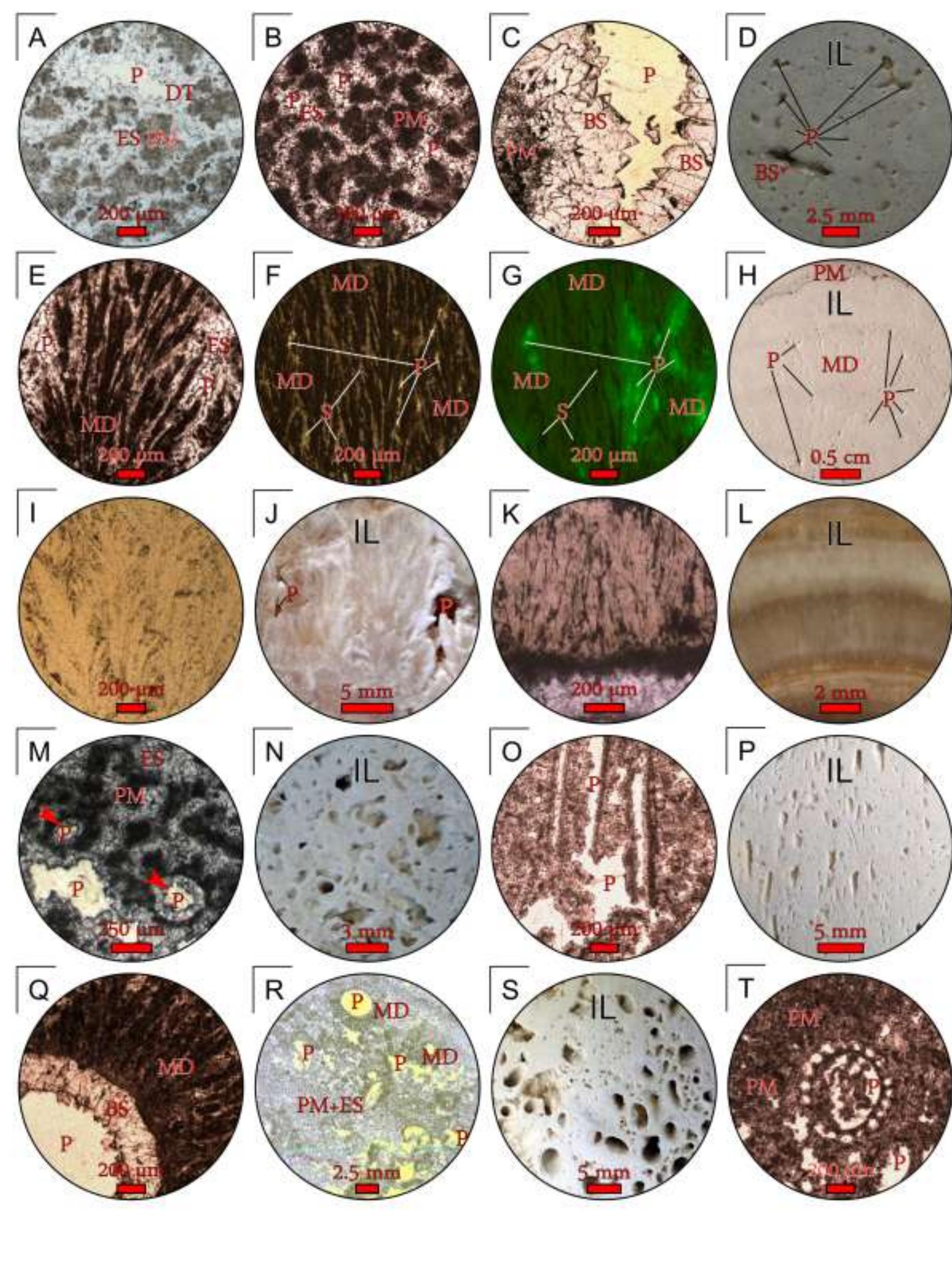
![Fig. 10. Field photographs of the MHS travertine vent facies. (A) Overview of the vent facies and apron and channel facies at Spring AT-1 in July 2003. (B) Hot-spring water emerges from the vent at 73°C to form a turquoise pool floored with mounded white travertine deposits. (C) Gradational contact of the mounded white travertine of the vent facies with the characteristic beige sinuous streamer travertine of the apron and channel facies. (D) The irregular bumpy surface of the mounded travertine deposits in the transitional region from the vent to the apron and channel facies [enlargement of the area shown in panel (C)]. The vent facies grades downstream into the apron and channel facies, which is floored by sinuous deposits of travertine called ‘streamers’ (Farmer, 2000) that reach tens of centimetres in length (Fig. 12A and B). Each streamer filament is composed of aragonite needle botryoids that are significantly smaller (<20 1m x 0-5 um) and less The vent facies is a shallow bowl-shaped depression (<5 m in diameter) surrounding the vent orifice (<3 cm in diameter) that is encrusted](https://figures.academia-assets.com/105373650/figure_008.jpg)
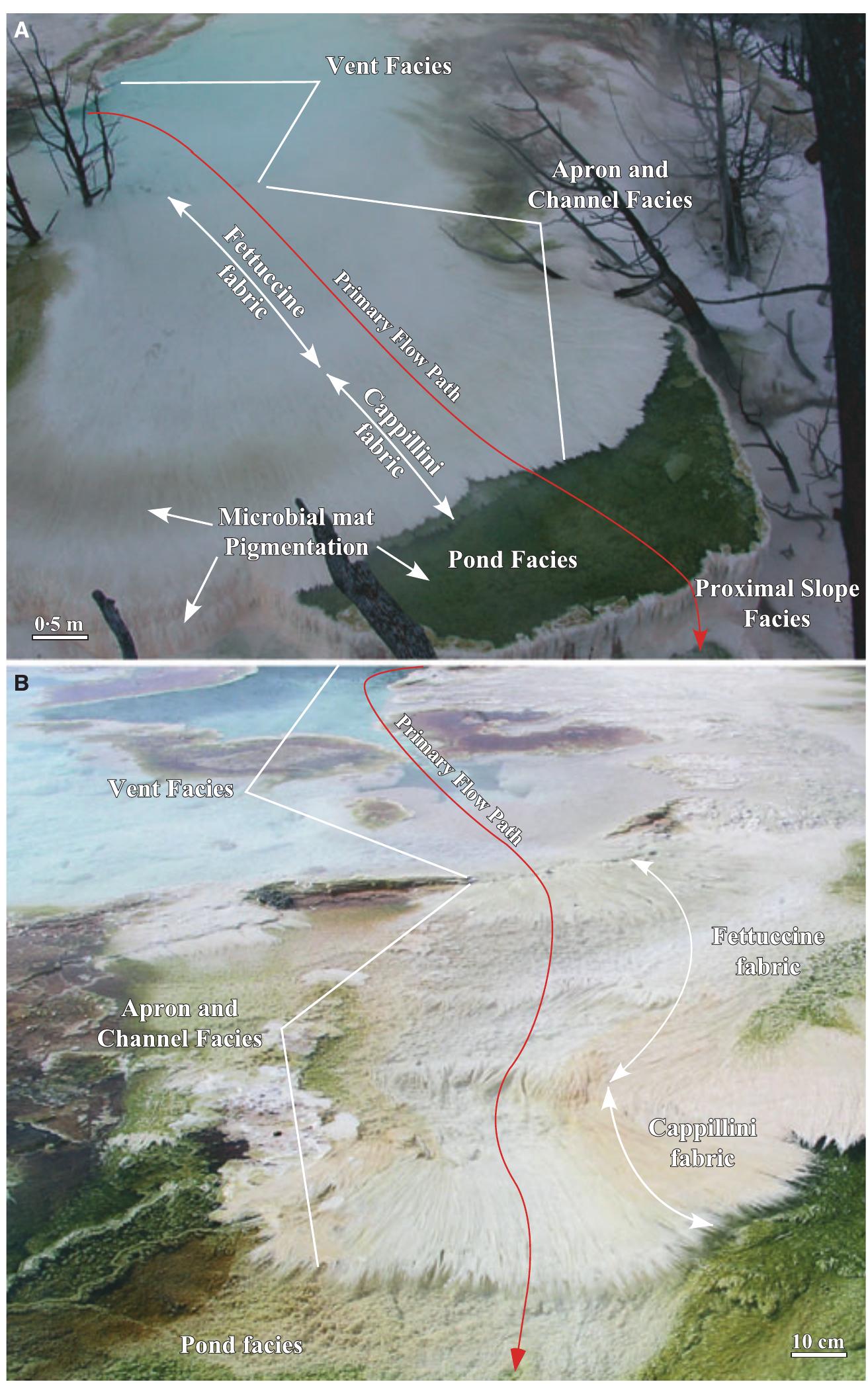
![Fig. 12. Field photographs of the Mammoth Hot Springs travertine apron and channel facies at Angel Terrace Spring AT-1 in January 2004. (A) Overview of the apron and channel facies. (B) The characteristic sinuous streamer fabric of the apron and channel facies [enlargement of the area shown in panel (A)]. (C) Schematic cross-section of the abrupt transition from the apron and channel facies to the pond facies, with filamentous microbial mats that grow near the water surface and extend into the deeper water of the pond. During periods of increased spring-water outflow from the vent, the apron and channel facies travertine progrades into the pond at and near the water surface, forming a ledge that will eventually completely cover the pre-existing terracette if hot-spring water flow remains high. (D) Photograph of the transition from non-encrusted to travertine encrusted microbial filaments.](https://figures.academia-assets.com/105373650/figure_010.jpg)






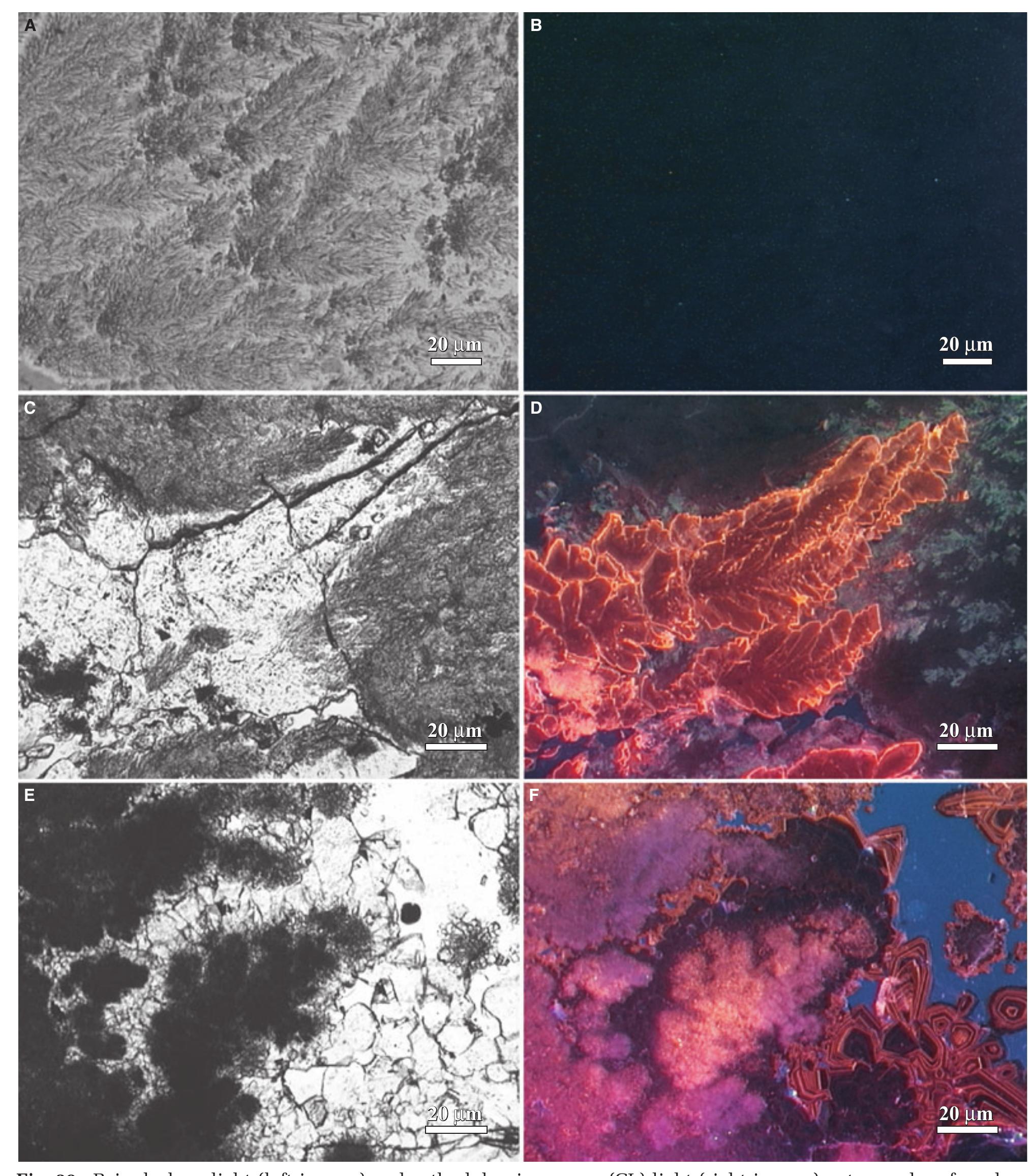
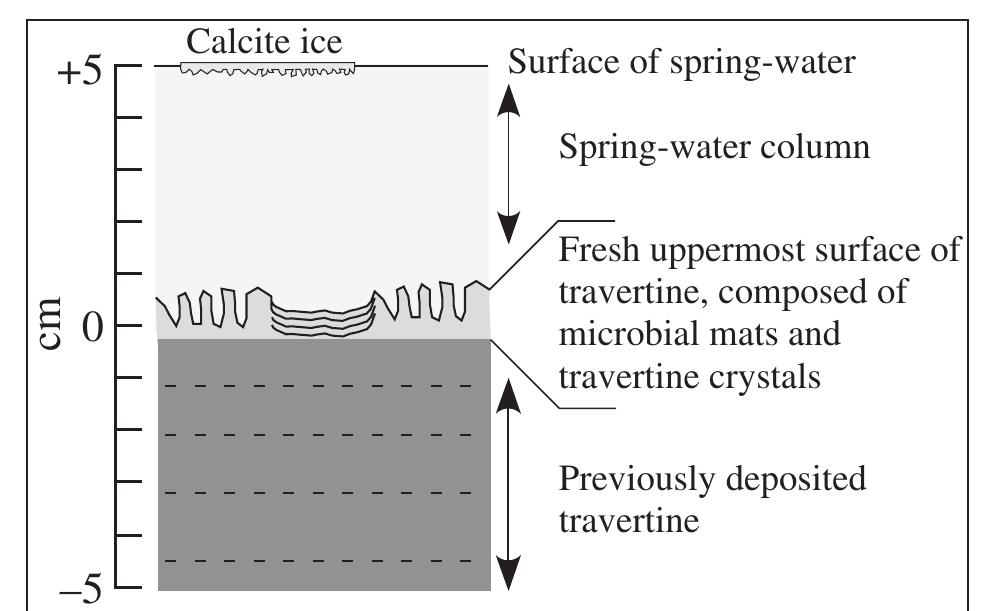
![Fig. 14. Petrography of travertine comprising encrusted bubbles, calcite ice and microterracette pond lips. (A) and (B) Paired polarized-light photomicrograph and SEM image of aragonite and calcite crystals encrusting a hot-spring water bubble in pond facies travertine [the field photograph is shown in panel (F)]. (C) and (D) Paired polarized-light photomicrograph and SEM image of aragonite and calcite crystals comprising calcite ice sheets. (E) and (F) Field photograph and plane-light photomicrograph of the lip of a microterracette in the proximal-slope facies. (ii) low-temperature (<57°C) ponds that form ridged networks of microcrystalline aragonite 1 to 2 cm in height (Farmer, 2000). Thin carbonate ‘ice-sheets’ and calcified bubbles (Fig. 13E and F) composed of 50 to 100 pm diameter blocky to columnar calcite crystals and small (<10 um) acicular aragonite (Fig. 14A to D) precipitate at the pond air—water interface (Allen & Day, 1935;](https://figures.academia-assets.com/105373650/figure_012.jpg)


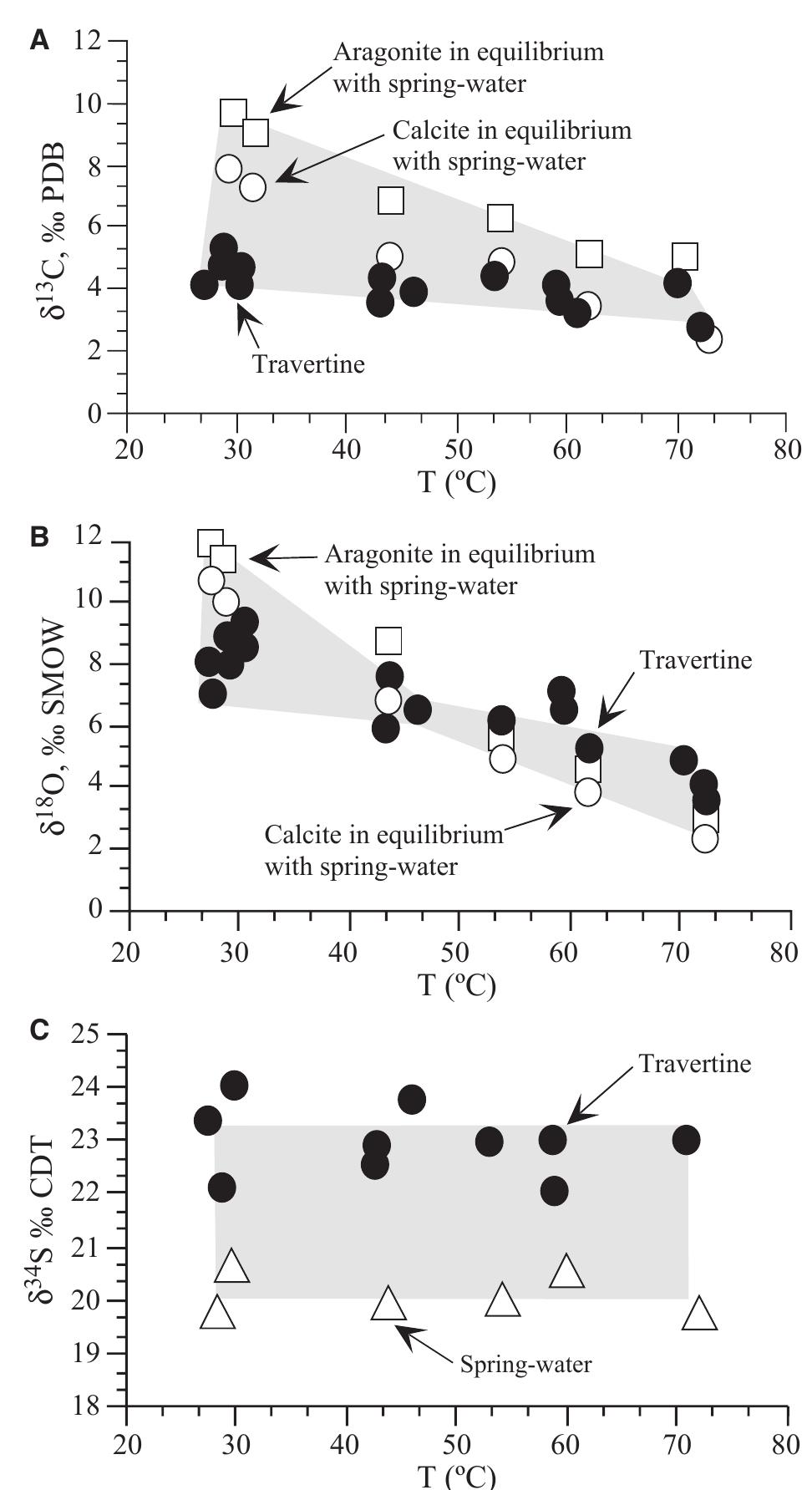




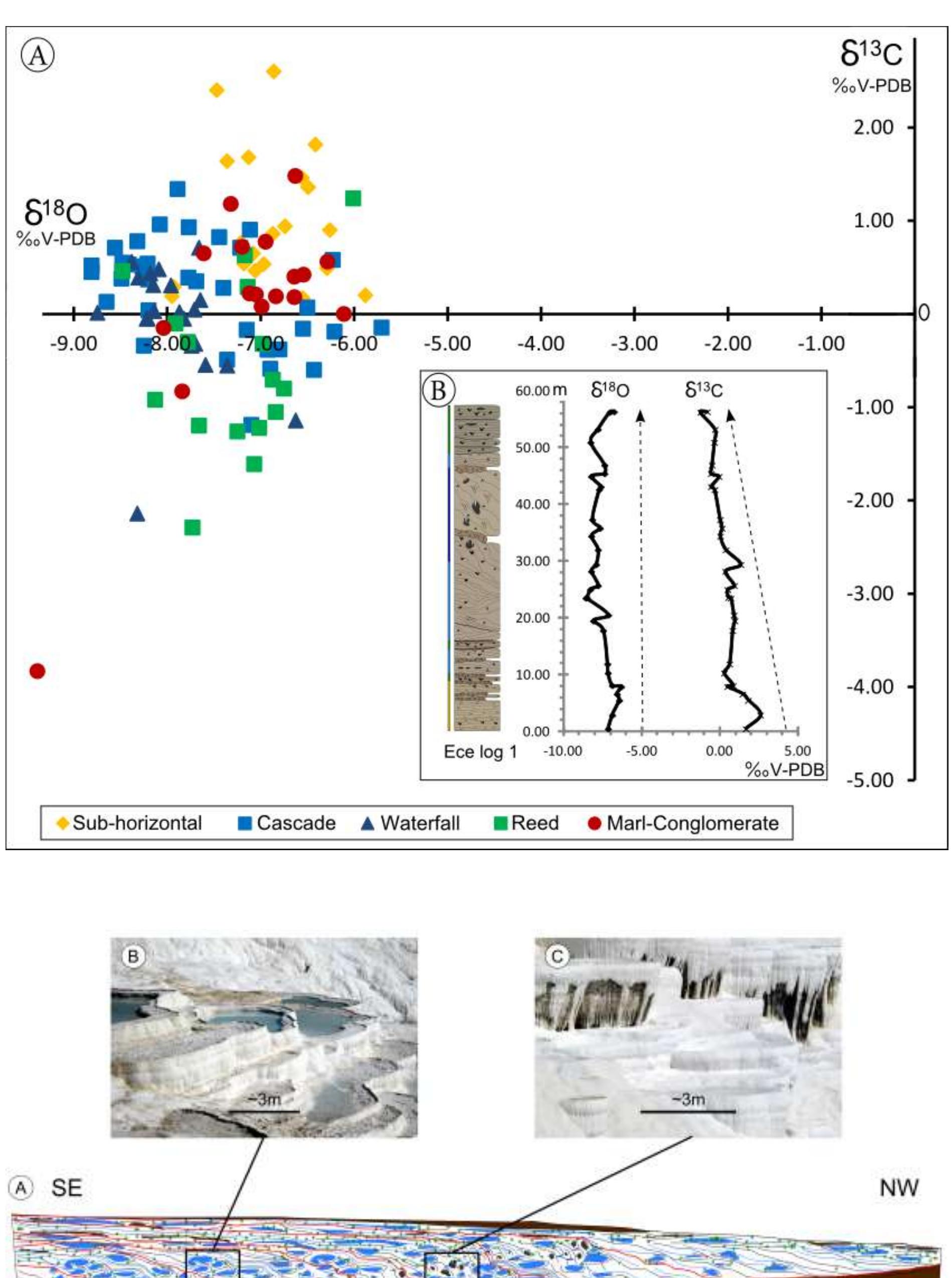
![Fig. 18. Travertine and spring-water geochemistry at Mammoth Hot Springs Spring AT-1 and Spring AT-3. (A) The distribution of 343 triplicate paired measurements of spring-water temperature and pH. Error bars indicate one standard deviation (modified from Veysey et al., 2008). (B) Co-variations in spring-water total dissolved inorganic carbon (DIC) versus its 57°C value. (C) Co-variations in spring-water oxygen versus carbon-isotopic composition. (D) Co-variations in travertine oxygen versus carbon-isotopic composition [(B), (C) and (D) modified from Fouke et al., 2000].](https://figures.academia-assets.com/105373650/figure_016.jpg)

















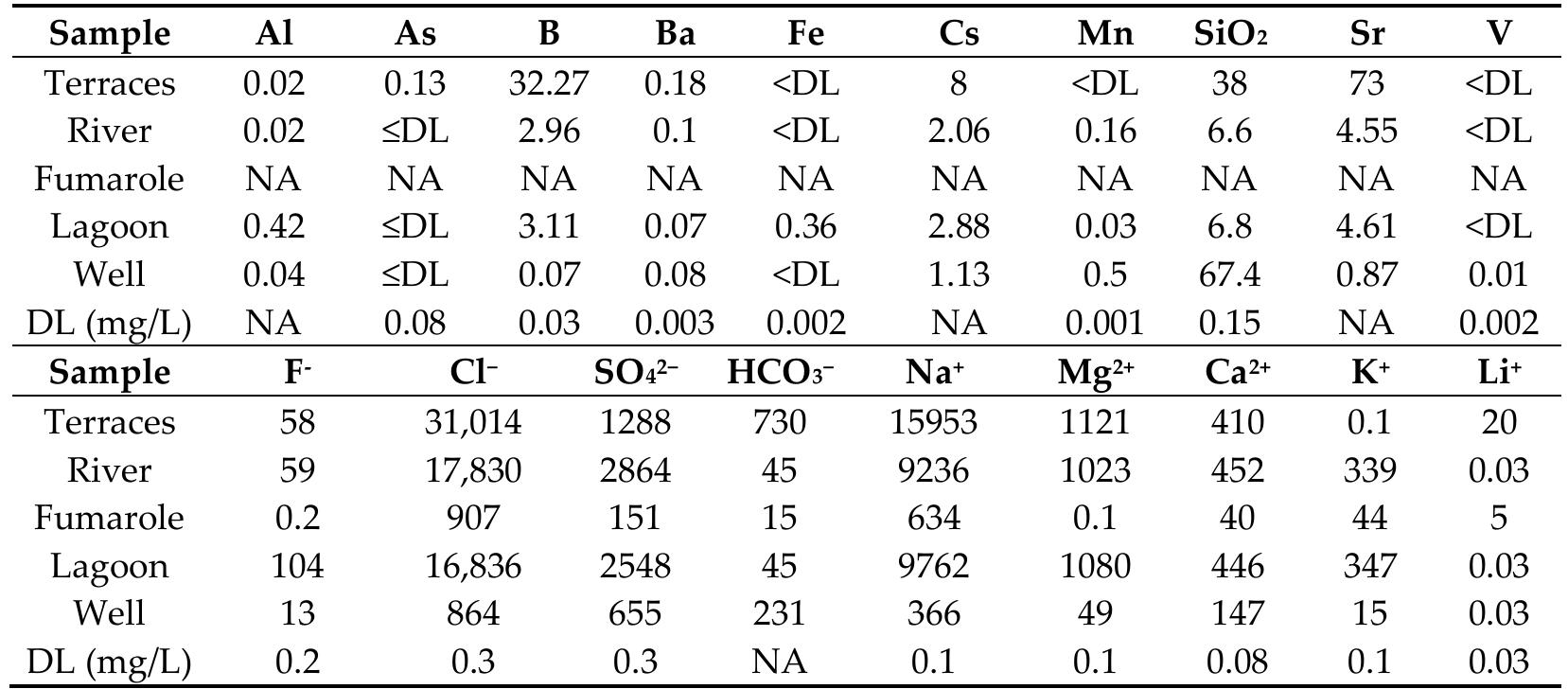
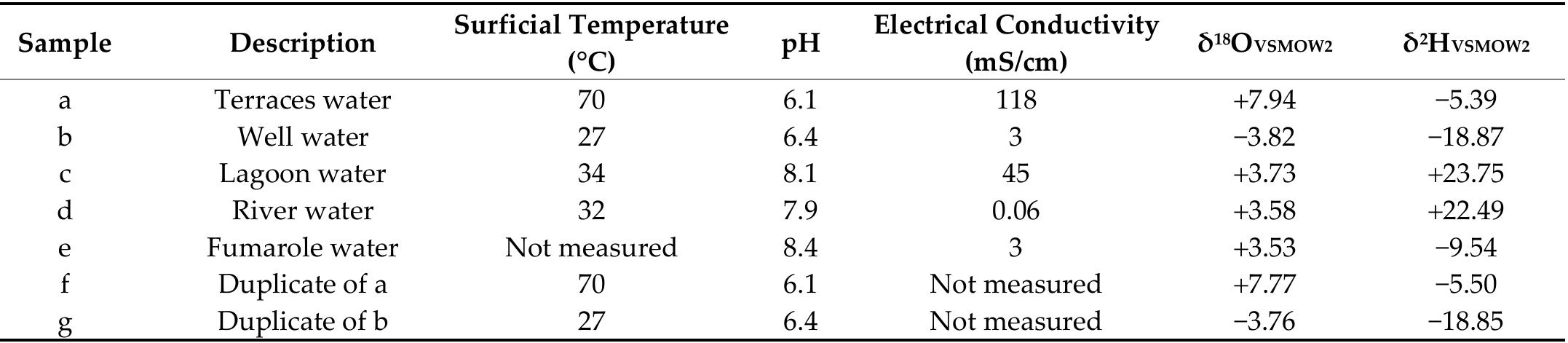
![Figure 10. Lanthanide concentrations of four aragonite samples and one sample of native sulfur normalized to chondrite CI meteorites (Wasson and Kallemeyn 1988) [52]. 4.2. Water System A Durov diagram [54] was constructed to visualize the chemis try of maj or ions and ) compare it with pH and total dissolved solids (TDS). The pH of the hydrothermal em- nation water in the aragonite terraces is slightly acidic (Figure 11a), s is the water from the well (meteoric water). In comparison, the wa re lagoon is slightly alkaline (Table 4). The temperature recorded in ter from t the three- approaching neutral, he river and point intake well, river, and lagoon) is similar, being between 27 °C and 34 °C (the average annual 1aximum temperature recorded at the climatological station of Maj agual in 7 Tamiahua is 9 °C; https://smn.conagua.gob.mx/es/informacion-climatologica-por-estado?estado=ve accessed 15 June 2022), while the minimum surface temperature of the aragonite terraces yas registered as 70 °C, similar to the outer part of the fumaroles in which steam ema- ates. Table 4 contains the physicochemical data taken and record ed in the field of the yater samples, except for the condensate from the fumarole, which was not determined ue to the limited amount of water collected.](https://figures.academia-assets.com/89264800/figure_010.jpg)





![Figure 2. Geologic map of the area. Modified from Maldonado Lee et al., (2004) [32].](https://figures.academia-assets.com/89264800/figure_002.jpg)
![Figure 11. (A) Durov diagram showing the dominant ions in relation to pH and Total Dissolved Solids (TDS) of the water samples from Tamiahua Lagoon. The plot was created with the AQQA (version 1.2.0). (B) Na-K-Mg diagram showing the chemical equilibrium of the waters of Tamiahua Lagoon. The plot was created with the Liquid Analysis v3 program from Powell and Cumming 2010 [58].](https://figures.academia-assets.com/89264800/figure_011.jpg)
![Table 2. Trace element composition of travertine samples including lanthanide concentrations of four aragonite samples and one native sulfur sample (Tami 2AS) normalized to chondrite-type me- teorites [52].](https://figures.academia-assets.com/89264800/table_002.jpg)
![Figure 13. Isotopic composition of different types of waters in Tamiahua Lagoon. Values for the waters from Los Humeros springs and wells were obtained from Martinez-Serrano [66]. water becomes enriched due to exchange with the surrounding host rocks during rising thermal waters. Furthermore, the loss of H by gases such as H25, Hz, or CH4—associated with hydrocarbon emanations —also provokes deuterium enrichment in water relative to the rocks and precipitates [70]. It is important not to discard the possible influence of deep brines associated with the hydrocarbon layers that were mixed with the surface water in the lagoon area due to the spill that occurred approximately 110 years ago.](https://figures.academia-assets.com/89264800/figure_014.jpg)
![Figure 14. Comparison of Tamiahua Ba/Sr values (purple stars) with the travertine and tufa litera- ture data (dashed area), modified from Teboult et al., (2016) [13].](https://figures.academia-assets.com/89264800/figure_015.jpg)

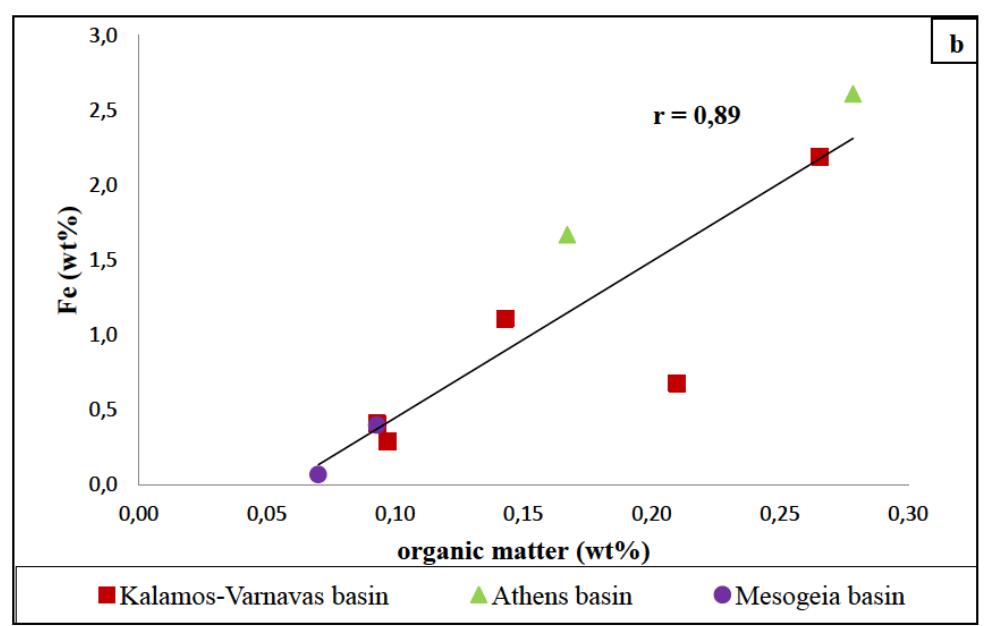

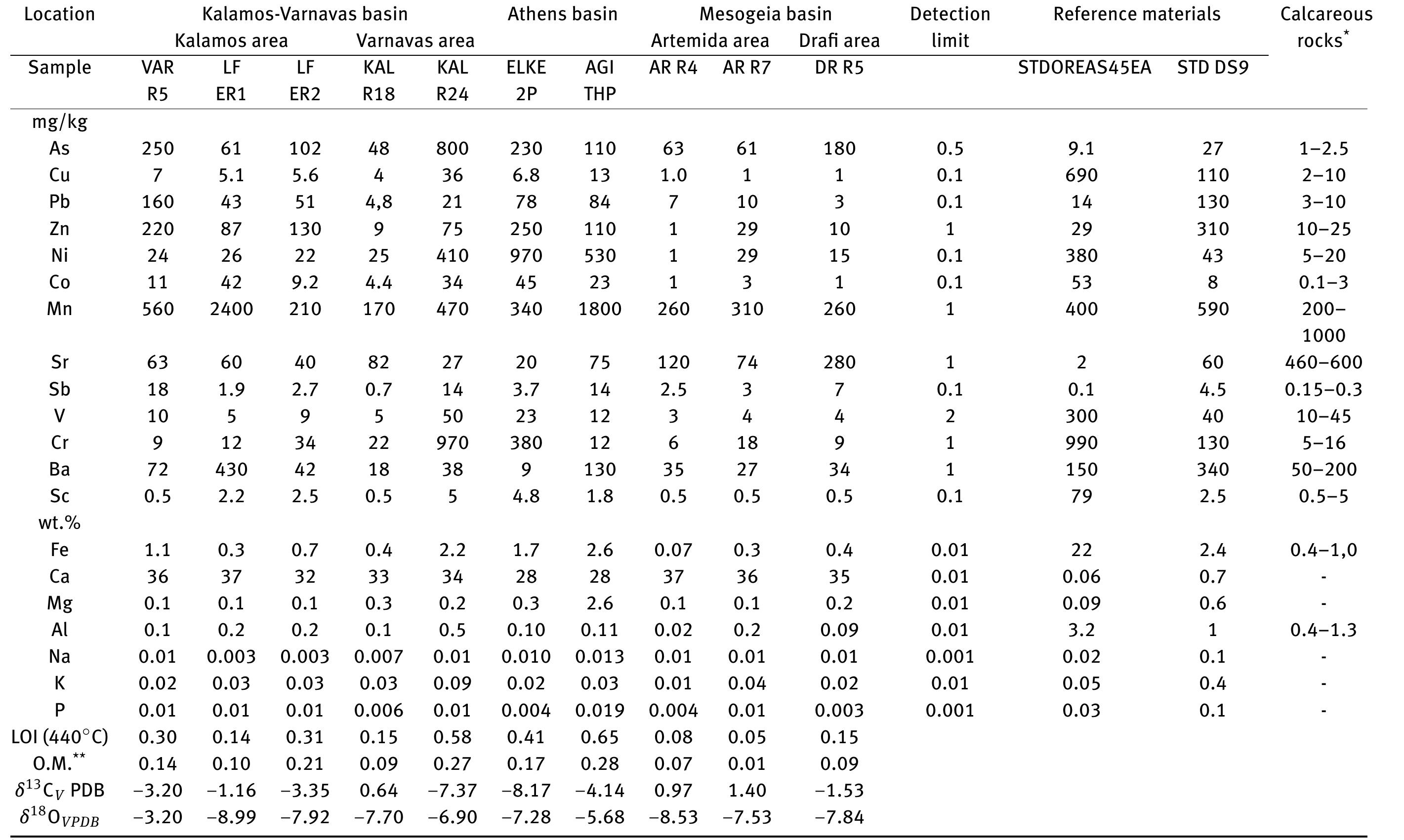

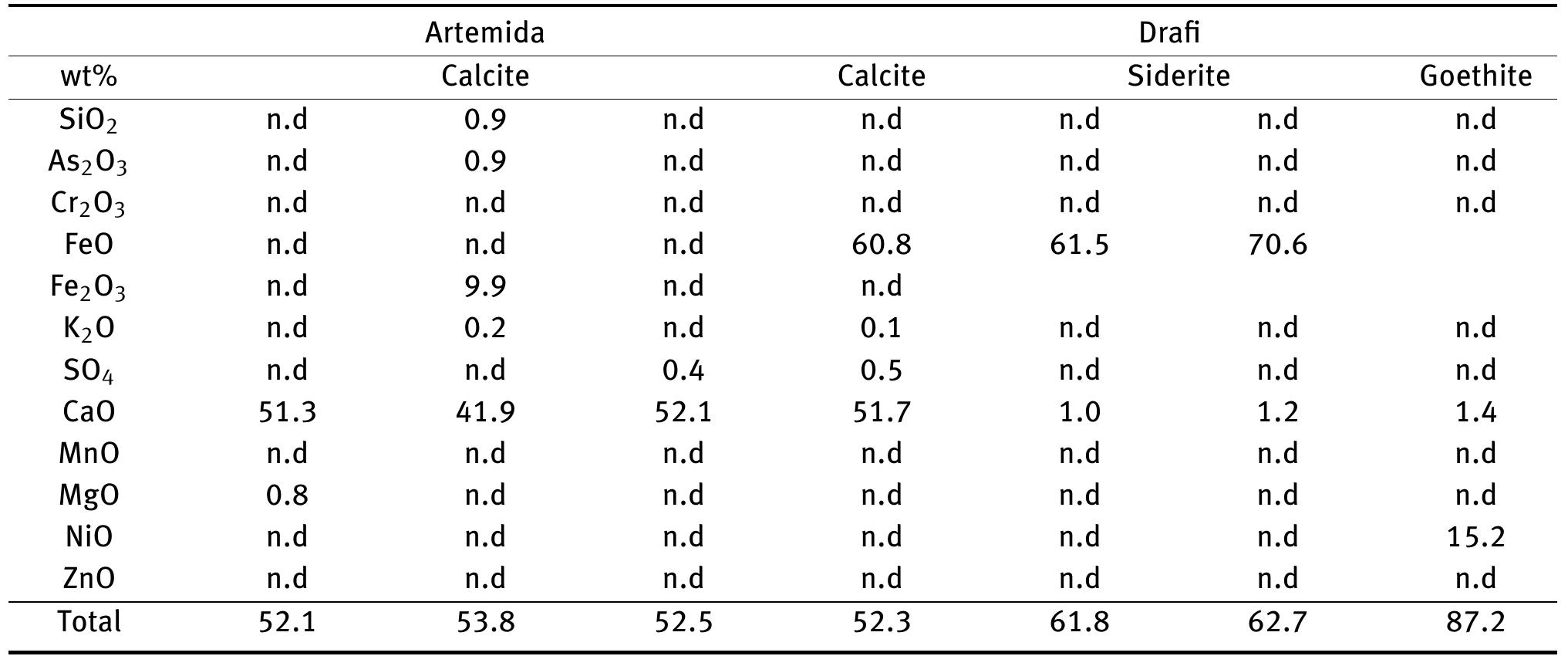
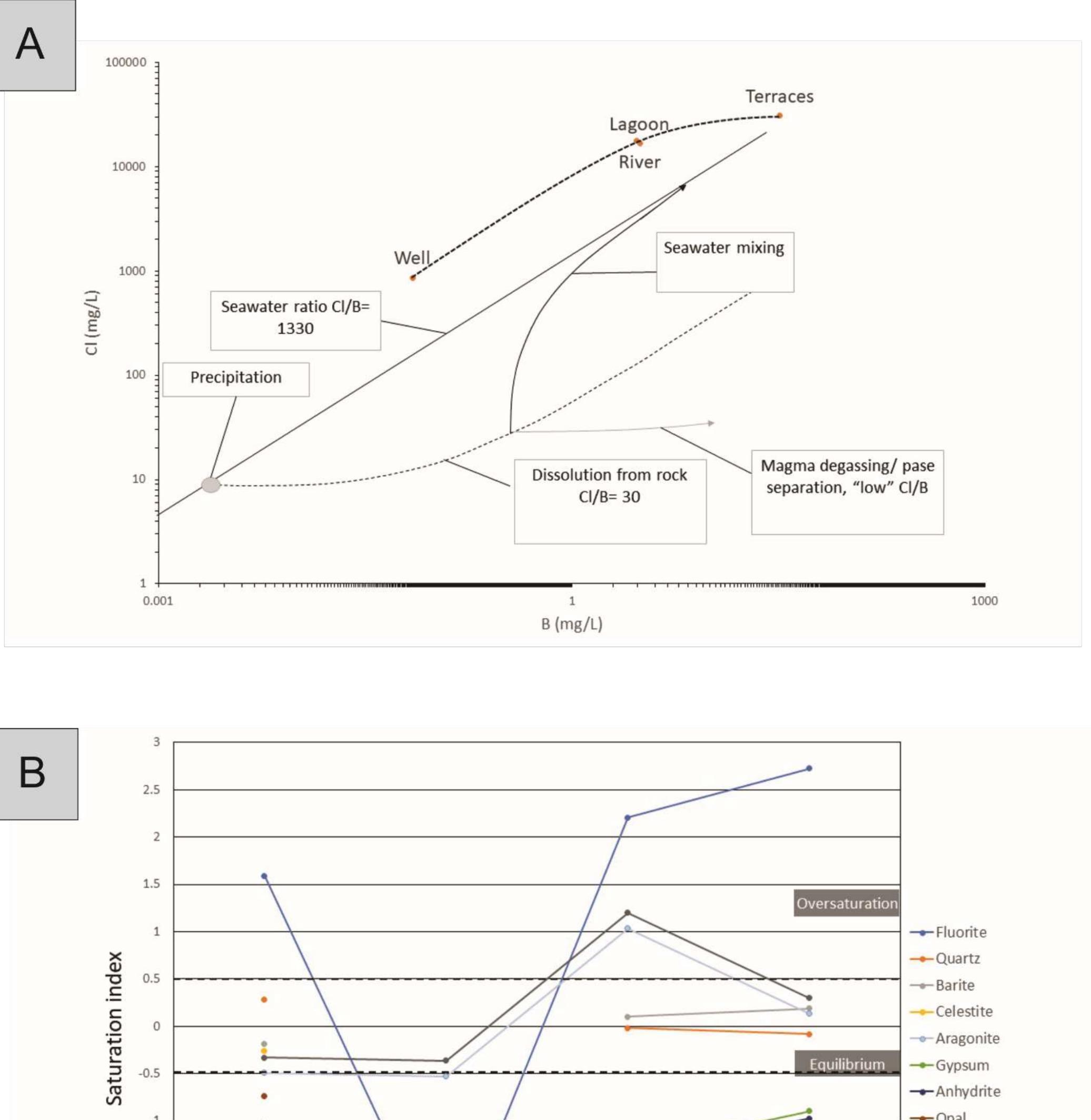
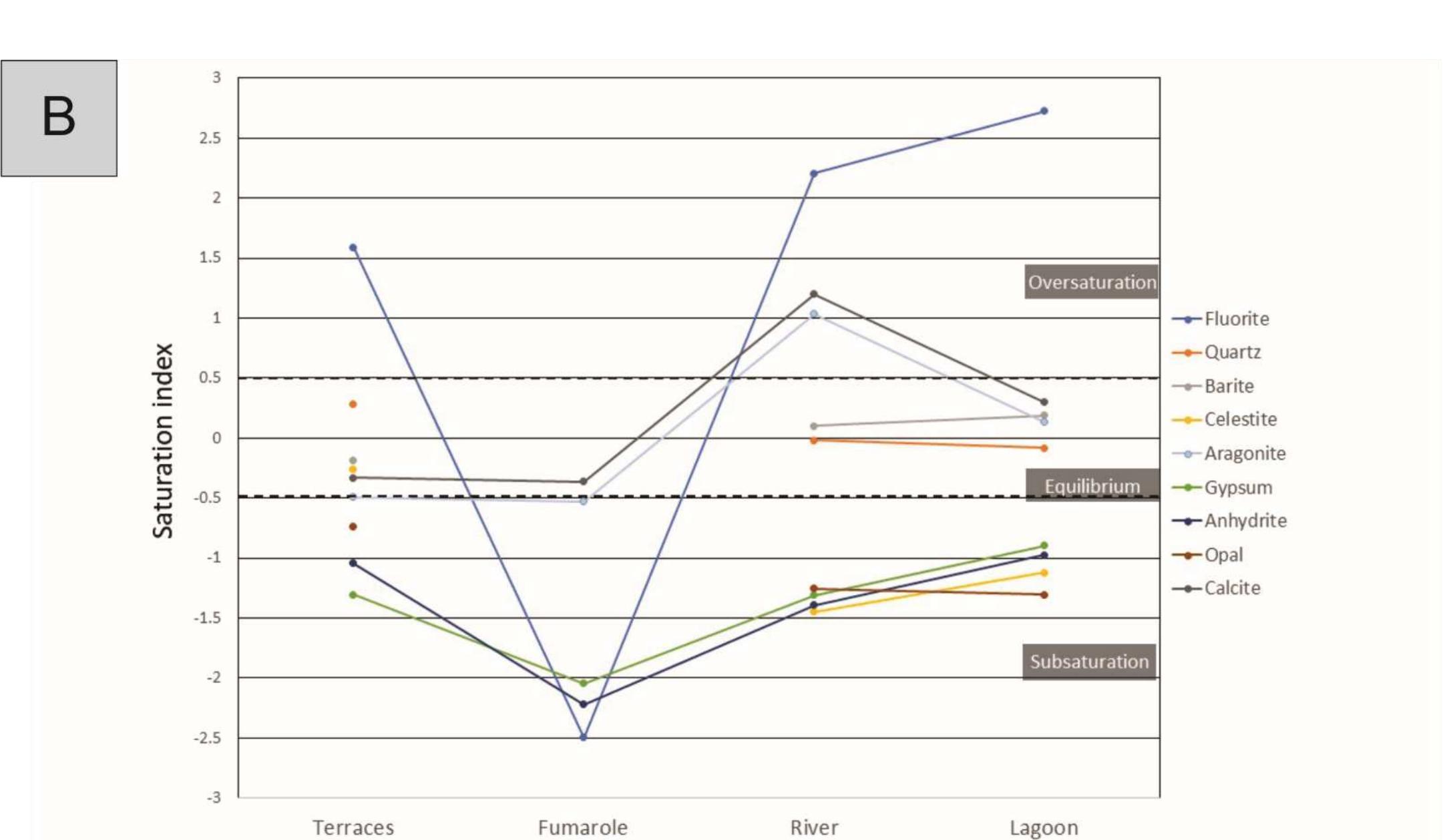
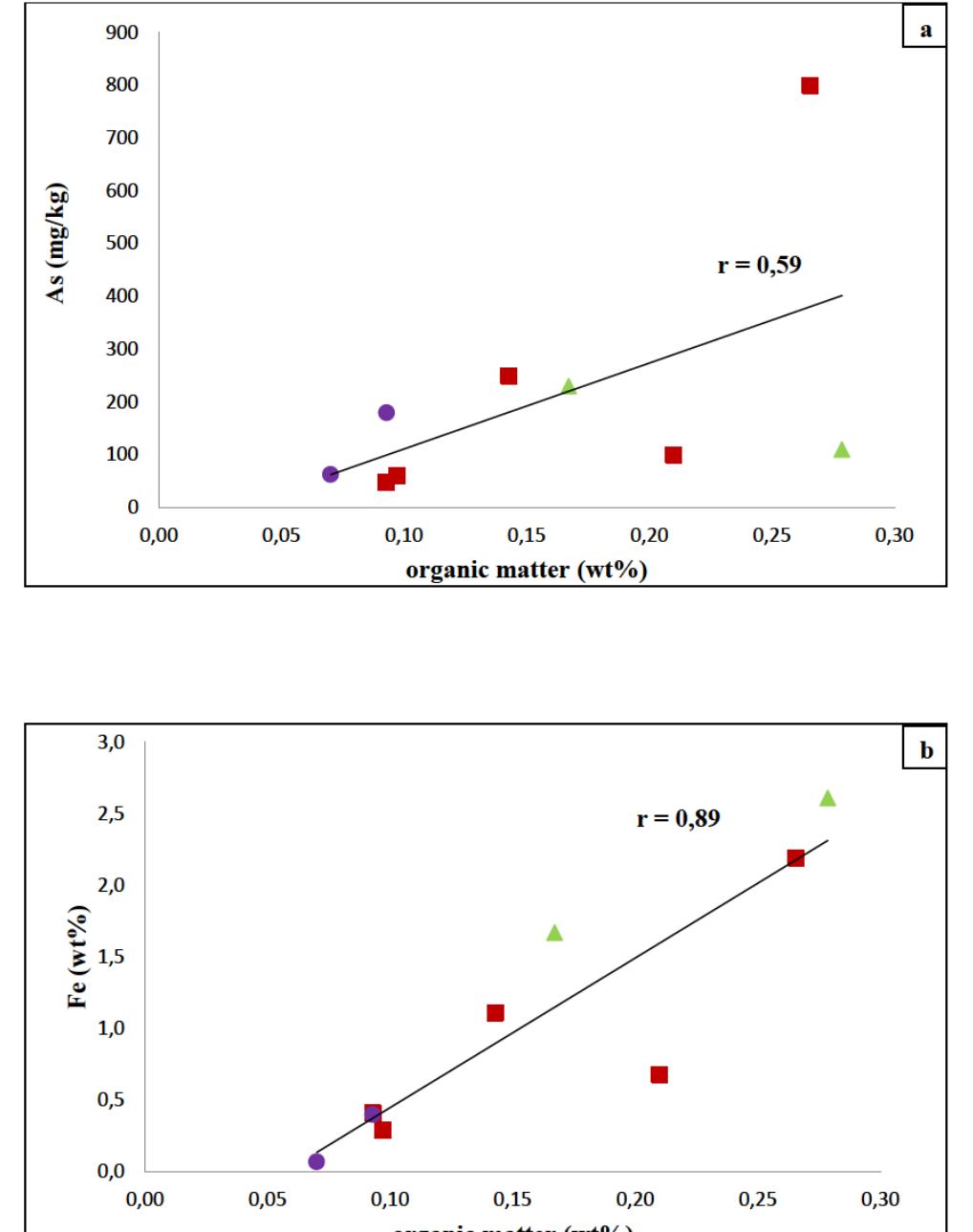
![Figure 8: Plots of Fe (a) and organic matter (b) versus 513 Cvppp. Data from Table 1 and Table 6. It is the contribution of such isotopically light car- bon to authigenic calcite have driven its carbon isotope values lower. This signal of lower 613°C calcite would u recorded in the sediment t cipitation. In the same envi topically light calcite wou creased export of As-bearing particulate Fe-hydroxides to the sediment, resul ingint formation that we consider to ltimately be transferred to and hrough primary carbonate pre- ronment, the deposition of iso- d have been coupled with in- he observed broad relationship between higher abundances of Fe and As, organic carbon and lower 61°C calcite. In we would also inter as linked mainly to distribution of As t ially plausible, but our new results. hrough must also stress, however, ion of a hydrothermal sys position of travertine in the Mesogeia basin and the ele- vated As contents measured therein [33] remains poten- cannot ight of the above arguments, pret much of the observed As anomaly primary processes of As-bearing ferric hydroxide deposition, and o a much lesser extent on re- ater diagenetic fluid-flow. We hat the alternative interpreta- em having influenced the de- be conclusively constrained by](https://figures.academia-assets.com/80365174/figure_009.jpg)


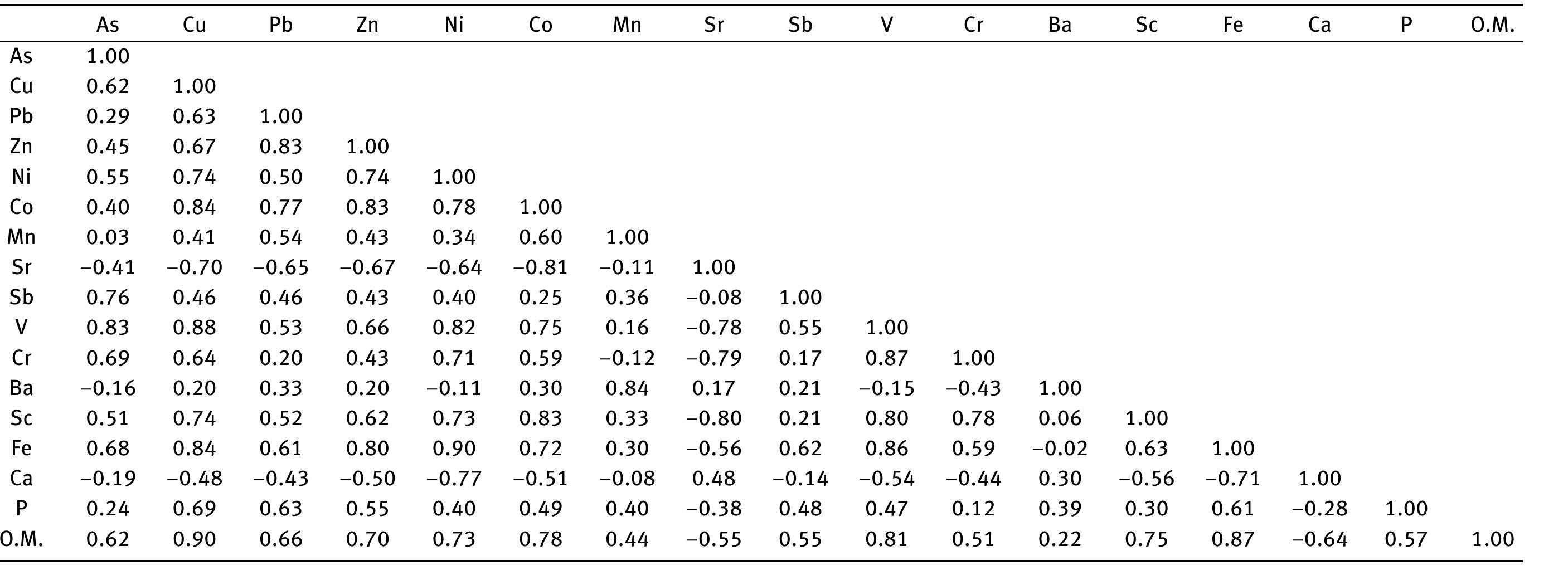
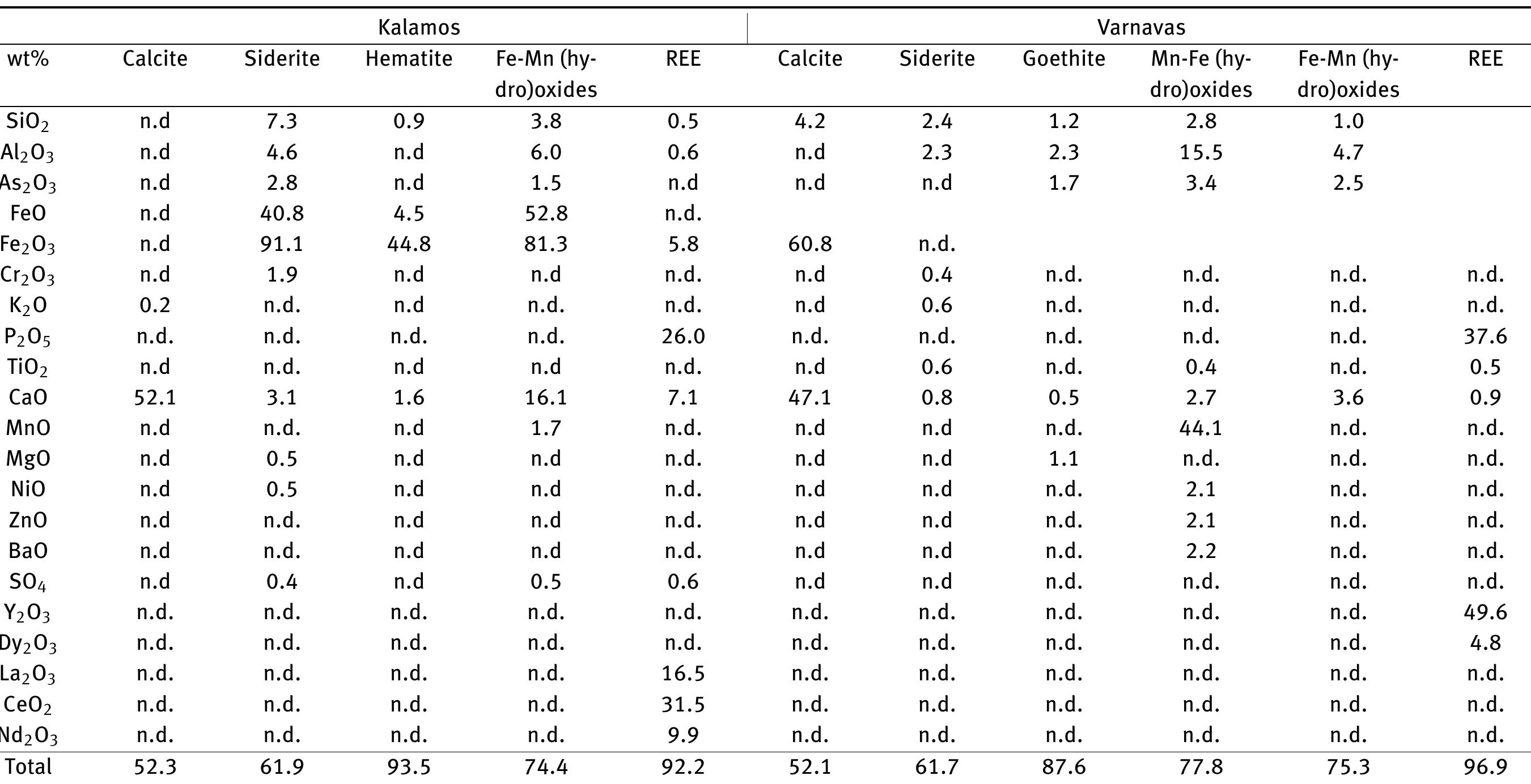
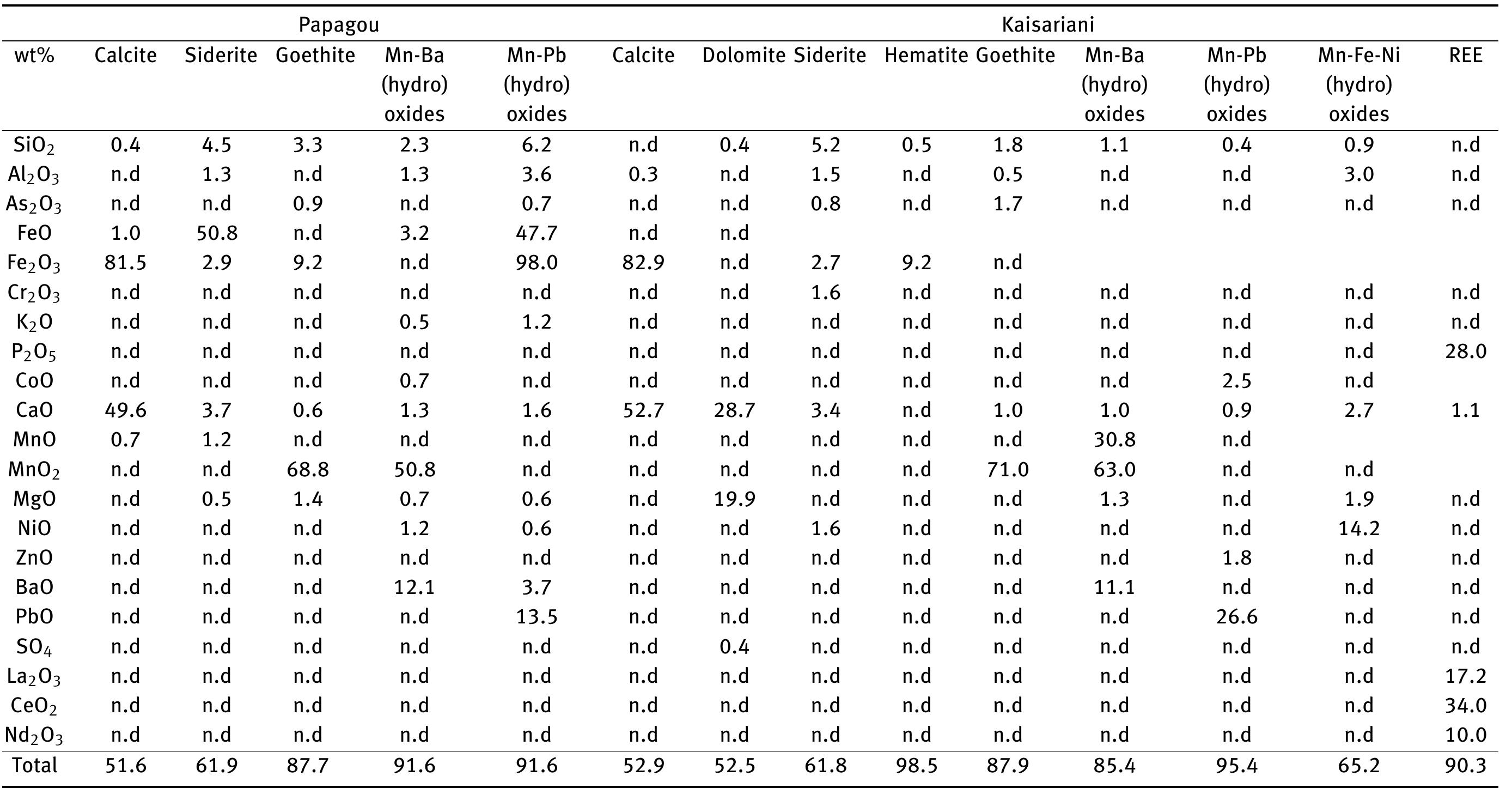
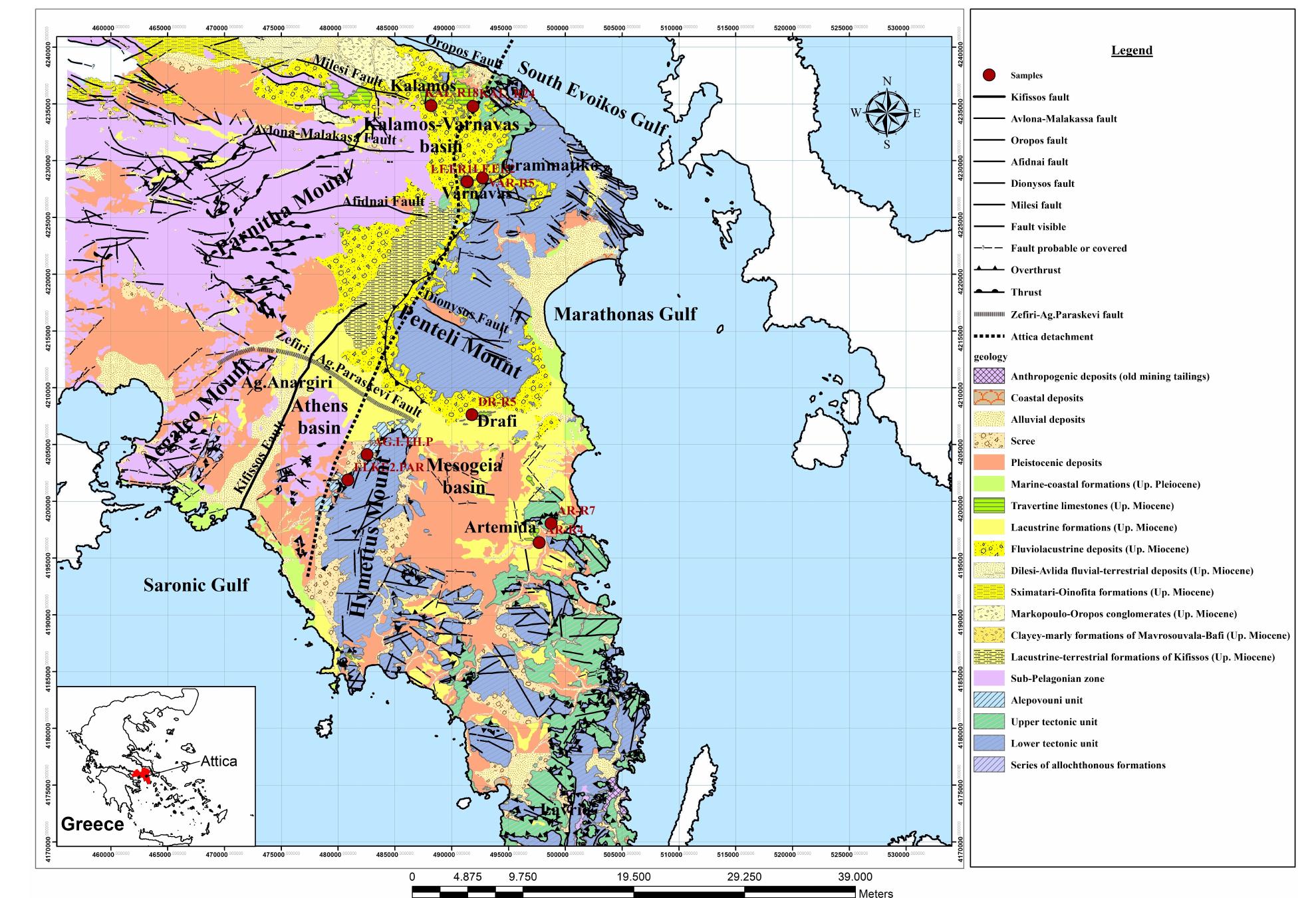
![Figure 7: Plot of 648Oyppg versus 613 Cyppg. Data from Table 1; Koukouvou (2012); Ozkiil et al. (2013). Variations in surface runoff and thus quantitative rection is thought to have extended to of the investigated region (Athens basin) and ly Miocene - omera he southern ransfer of As-enriched Fe/Mn hydroxides into the Neo- zene basins of Attica would have been primarily a func- ion of changing climatic/tectonic conditions t ifetime of these basins in combination with source rock composition at the hinterland. The Ear Pliocene is thought to have marked a period acustrine deposit formation in the study area (marls, marl imestone, travertine or tufa, clays and cong affected by syn-depositional tectonic activity. The Ze Ag. Paraskevi fault (Figure 1) with a WNW-ES hrough the Late of fluvio- es), E strike di- part created a graben in the northern part (Kalamos-Varnavas basin and the larger basin of Assopos). A key consequence of this tec- tonic regime was a topographically elevated area to the south, which functioned as a incursion of marine wa mately responsible ers. T physical barrier preventing his natural “dam” was ulti- for the formation of seasonal lakes fur- ther north in the Kalamos-Varnavas basin [39]. Paleoflora studies have revea climate was humid soil activity was stronger t ative 613C values [26] akin ied carbonates. Suc ed tha h clima the concurrent Late Miocene and warm [34]. During wet periods, herefore resulting in more neg- to those recorded in our stud- ic conditions would have prob- ably also favored the formation of marly limestone with lignite intercalations such as those seen in Almyropota- mos, Pikermi, Mavrosouvala, and Milesi [62, 63]. The lakes therefore seem to have formed in dynamic response to an integrated environmental, climatic and tectonic realm, and their sedimentary archive thus provides a continuous record of local and regional change [64].](https://figures.academia-assets.com/80365174/figure_008.jpg)


