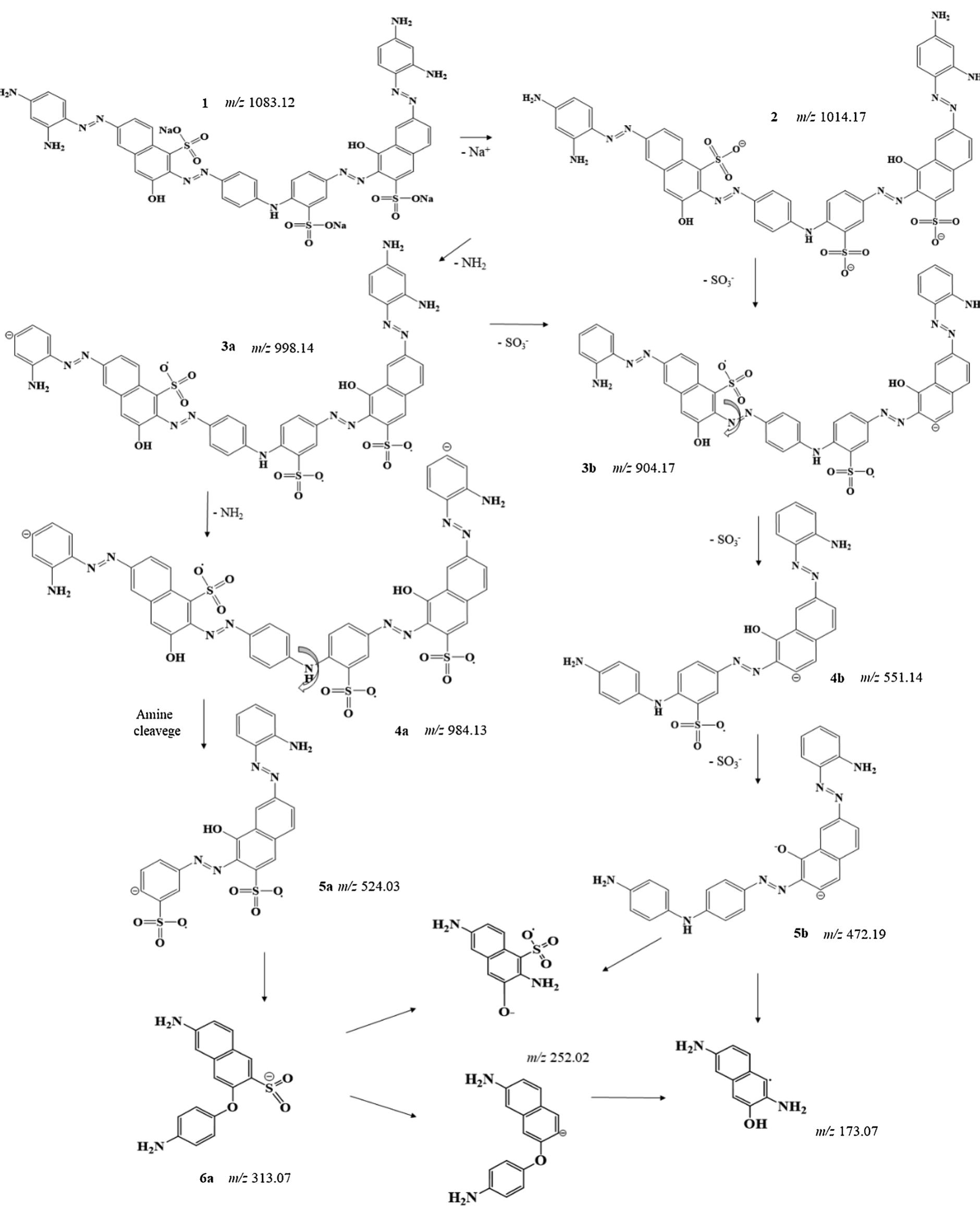
The increasing presence of organic pollutants in wastewater has become a major concern, primarily due to rapid population growth and ongoing technological advancements that deplete water resources. Consequently, water bodies are becoming... more
The textile industry produces large amounts of wastewater during all phases of textile production and finishing. Major pollutants in textile wastewaters are high suspended solids, chemical oxygen demand, heat, color, acidity, and other... more
Azo dyes account for about 60-70% of all dyes in textile, food, pharmaceutical, leather, cosmetics and paper industries and are the most common synthetic colorants released into the environment, causing undesirable colouring of surface... more
The effective treatment of dye-laden industrial effluents remains a critical challenge in environmental engineering. This study introduces a novel hybrid system integrating ultrasound (US) and nanofiltration (NF) technologies for the... more
This article describes the potential of KMnO 4 for oxidation of the thiazine dye (Toluidine blue TB) in an acidic medium. The effect of different operational parameters like initial concentration of dye, potassium permanganate, pH, ionic... more
BACKGROUND: Nitrogen doped titanium dioxide (N-TiO 2), iron doped titanium dioxide (Fe-TiO 2), and undoped titanium dioxide (TiO 2) nanocatalysts were synthesized using sol-gel method aided with ultrasound (US). The effects of duty cycle,... more
Microbial fuel cells (MFCs) represent a transformative technology at the intersection of wastewater treatment and renewable energy, offering a sustainable pathway to achieve Sustainable Development Goals 6 (clean water and sanitation) and... more
Dyes are those synthetic and natural compounds which applied in form of aqueous based solutions with many additives to strong attachment of dye molecule with a fabric for persistence the colour on it while pigments are usually insoluble.... more
Lawrence K. Wang, Mu-Hao Sung Wang, Yung-Tse Hung (2021). Integrated Natural Resources Research, Springer Cham, NY, USA; 651 pages, ISBN: 978-3-030-61001-2; https://doi.org/10.1007/978-3-030-61002-9 .... ABSTRACT: The book will... more
The aqueous degradation of Reactive Yellow 84 (RY84) by potassium peroxydisulfate (K 2 S 2 O 8) has been studied in laboratory scale experiments. The effect of the initial concentrations of potassium peroxydisulfate and RY84, pH and... more
A nanocrystalline anatase hydrate (Titanhydrat-0, TH) is a highly efficient catalyst for low intensity sonochemical degradation of 4-chlorophenol (4-CP). The initial disappearance rate of 4-CP increases by a factor of 95 under the TH... more
Neutral Red was subjected to two different oxidative processes, namely the photo-oxidative process and the Fenton process. The dye was found to undergo substantial and rapid decoloration by both the methods. Conditions were optimized in... more
The degradation of Reactive Black 5 (KN-B) in water using double-dielectric barrier discharge (DDBD) was studied. Experimental results showed that the KN-B degradation rate increased as the initial pH decreased. Low concentrations of Fe2+... more
Highlights: •O3-based AOPs constitutes a good process to treat textile wastewater. •The wastewater matrix caused lack of summary effect of O3, UV and H2O2 within AOPs. •Ozone treatment was not influenced by textile auxiliaries. •Textile... more
Wastewater-based microbial fuel cell is a promising green technology that can potentially be used to treat recalcitrant wastewater such as textile wastewater through in situ Fenton oxidation while generating net positive energy. One of... more
In the present study, a comparison of central composite design (CCD) and Taguchi method was established for Fenton oxidation.Dyeini, Dye : Fe+2, H2O2 : Fe+2, and pH were identified control variables while COD and decolorization efficiency... more
Application of the Fenton process for textile wastewater treatment is limited due to high treatment cost, substantially contributed by the un‐availability of cheap hydrogen peroxide. Therefore, alternative methods for hydrogen peroxide... more
Wastewater-based microbial fuel cell is a promising green technology that can potentially be used to treat recalcitrant wastewater such as textile wastewater through
The main objective of this work was to study the degradation of Acid Orange 7 (AO7) dye in aqueous solution by hydrogen peroxide using HFe 2.5 P 2 W 18 O 62 23.H 2 O as a catalyst. HFe 2.5 P 2 W 18 O 62 23.H 2 O is a recyclable... more
The effectiveness of peroxymonocarbonate ( HCO 4 − ) on the degradation of Reactive Blue 19 (RB19) textile dye was investigated in this study. The formation kinetics of HCO 4 − produced in situ in a H 2 O 2 − HCO 3 − system was studied to... more
Your article is protected by copyright and all rights are held exclusively by Springer Science +Business Media Dordrecht. This e-offprint is for personal use only and shall not be selfarchived in electronic repositories. If you wish to... more
Abstract: The photocatalytic degradation of lissamine fast yellow has been carried out in the aqueous suspension of ZnO under artificial light. The effects of process parameters such as catalyst loading, H 2 O 2, FeCl 3, etc., on the... more
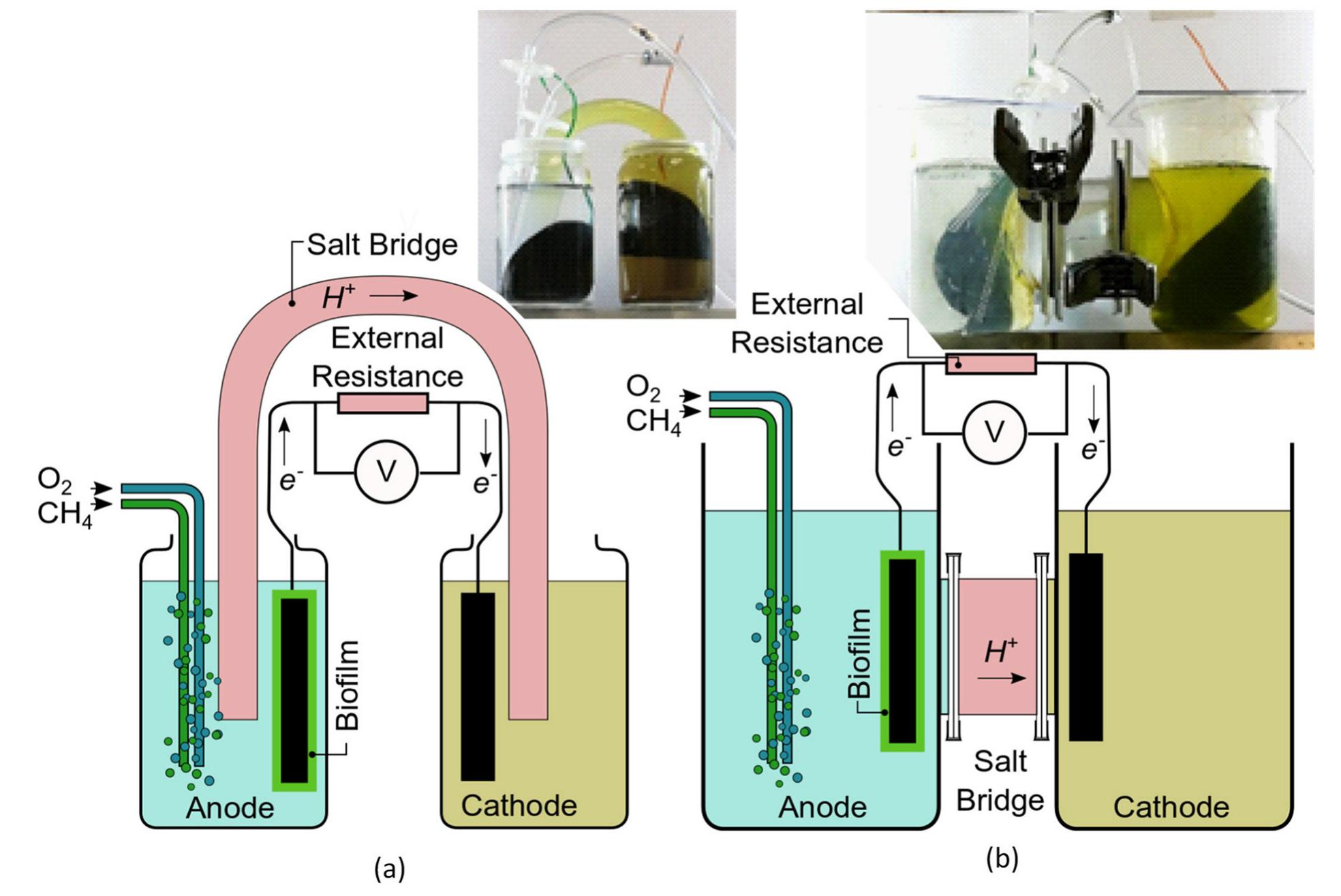
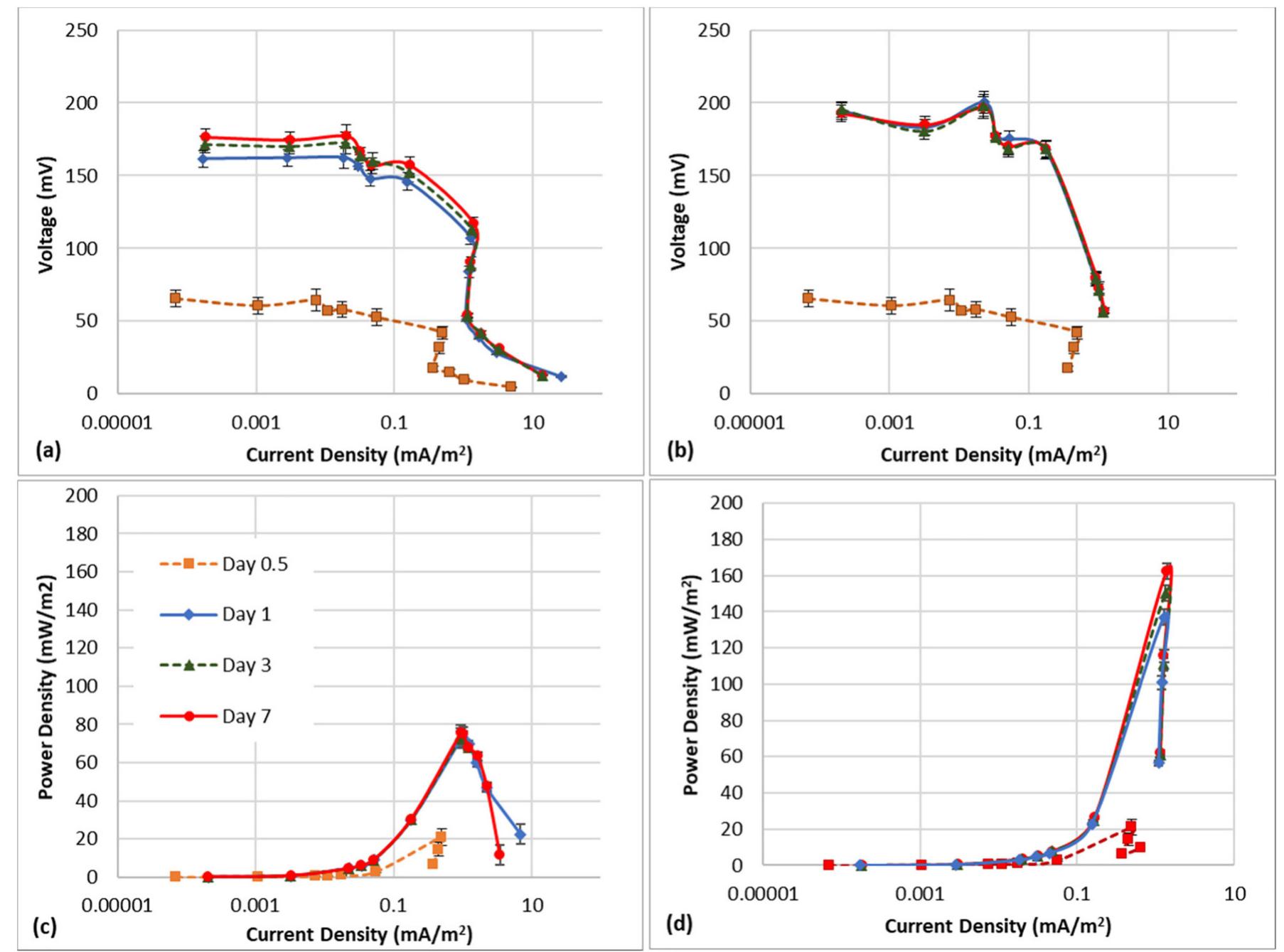
Microbial fuel cells (MFCs) are typically designed as a two-chamber system with the bacteria in the anode chamber separated from the cathode chamber by a polymeric proton exchange membrane (PEM). Most MFCs use aqueous cathodes where water... more
Advanced oxidation processes are eco-friendly methods of destroying organic pollutants by using semiconductor and visible light. Photocatalytic degradation of methylene violet has been investigated in aqueous heterogeneous suspension. The... more
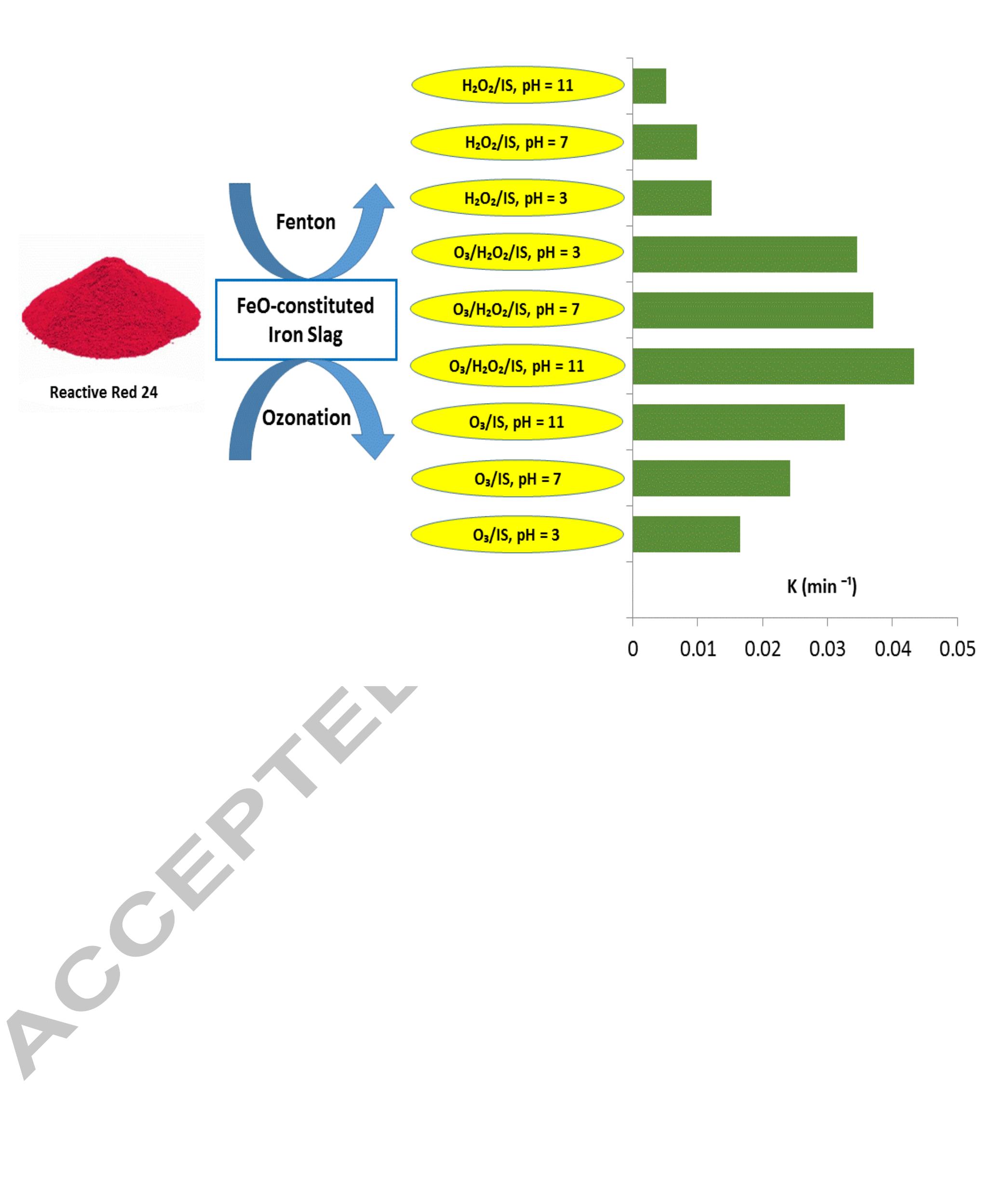
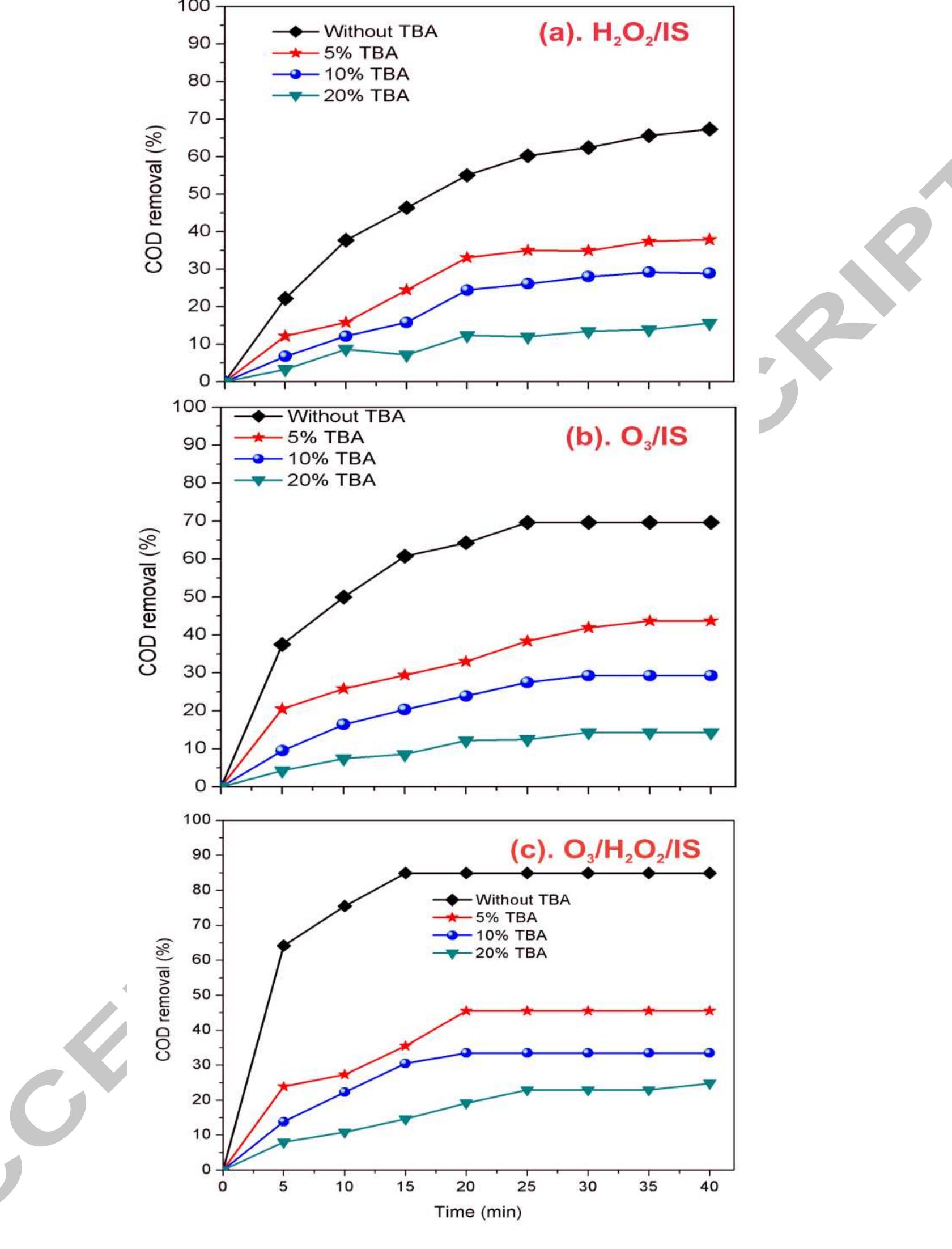
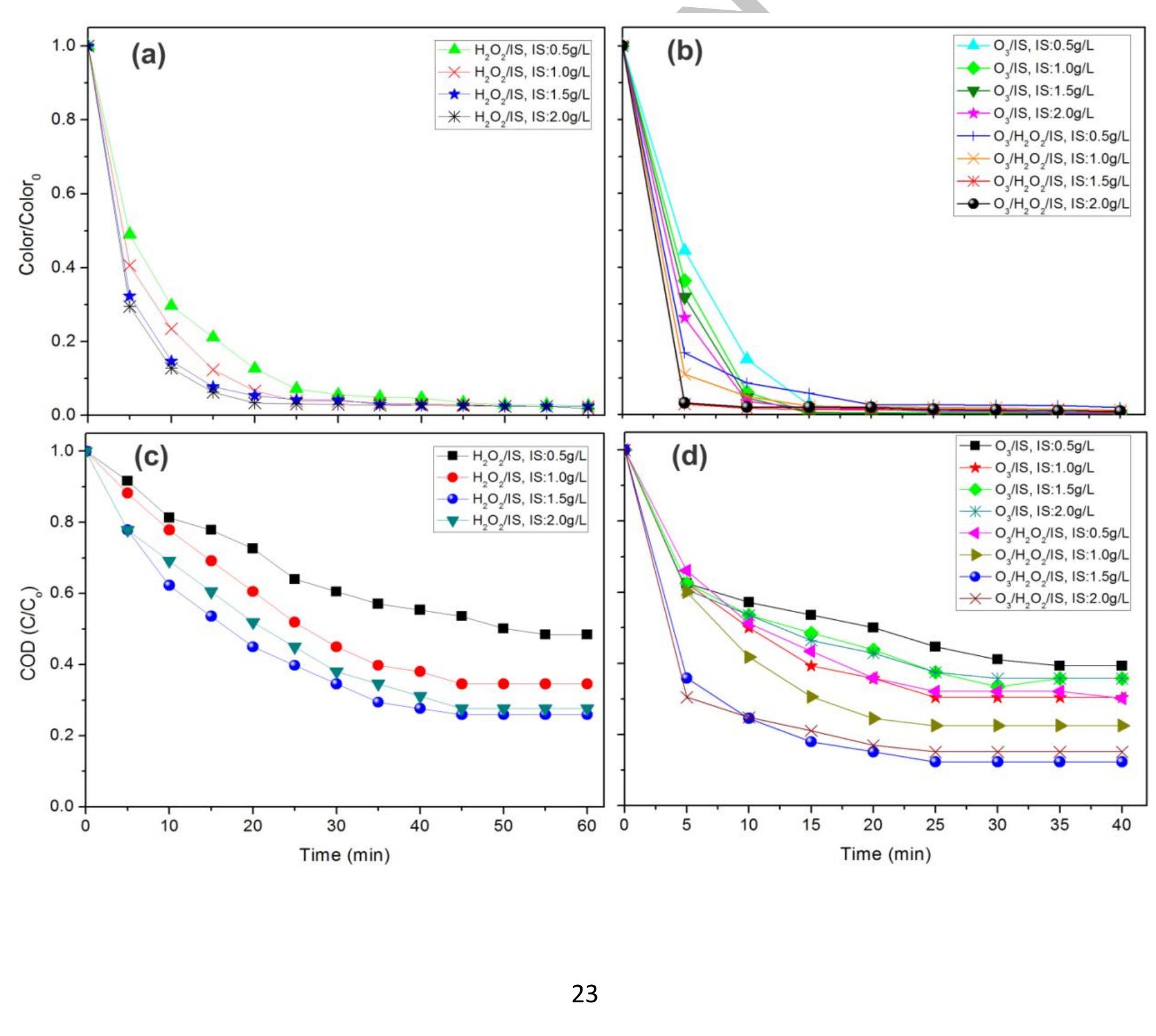
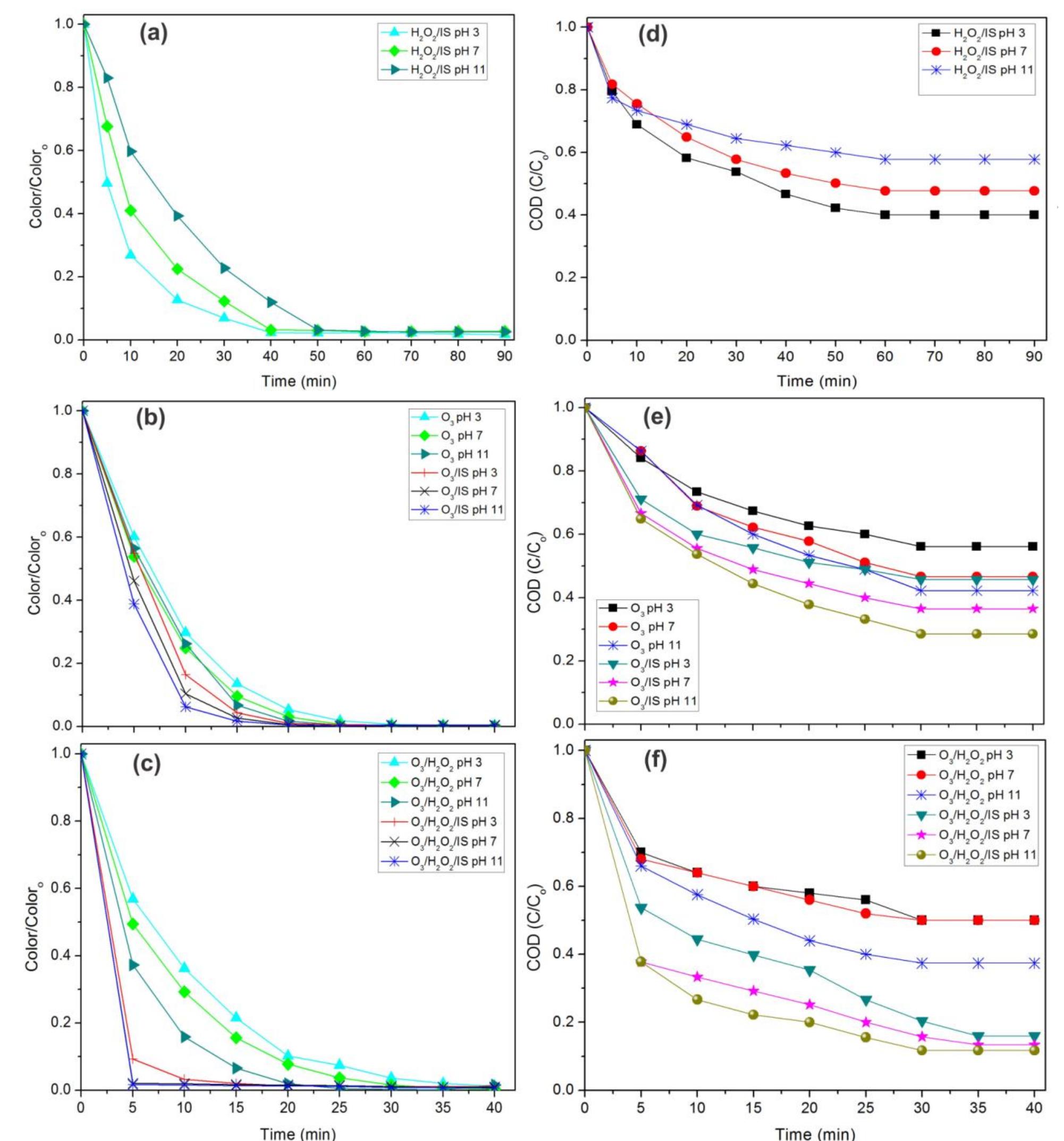
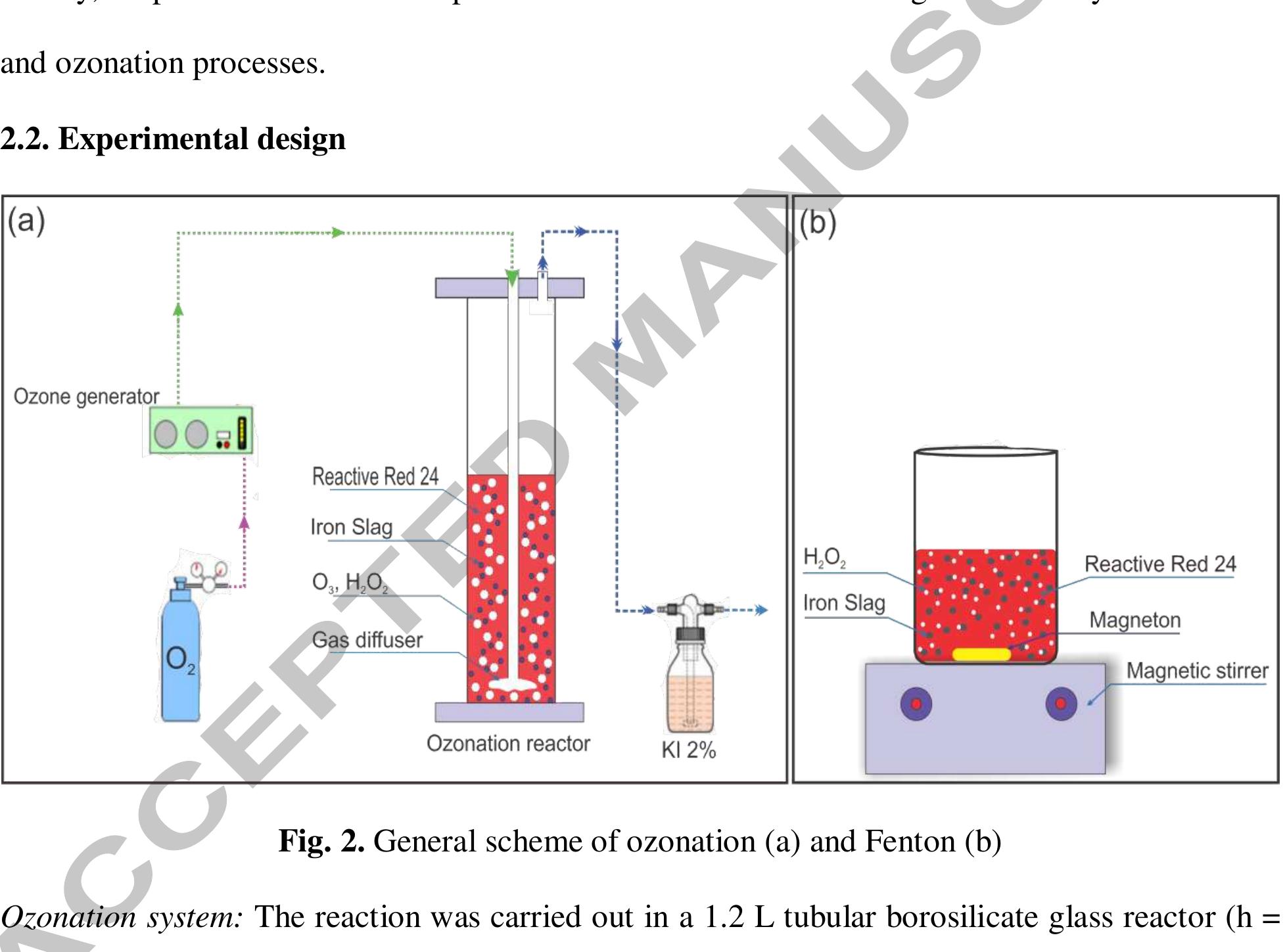
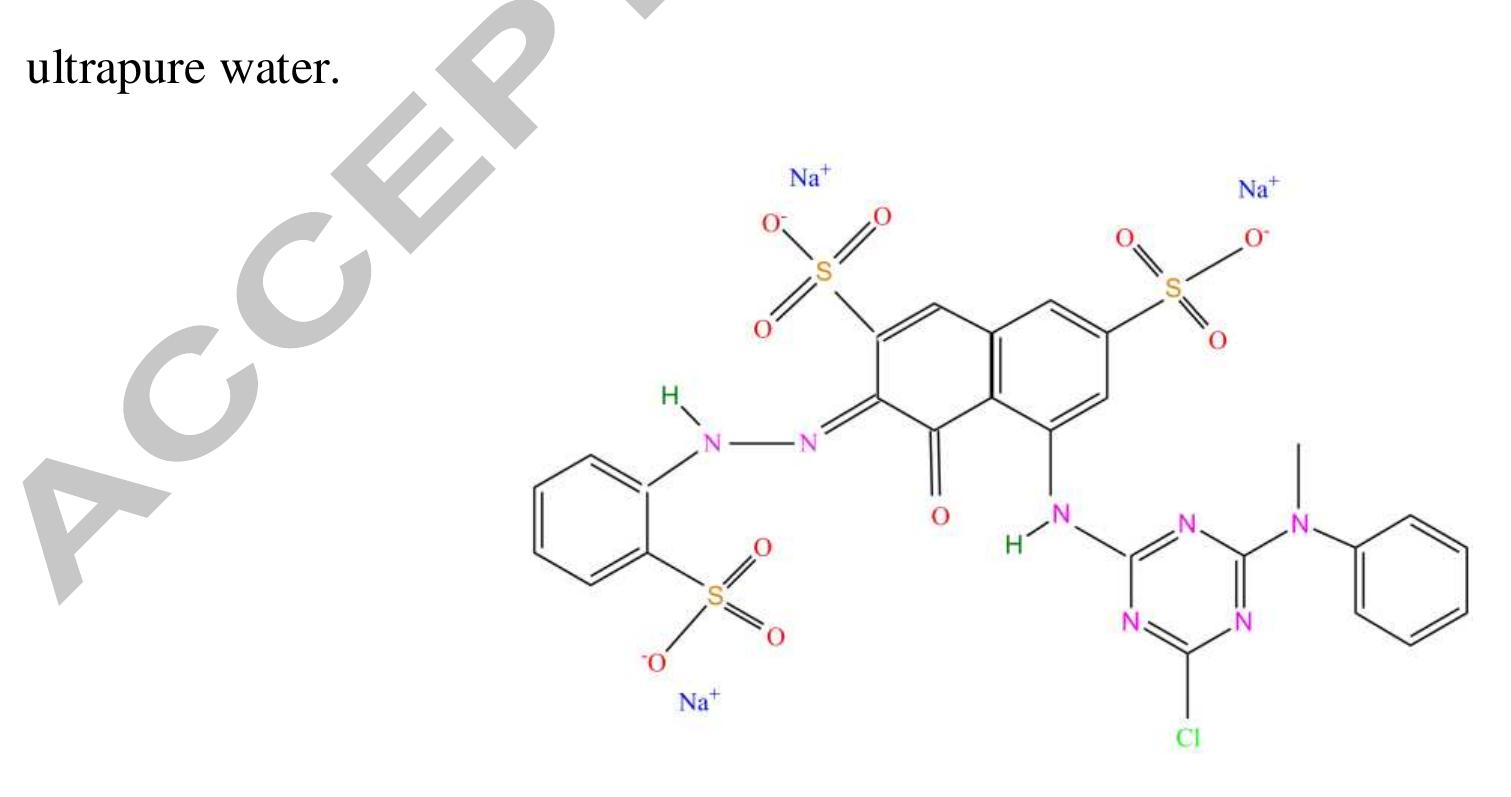
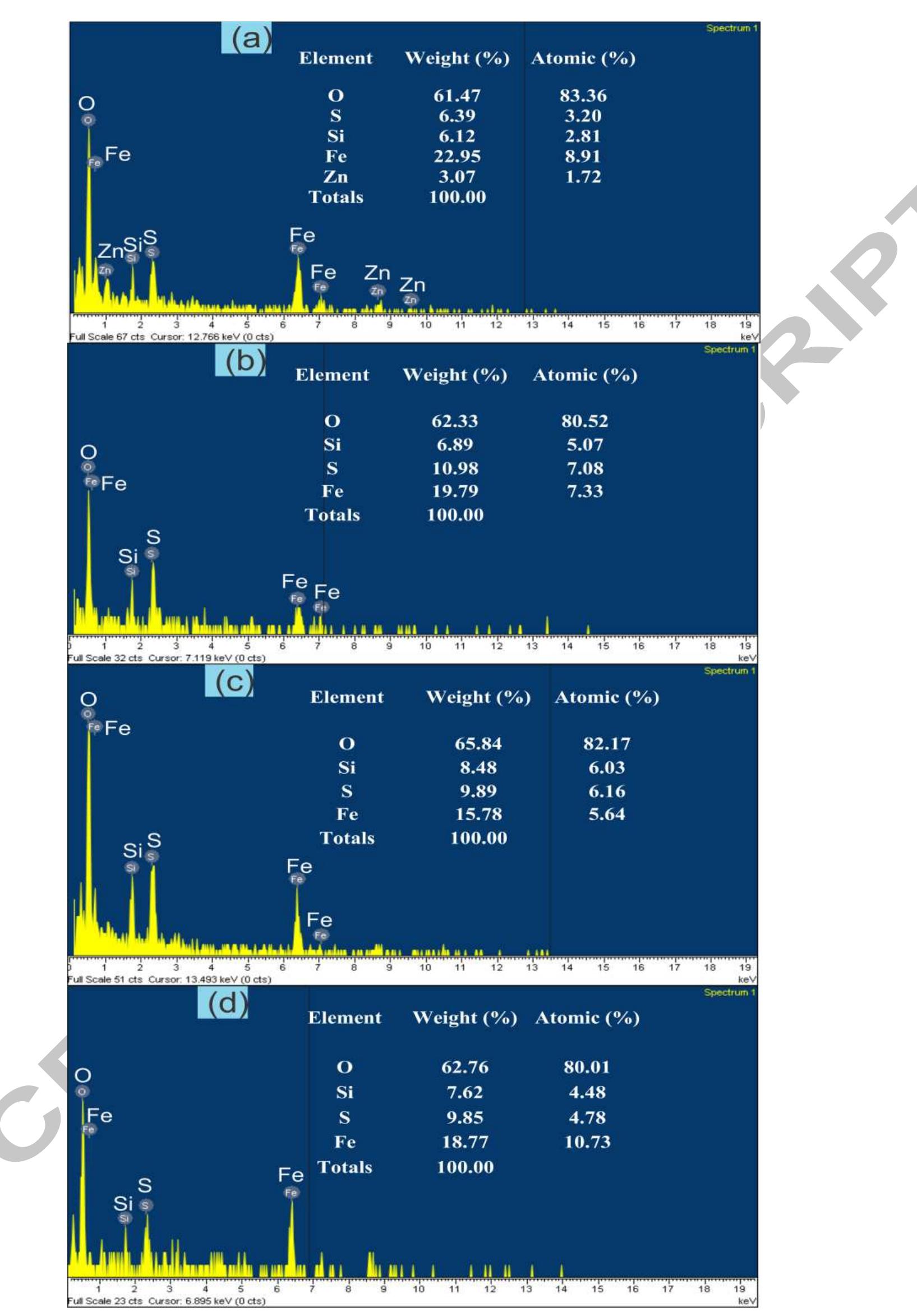
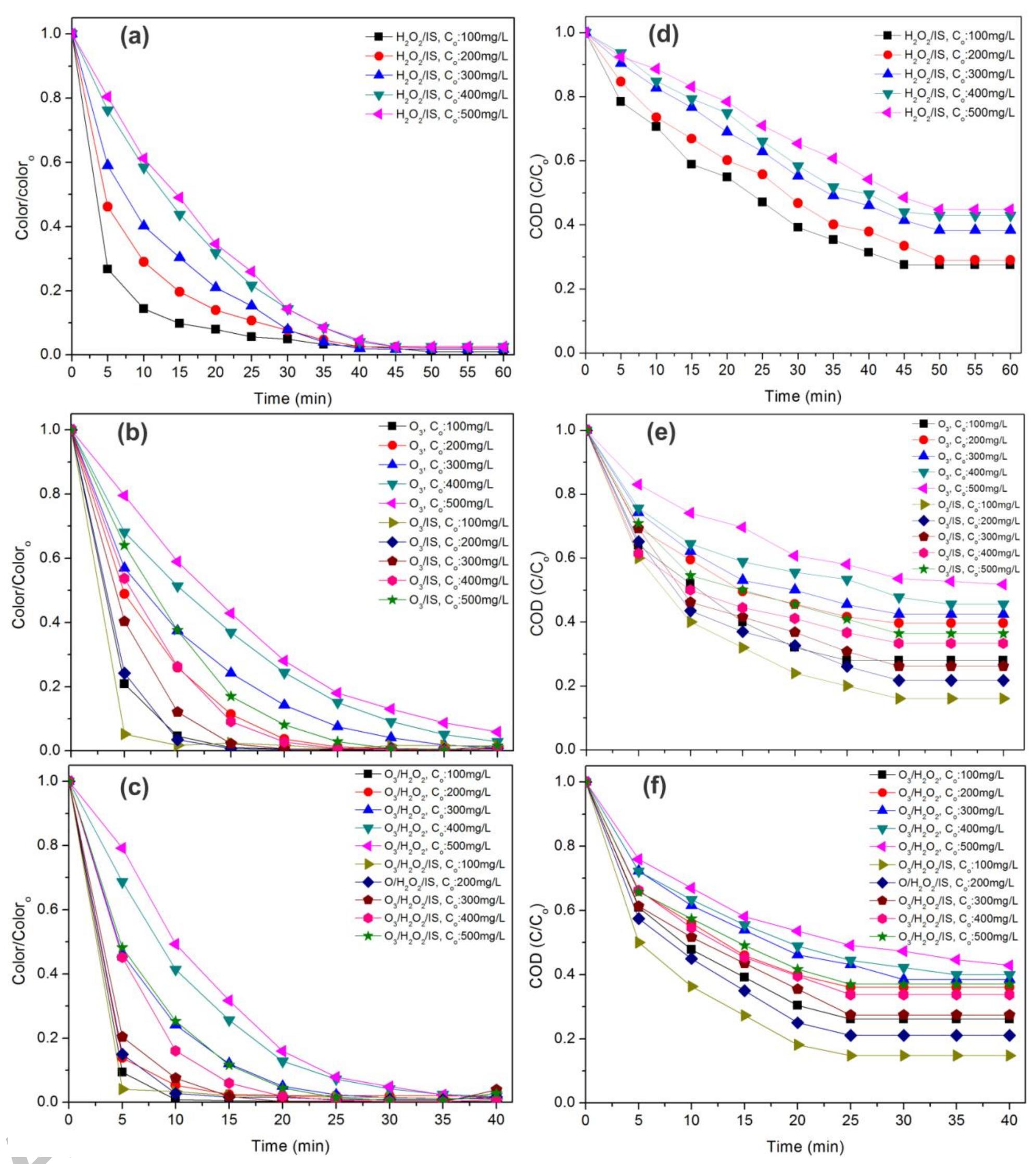
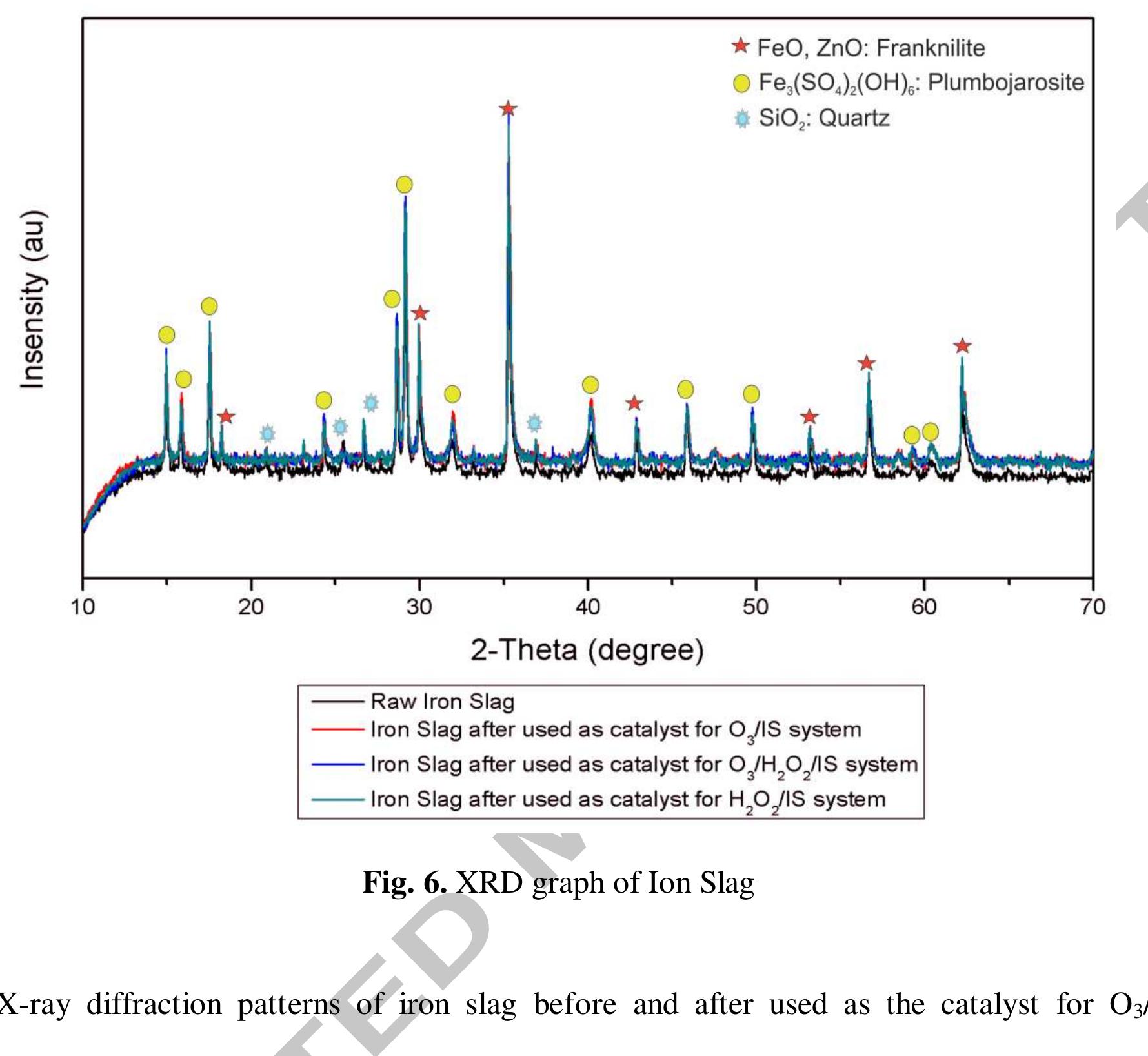
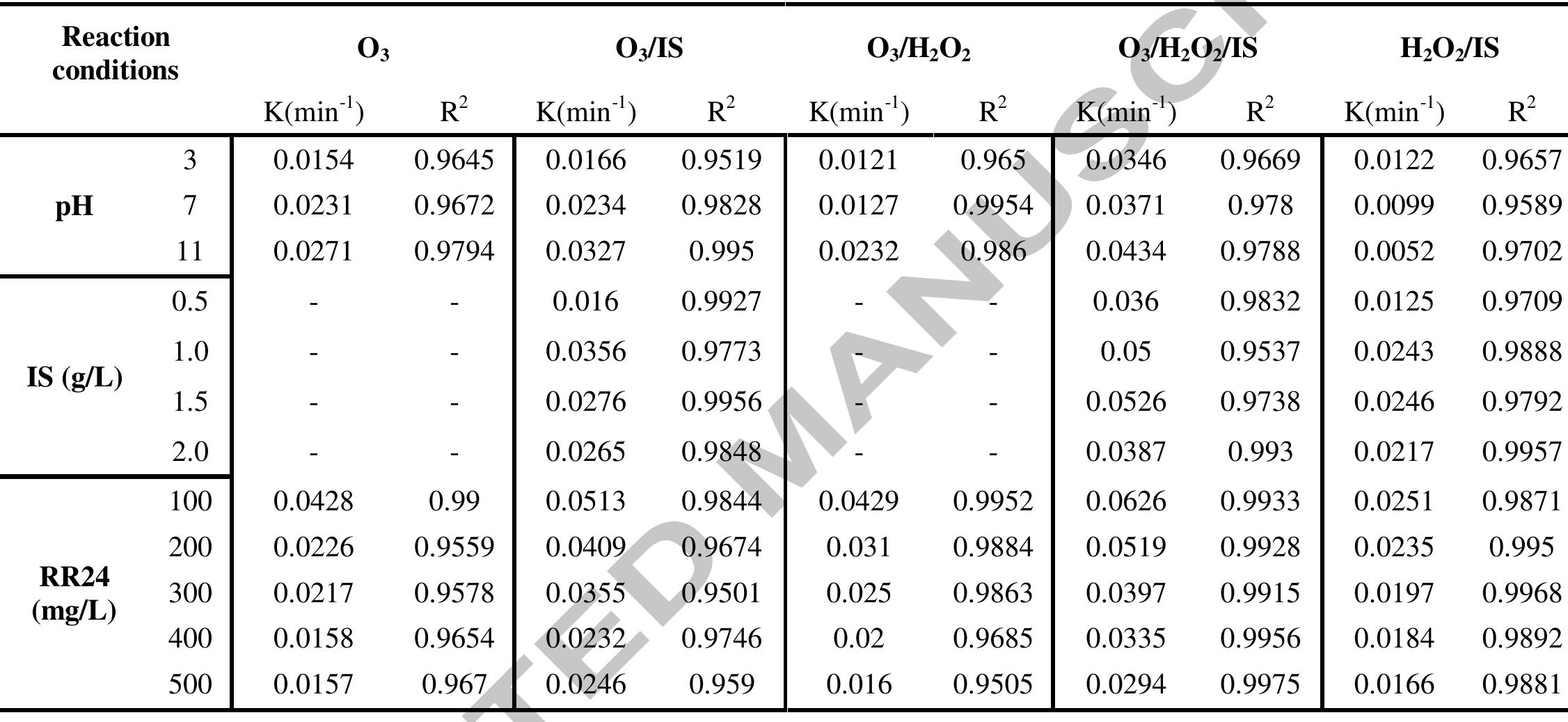
Using FeO-constituted Iron Slag wastes as heterogeneous catalyst for Fenton and Ozonation processes to degrade Reactive Red 24 from aqueous solution, Separation and Purification Technology (2019), doi:
The production of an efficient, reusable, stable and easily separable catalyst in the ozonation process through a reliable procedure is one of the essential requirements of the catalytic ozonation process (COP). In this study, the... more
Photocatalytic degradation of pollutants under solar light irradiation is an economically viable process and a very promising clean wastewater treatment technology. The aim of this study is to evaluate photocatalytic degradation of... more
In this investigation, nano ZnO was sonochemically synthesized by a novel method using a methionine precursor. A narrow size distribution (41-50 nm) of nano ZnO was achieved that was immobilized on perlite and applied as a catalyst in... more
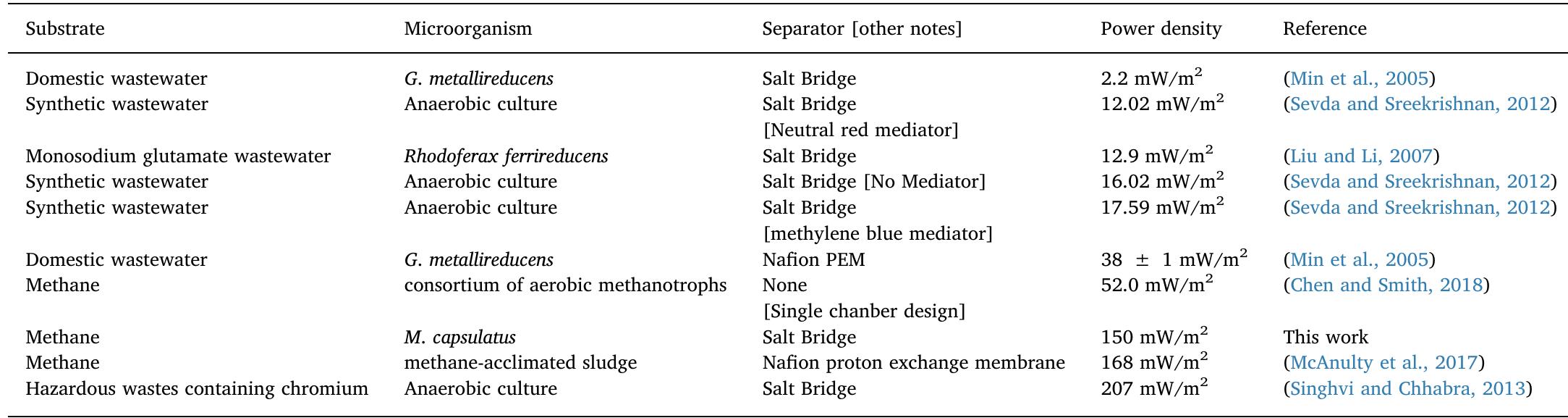
Methane is a widely available energy resource that can be obtained biologically as well as via natural gas. However, using methane as a fuel in low-temperature fuel cells has been a challenge due to the high thermodynamic stability of... more
In this investigation, nano ZnO was sonochemically synthesized by a novel method using a methionine precursor. A narrow size distribution (41-50 nm) of nano ZnO was achieved that was immobilized on perlite and applied as a catalyst in... more
In this investigation, nano ZnO was sonochemically synthesized by a novel method using a methionine precursor. A narrow size distribution (41-50 nm) of nano ZnO was achieved that was immobilized on perlite and applied as a catalyst in... more
Wastewater from the textile industry containing a high concentration of organic and inorganic chemicals has strong color and residual chemical oxygen demand (COD). Therefore, advanced oxidation processes (AOPs) are very good candidates to... more
In this study, the photo‐Fenton process was used to identify degradation conditions of Reactive Blue 19 (RB19) and Reactive Red 21 (RR21). The effects of pH, initial H2O2 and FeSO4 concentrations, time, and UV light intensity in... more
The photocatalytic performance of copper ferrite graphene oxide catalyst, CFXGO (CF: Copper ferrite, CuFe 2 O 4 ; GO: Graphene oxide; X: GO weight percentage) was tested for photo Fenton-like oxidation of Reactive Black 5 by using UV... more
Reactive Black 5 was effectively degraded by the CWAO using LaNiO 3 catalyst. The most suitable reaction conditions were 1 g/L of LaNiO 3 , 50 C, 1 atm, and pH ¼ 3. 65.4% degradation and 89.6% decolorization efficiencies were achieved.... more
Several processes have been used for dye and chemical oxygen demand (COD) removal from wastewater. Comparision of these methods are important in terms of efficiency and energy consumption. In recent years, advanced oxidation processes... more
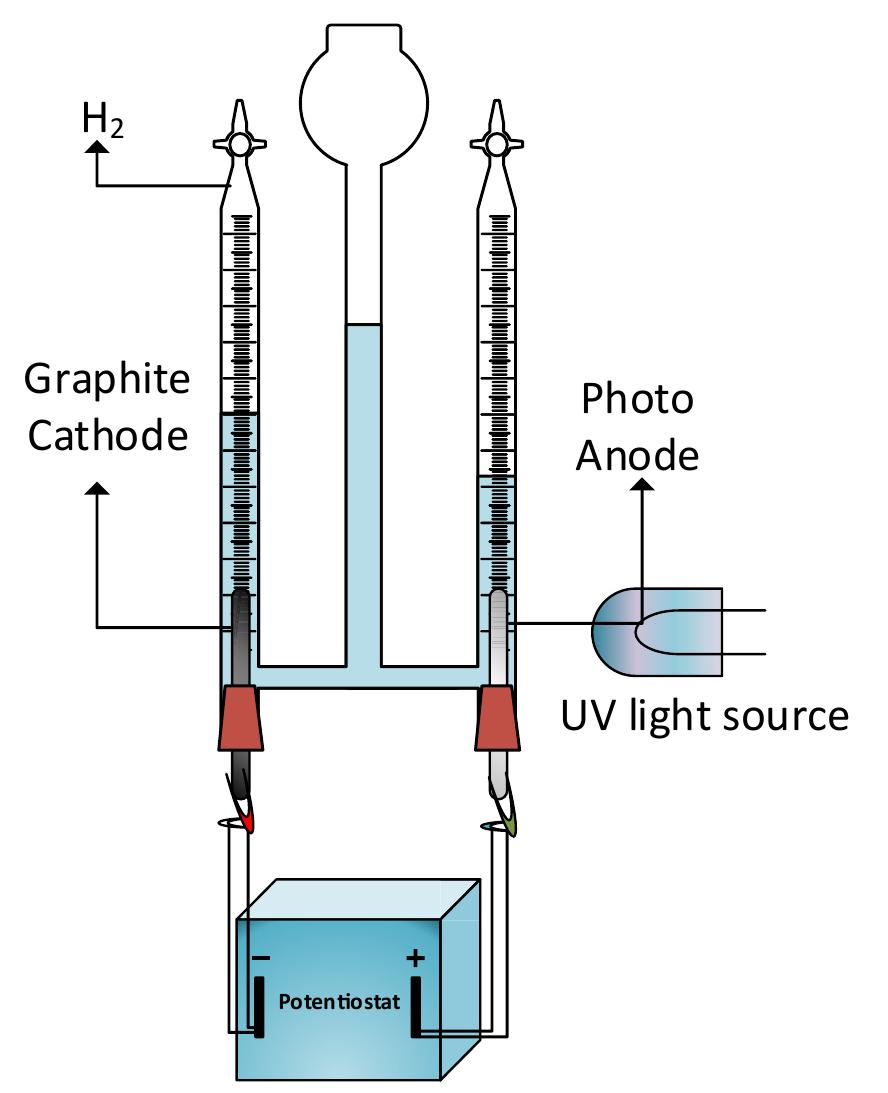
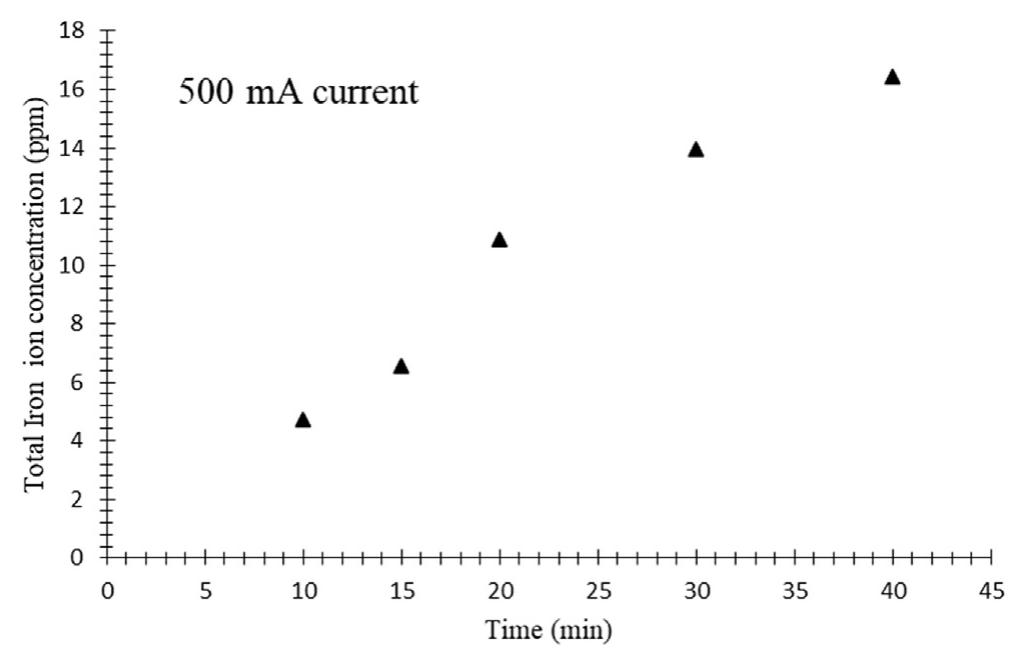
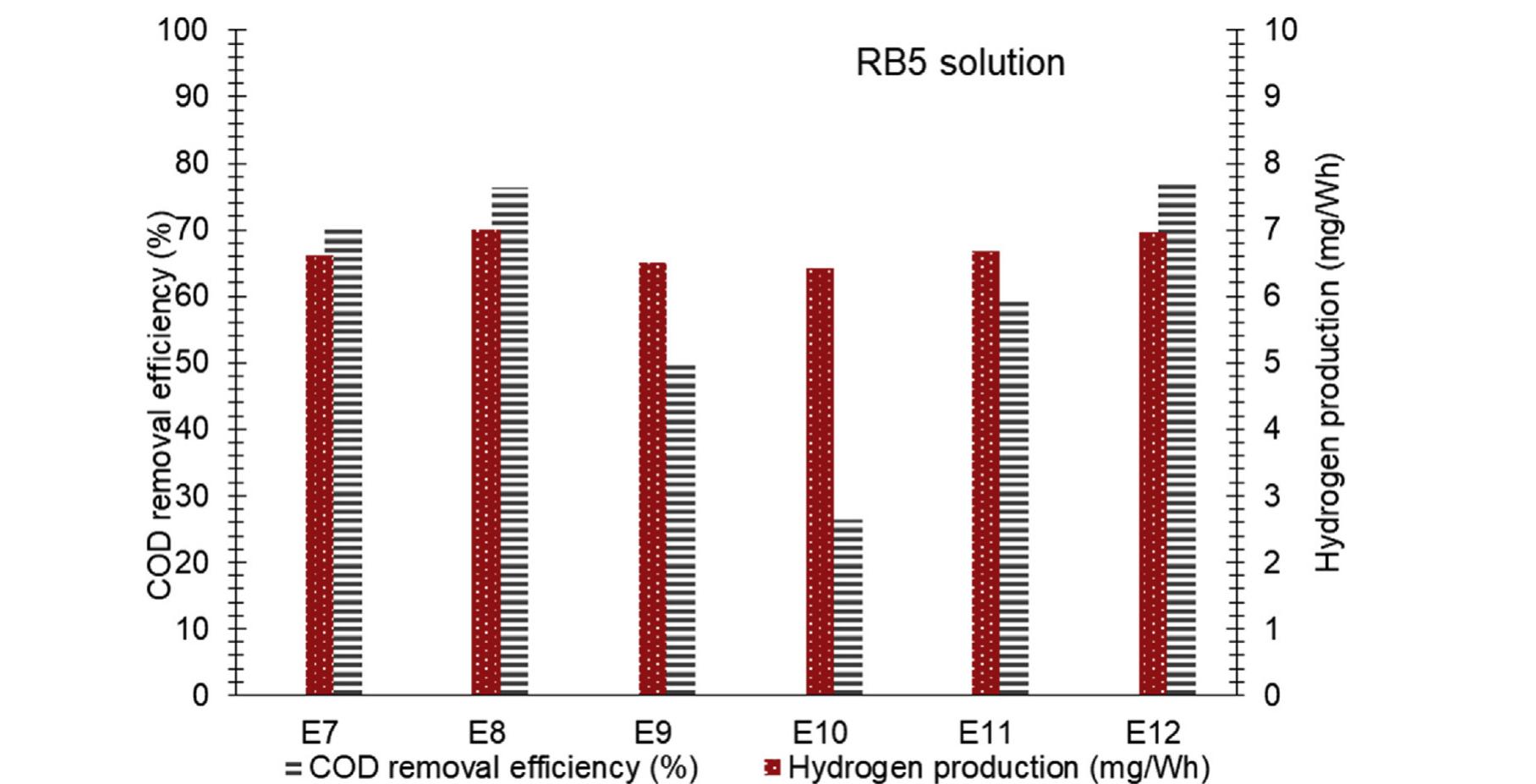
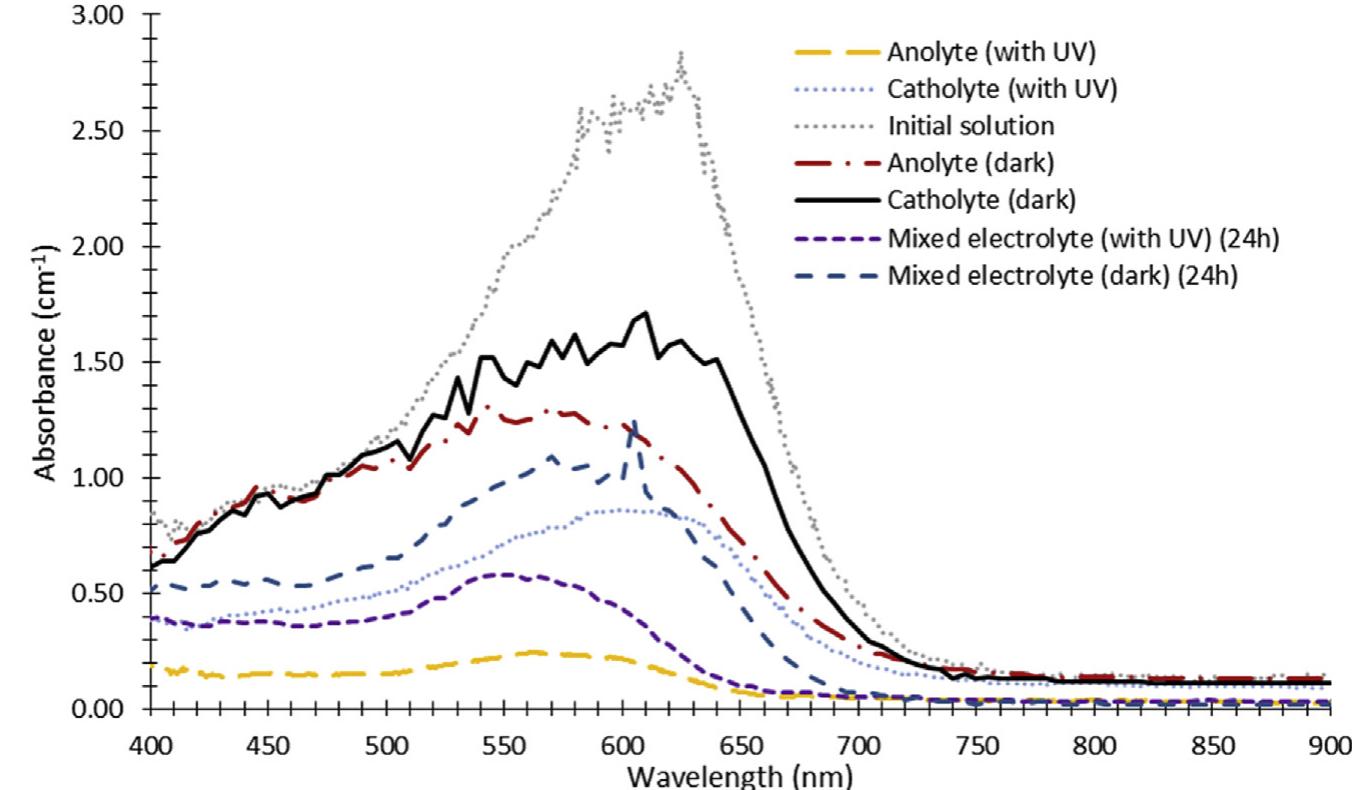
h i g h l i g h t s Investigation of synergistic effects of AOPs for H 2 production and wastewater treatment processes. TiO 2 NPs utilization in combination with Fenton reactions for wastewater treatment. Wastewater treatment systems with... more
Nowadays, leaving industrial wastewater into receiving environment causes serious environmental problems. In this study, experimental studies on color and chemical oxygen demand (COD) removal of paper industrial wastewater were carried... more
The objective of this study was to investigate mineralization of hazardous water soluble azo dye Acid Orange 7 (AO7) under photocatalytic process. In this experiment, sunlight was used as the source for UV irradiation and titanium... more
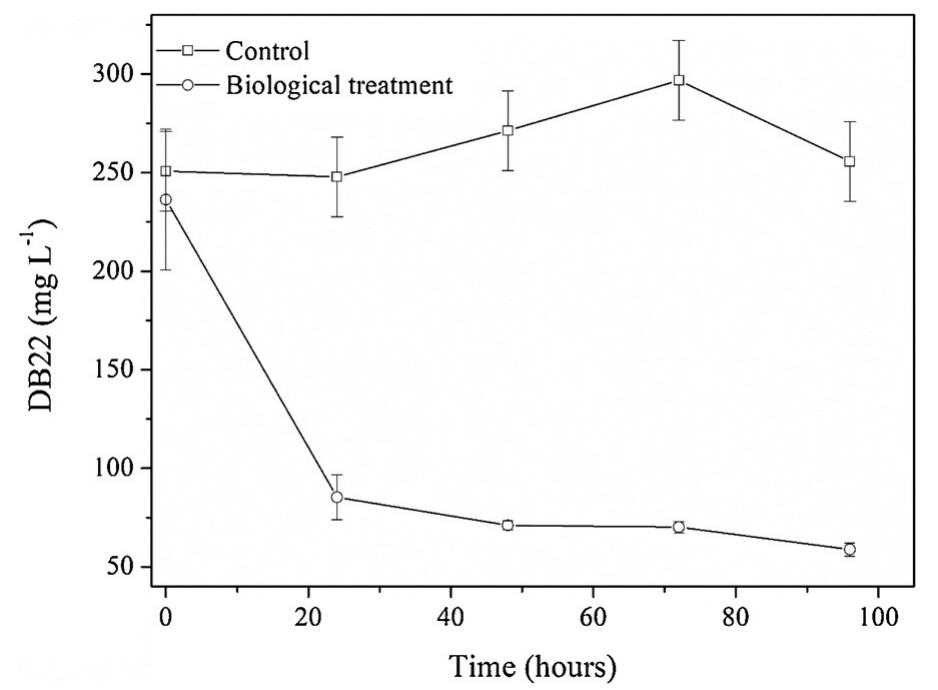
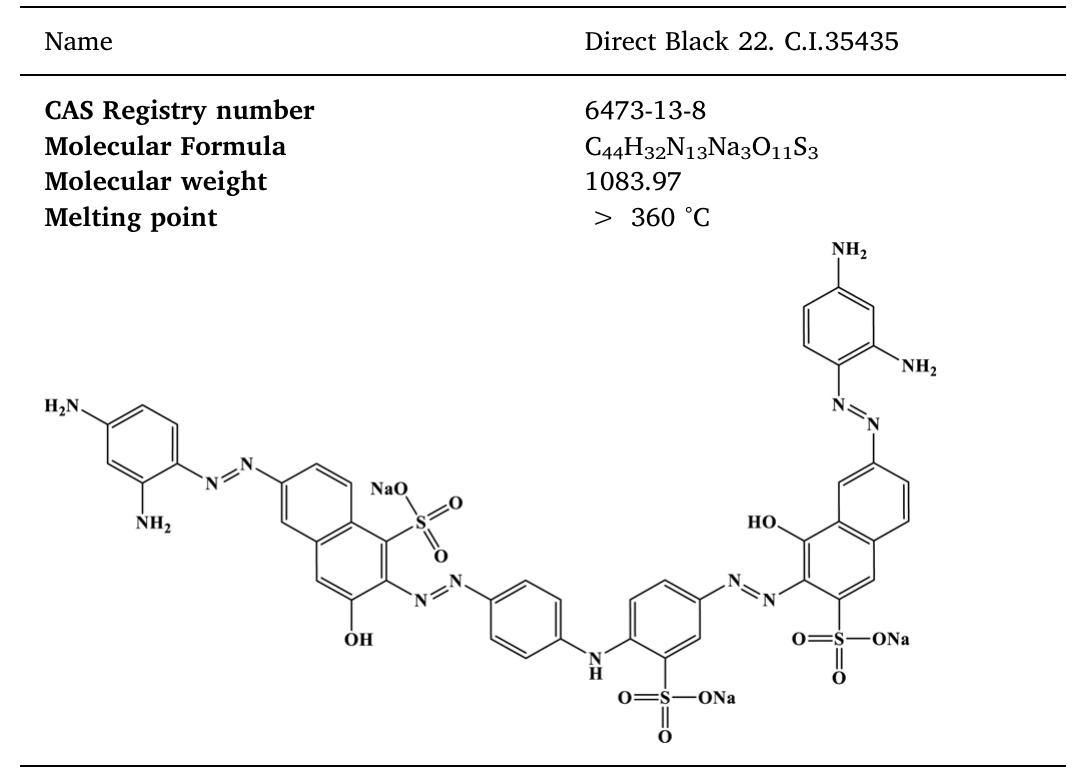
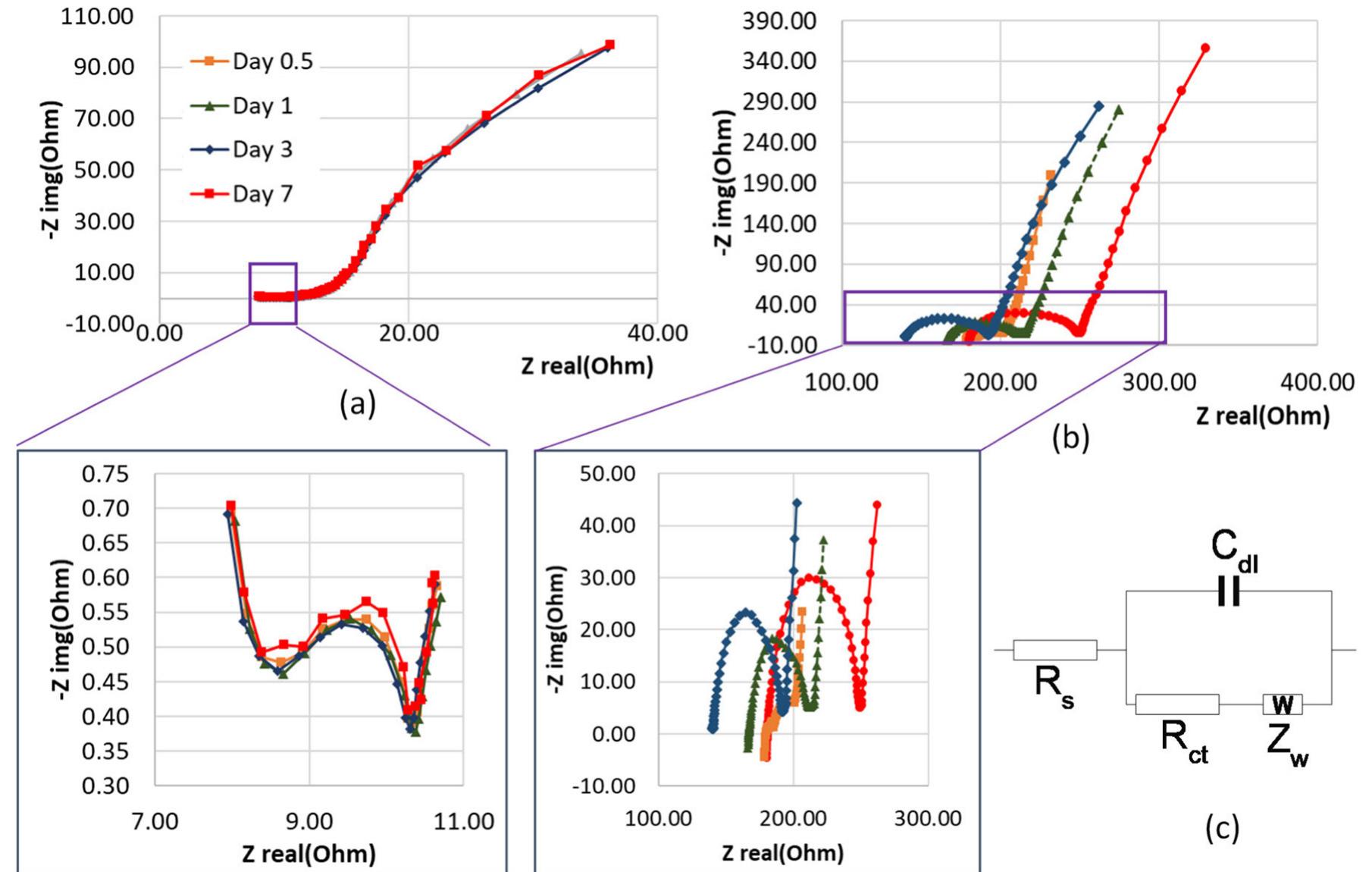
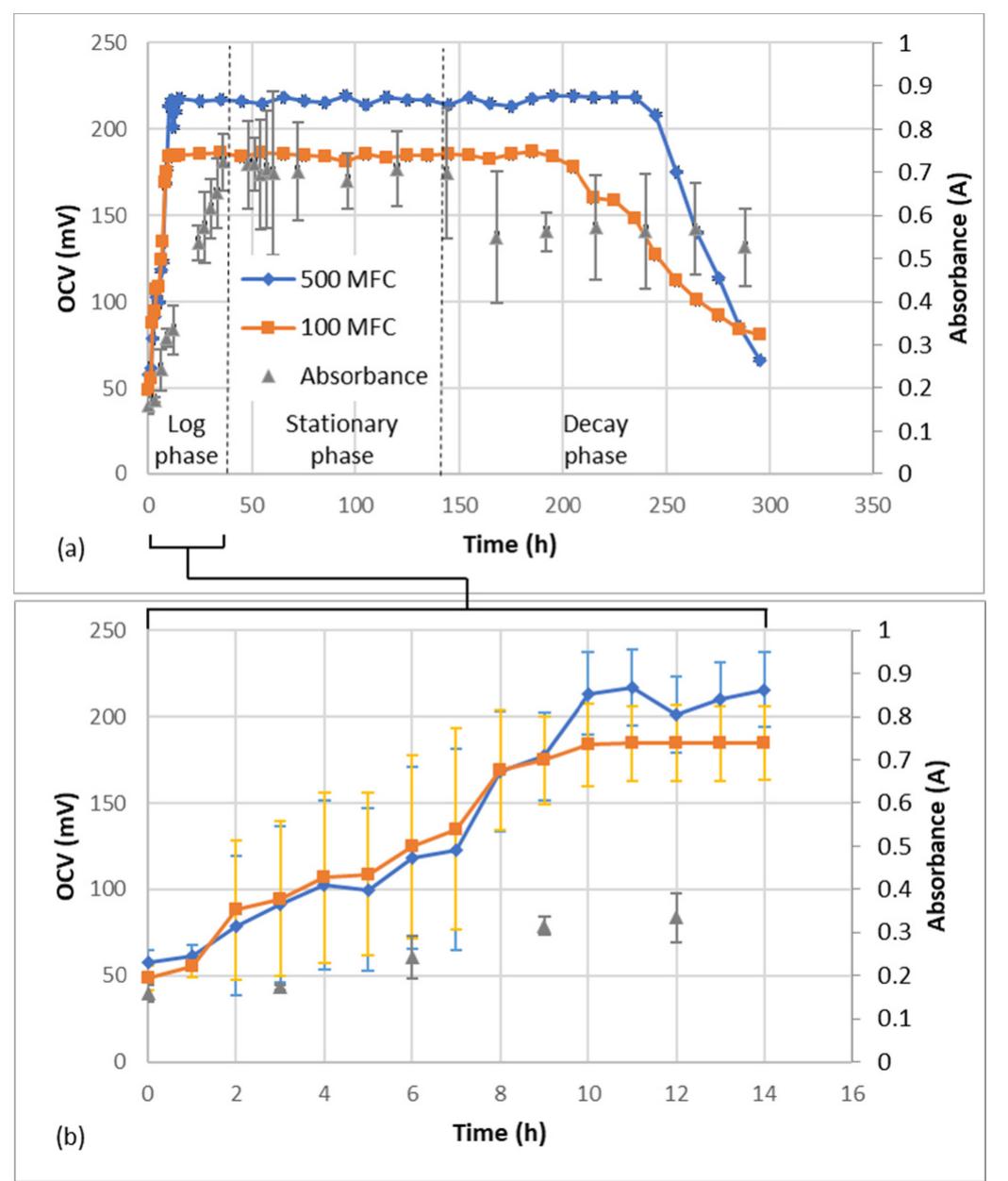
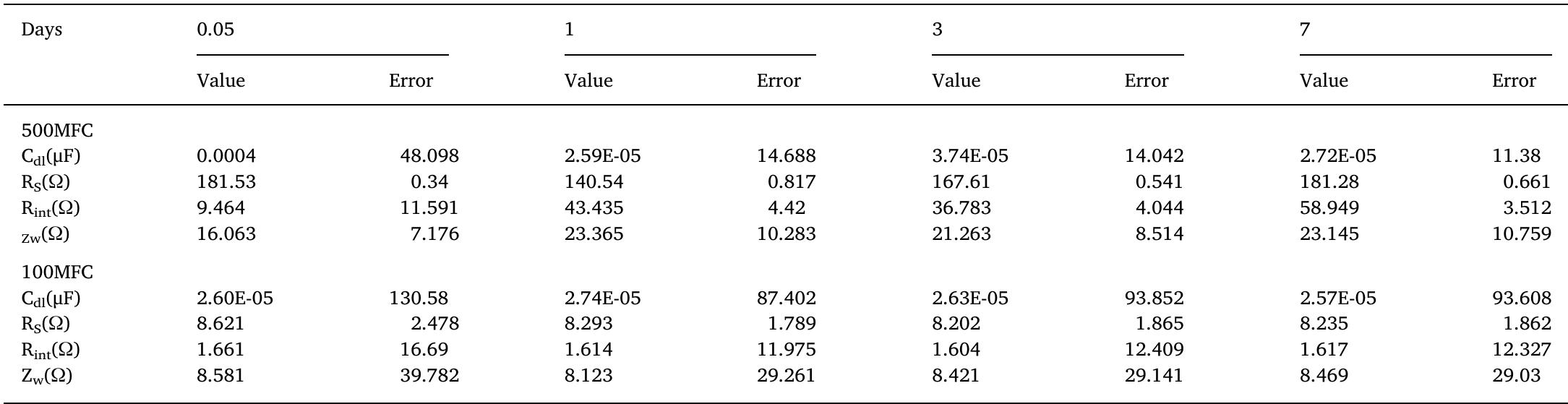
The treatment of textile wastewater (TWW) loaded with recalcitrant azo dyes in bioelectrochemical systems (BES) rather than in physicochemical processes is a low-cost and environmentally friendly process. The main objective of this study... more
Availability of clean water is of concern due to pollution and diminishing supply pollution. However, purification is possible depending on the incorporated contaminants. Domestic wastewater contains dissolved organic matter and its... more
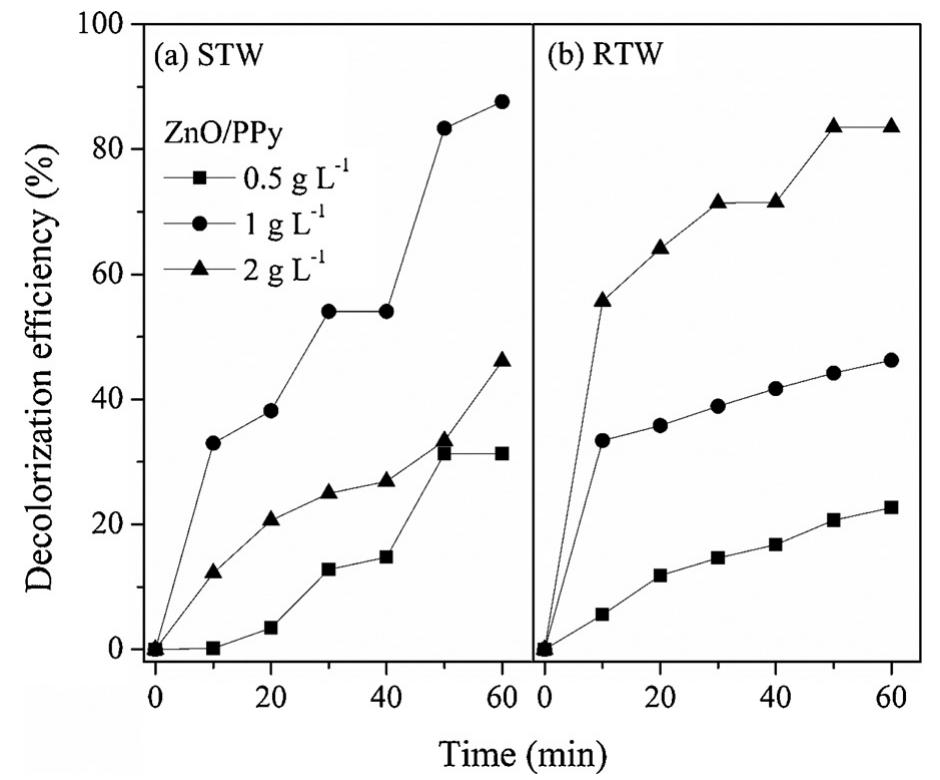
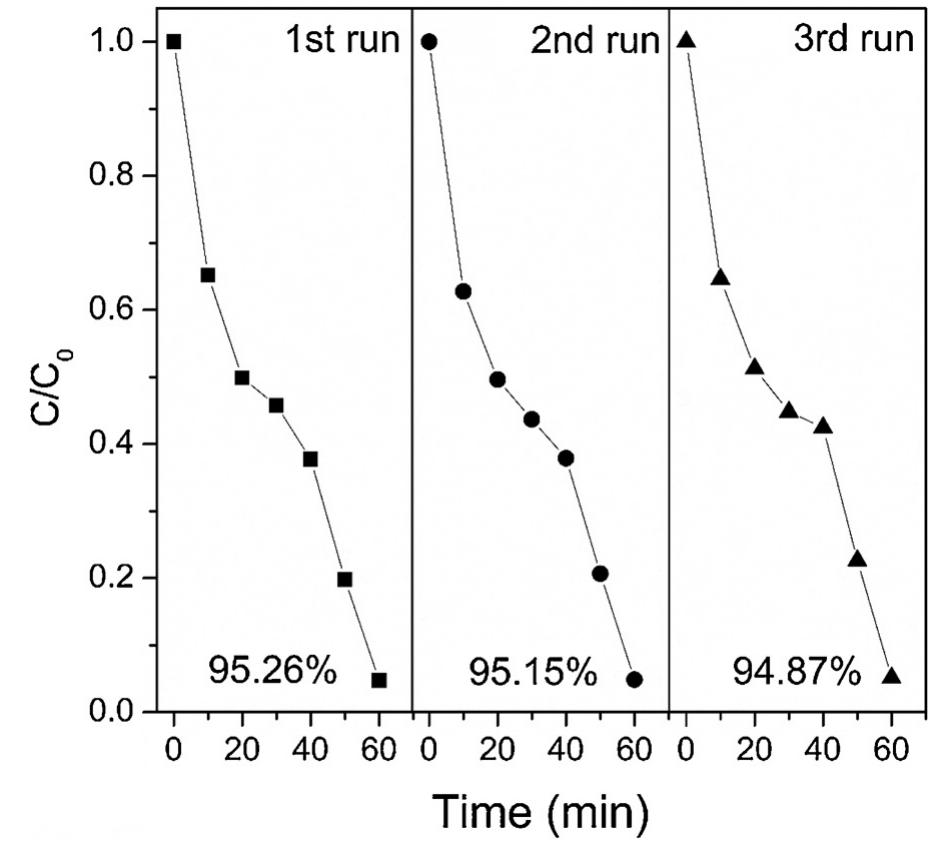
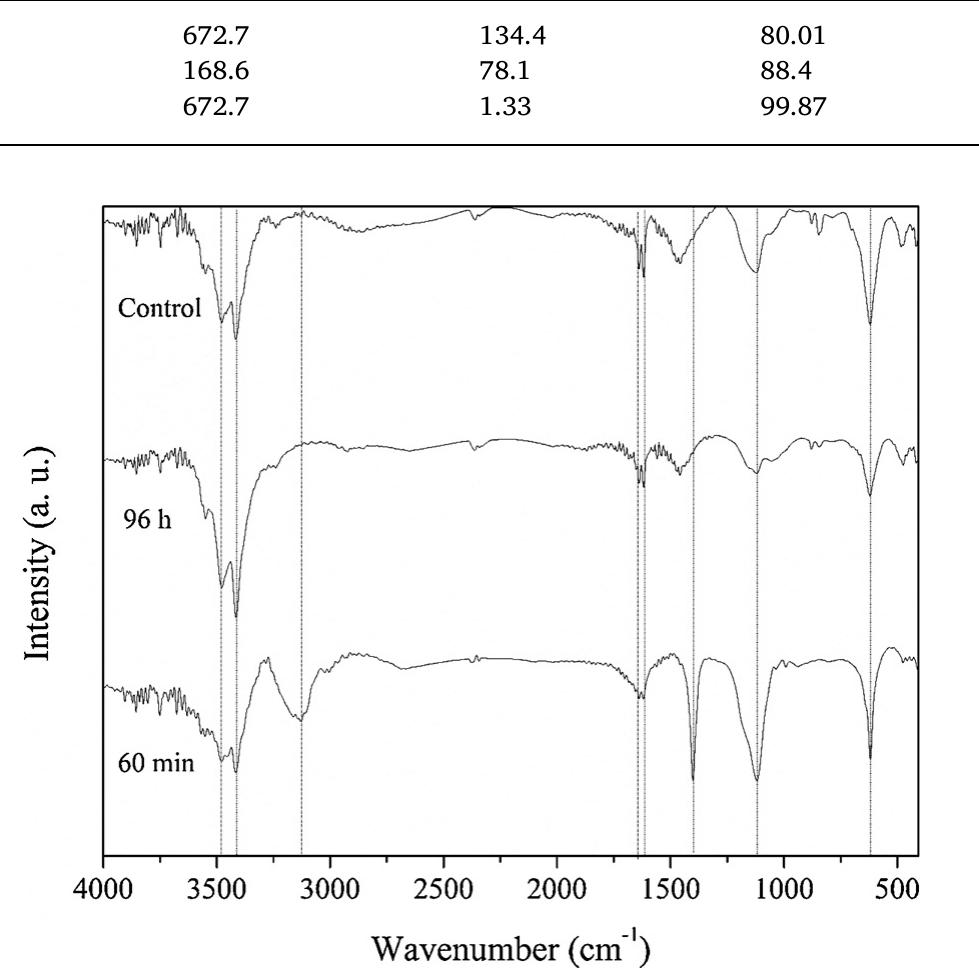
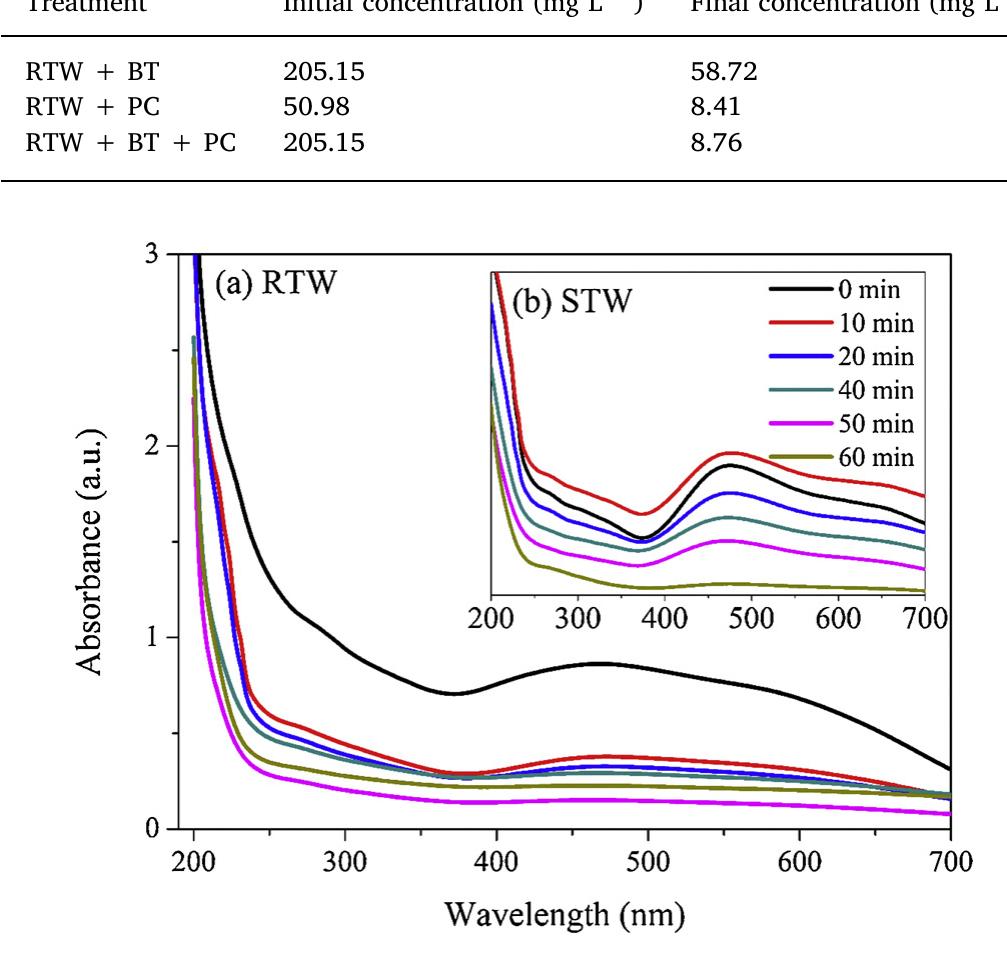
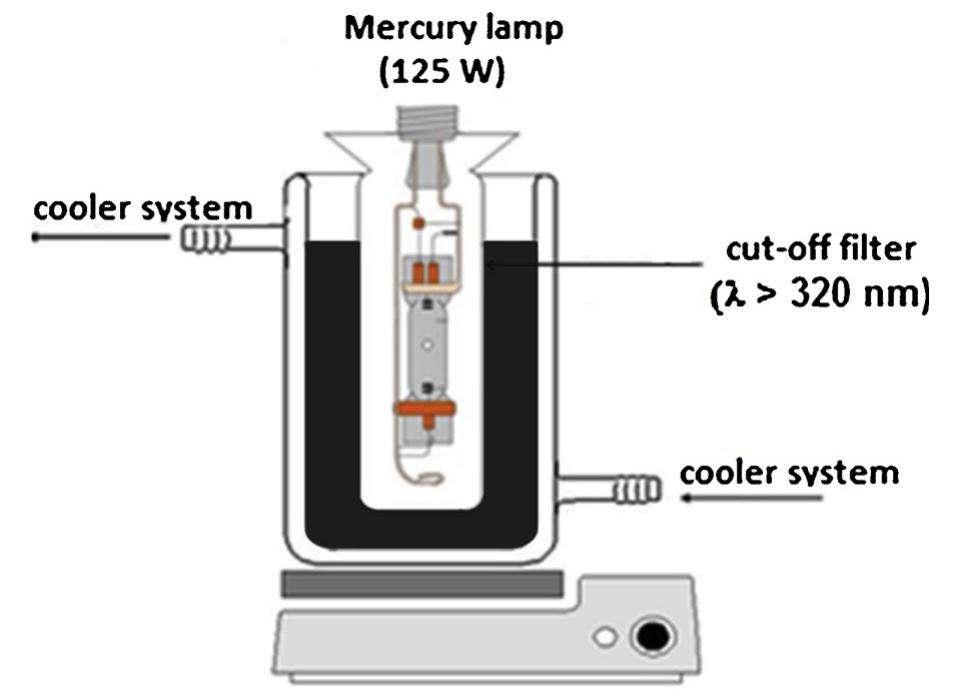
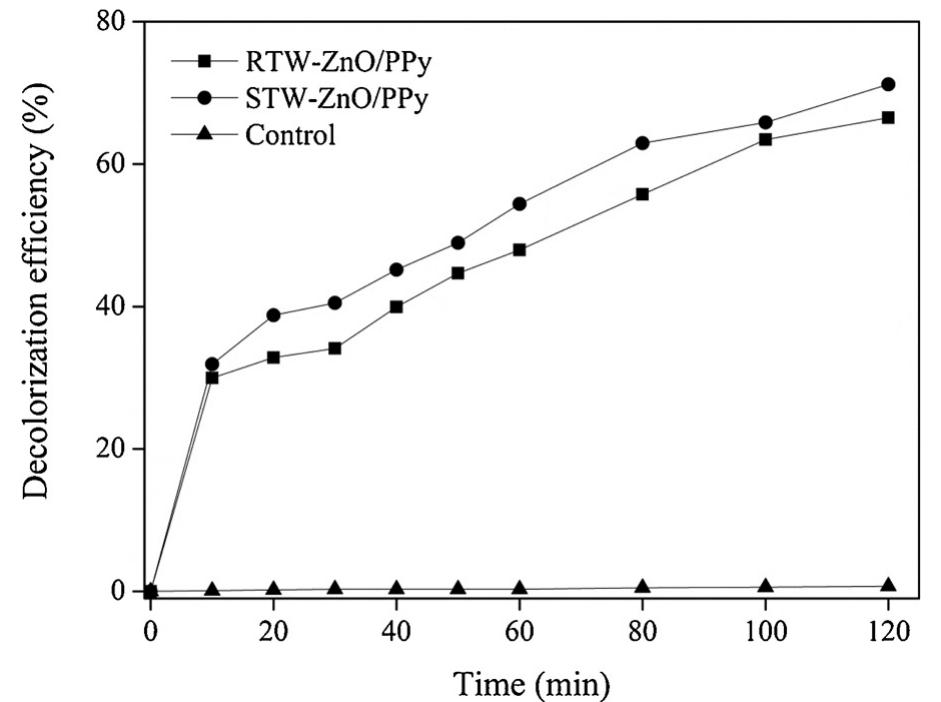
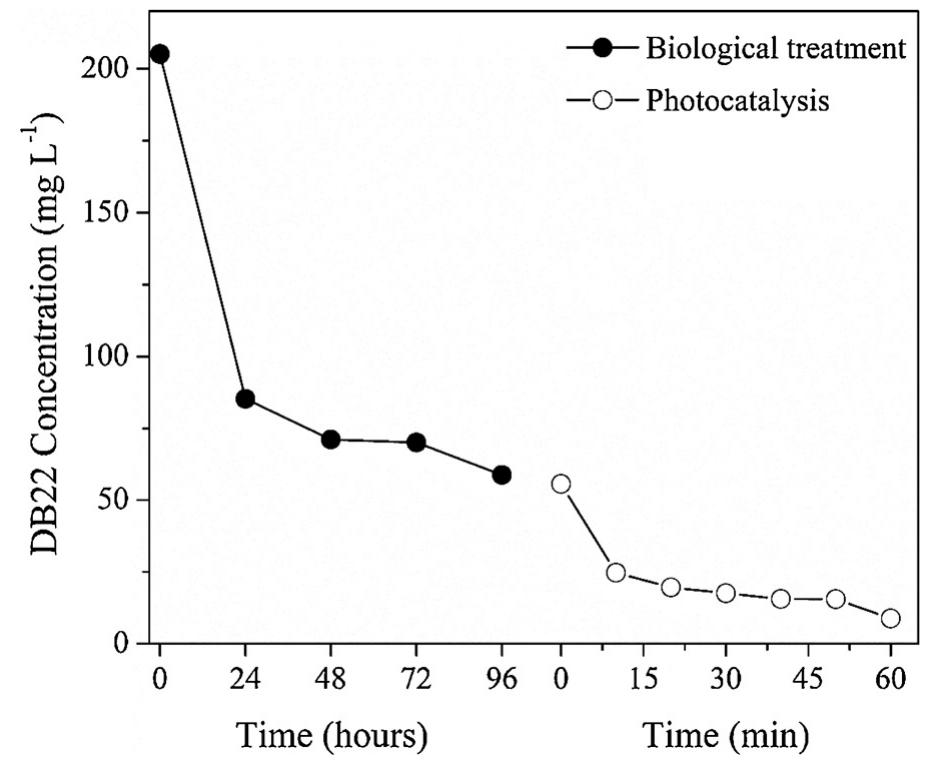
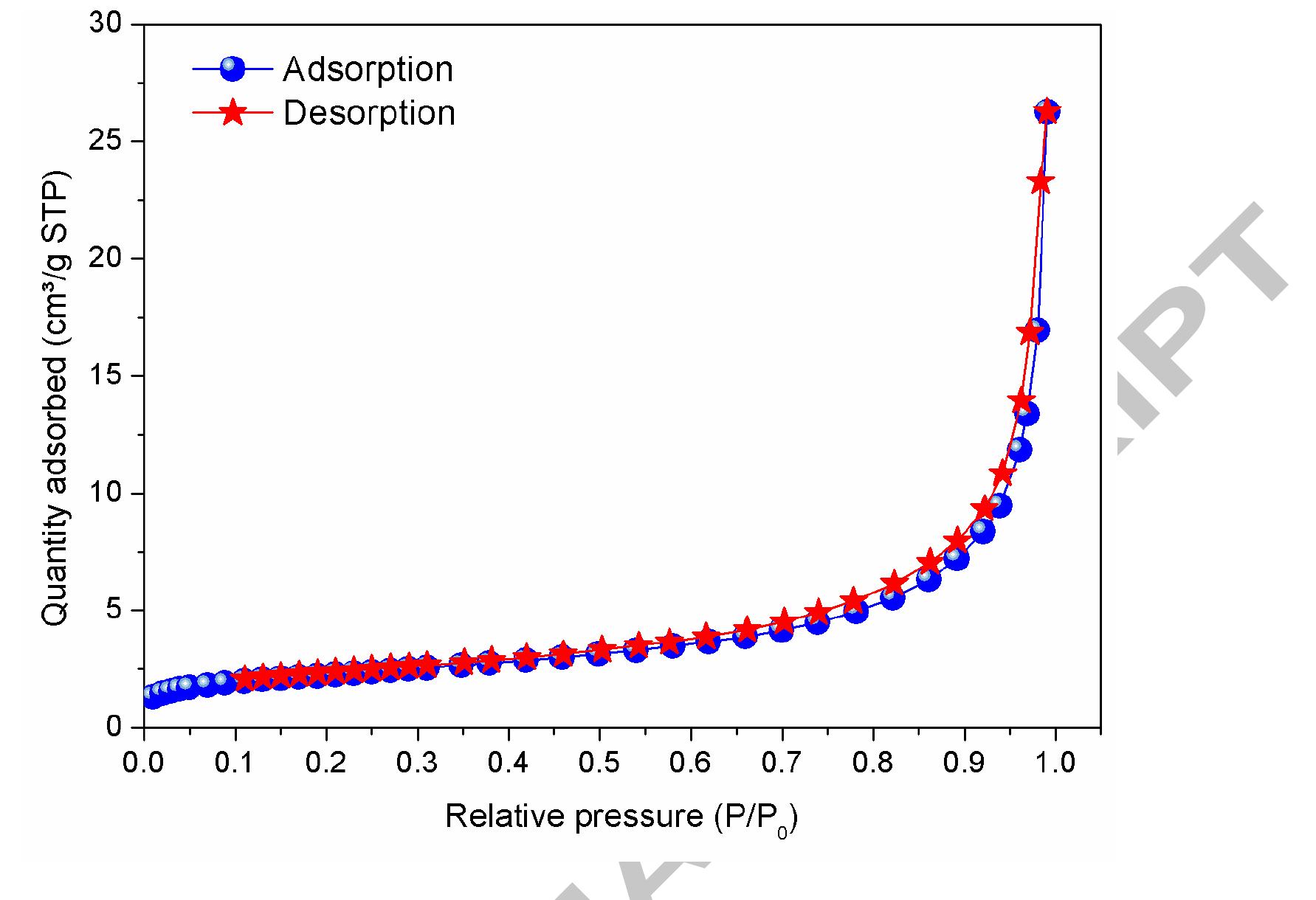
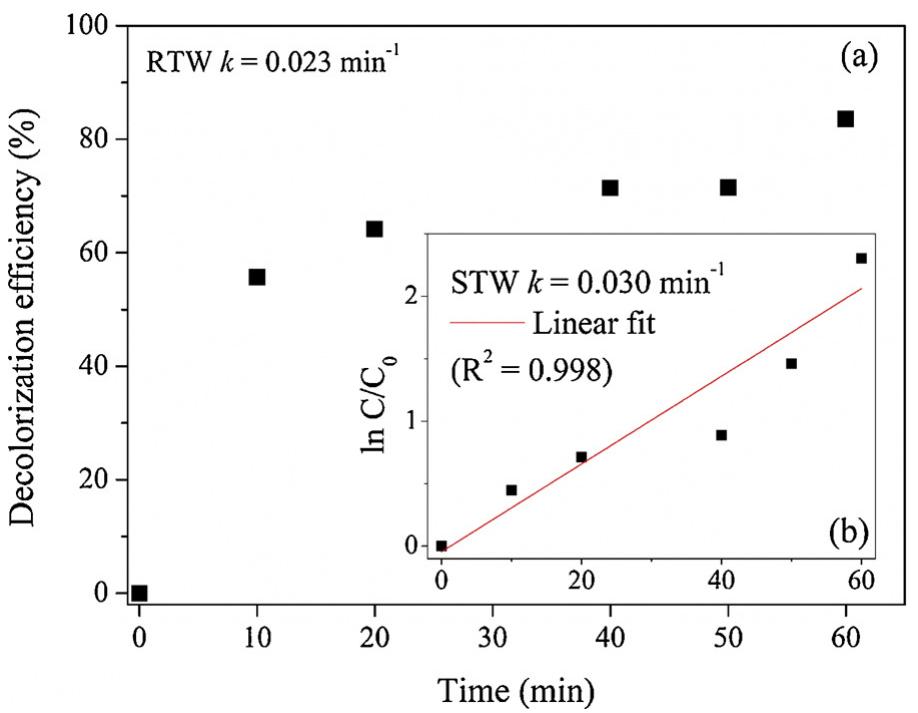
The novelty of this work lies in the use of a real textile wastewater (RTW), without a pretreatment, without additional carbon sources and undiluted, with coupling a photocatalysis by using an unprecedented composite for this application.... more
Decolorization of C.I. Acid Orange 7 (AO7) via TiO2-assisted photocatalysis under UV illumination in the presence of H2O2 has been investigated. The effect of pH on decolorization efficiency of AO7 was investigated in TiO2/UV/H2O2... more
In this study sunlight and UV radiation were used to compare the efficiency of decolorization of textile wastewater containing brilliant reactive red dye K-2BP (λmax = 534 nm) by the advanced oxidation process (AOP) using (H2O2/sunlight,... more
The discharges of the wastewater from the textile industry generate a substantial quantity of effluents which can cause various environmental problems, if disposed of without any prior treatment. Therefore, the treatment of these textile... more




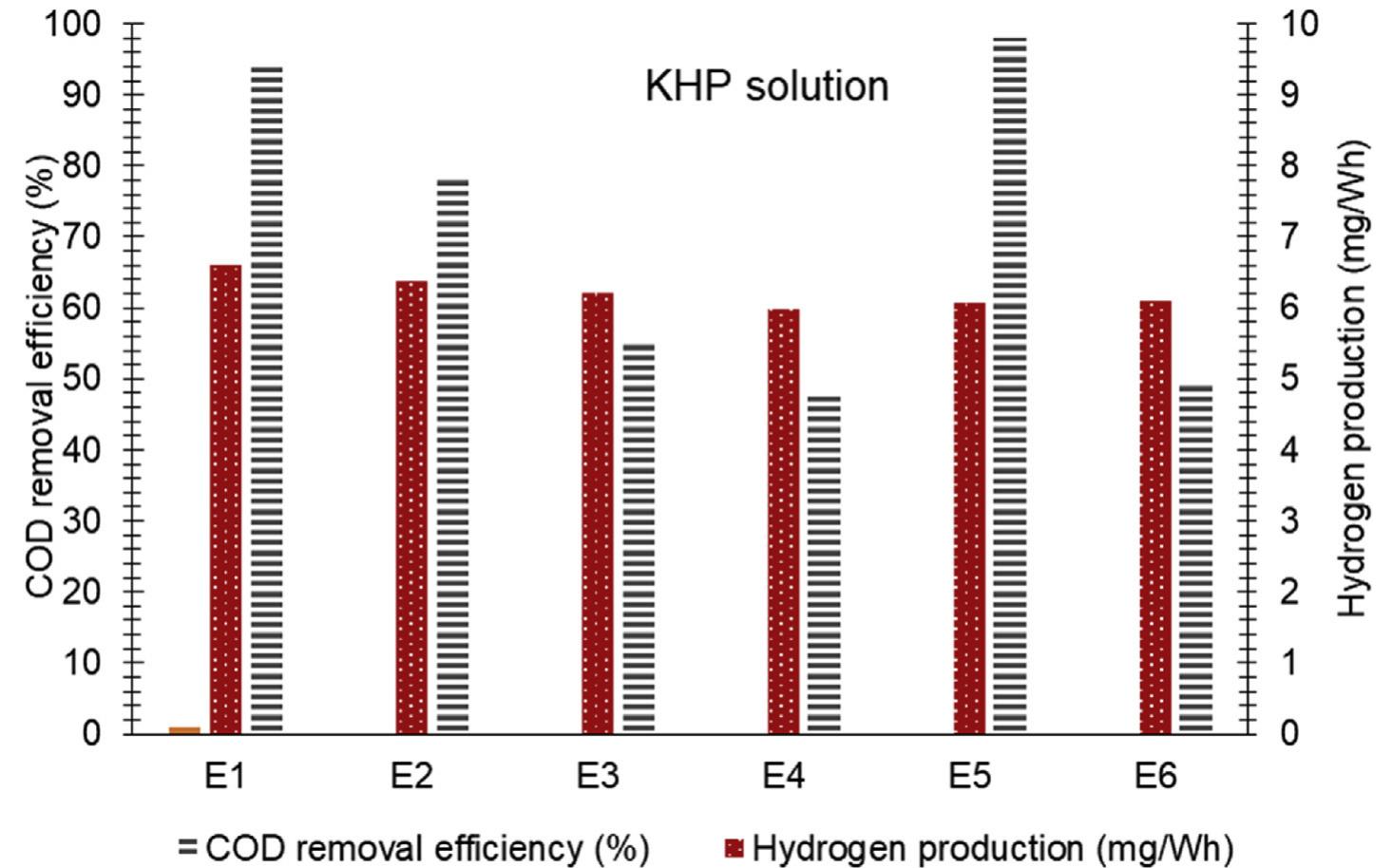
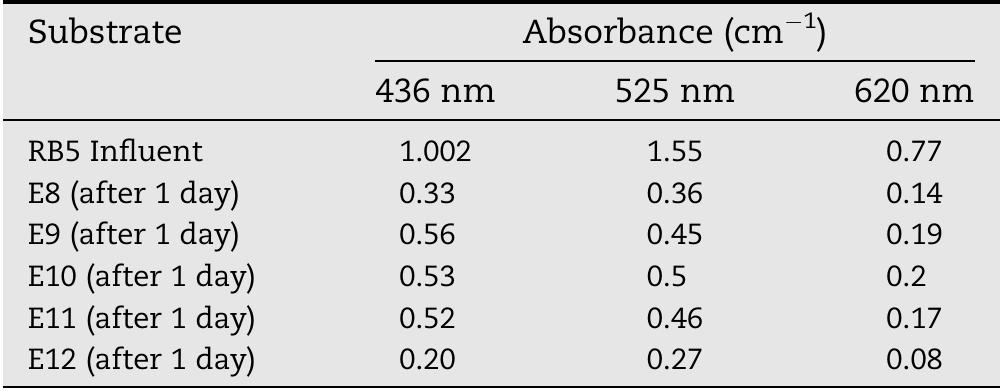
![Fig. 3 — Molecular structure of Potassium hydrogen phthalate (KHP) (a) and Reactive black 5 (RB5) (b). KHP solution is prepared by mainly following the proced- ure given by Standard Methods Committee [15], the concen- tration of the KHP is set twice as the standard COD solution. 850 mg of KHP is dissolved in deionized water and diluted to 1L. Potassium hydrogen phthalate theoretically has a COD of 1.176 mg O2/mg [26] therefore this solution has a theoretical chemical oxygen demand of 1 mg O,/mL [16]. Similar to the RBS solution, pH of the KHP solution is adjusted to 3 with the addition of H2SOx. The electrical conductivity of the solution is The absorption of photons possessing energy equal to or higher than that semiconductor which is 3.2 eV for titanium dioxide [24]:](https://figures.academia-assets.com/108733766/figure_003.jpg)
![‘ig. 1 — A classification of advanced oxidation processes. There has been a significant interest in sustainable develop- ment to meet increasing global energy and clean water de- mand due to population growth and higher living standards. As the depletion of fossil fuels for energy production resulting environmental problems such as global warming, alternative clean and renewable energy sources are needed. Hydrogen gas is an important alternative for fossil fuels since it is clean and renewable energy source. At the same time, due to the rising amounts of industrial, agricultural and domestic wastewaters generated daily, there is a growing concern. There is a strong need to treat wastewater and convert it into useful com- modities. In order to avoid the pollutant level causing severe health problems, wastewater quality standards are protected under regulations [1]. Conventional wastewater treatment methods such as physical treatment methods, biological processes, stripping and adsorption processes, and oxidation processes with chemicals for removing the organic contami- nants, consume high amounts of chemicals and energy. Recently, researchers are focused on processes that can pro- vide simultaneous hydrogen production and wastewater treatment since wastewater is a potential energy source due to its high organic content that can be transformed into hydrogen or methane gases. Electrohydrolysis process has been used to treat domestic [2] and olive mill wastewater [3] while producing hydrogen simultaneously. Iervolino et al. [4] produced hydrogen from glucose degradation in water and wastewater by using Ru-LaFeO;/Fe,03 magnetic particles photocatalysis and heterogeneous photo-Fenton processes. Besides hydrogen production, the efficiency of the wastewater treatment method is an important factor in order to protect environment. Since, chemical oxidation processes mineralize the pollutants to carbon dioxide, water and inorganics or transform them into harmless and nontoxic products [5], they are significant processes that can be utilized for simultaneous wastewater treatment and hydrogen production. Advanced oxidation processes (AOP) are special class of oxidation methods that utilize hydrogen peroxide, ozone and/or ultra- violet (UV) radiation to degrade the contaminants in the wastewater. AOPs are considered as preferable methods due to lack of toxicity of the reagents, their ability to oxidize in a variety of compounds, the absence of mass transfer limitation because of their homogeneous catalytic nature and the simplicity of the technology [6,7]. Even though advanced](https://figures.academia-assets.com/108733766/figure_001.jpg)
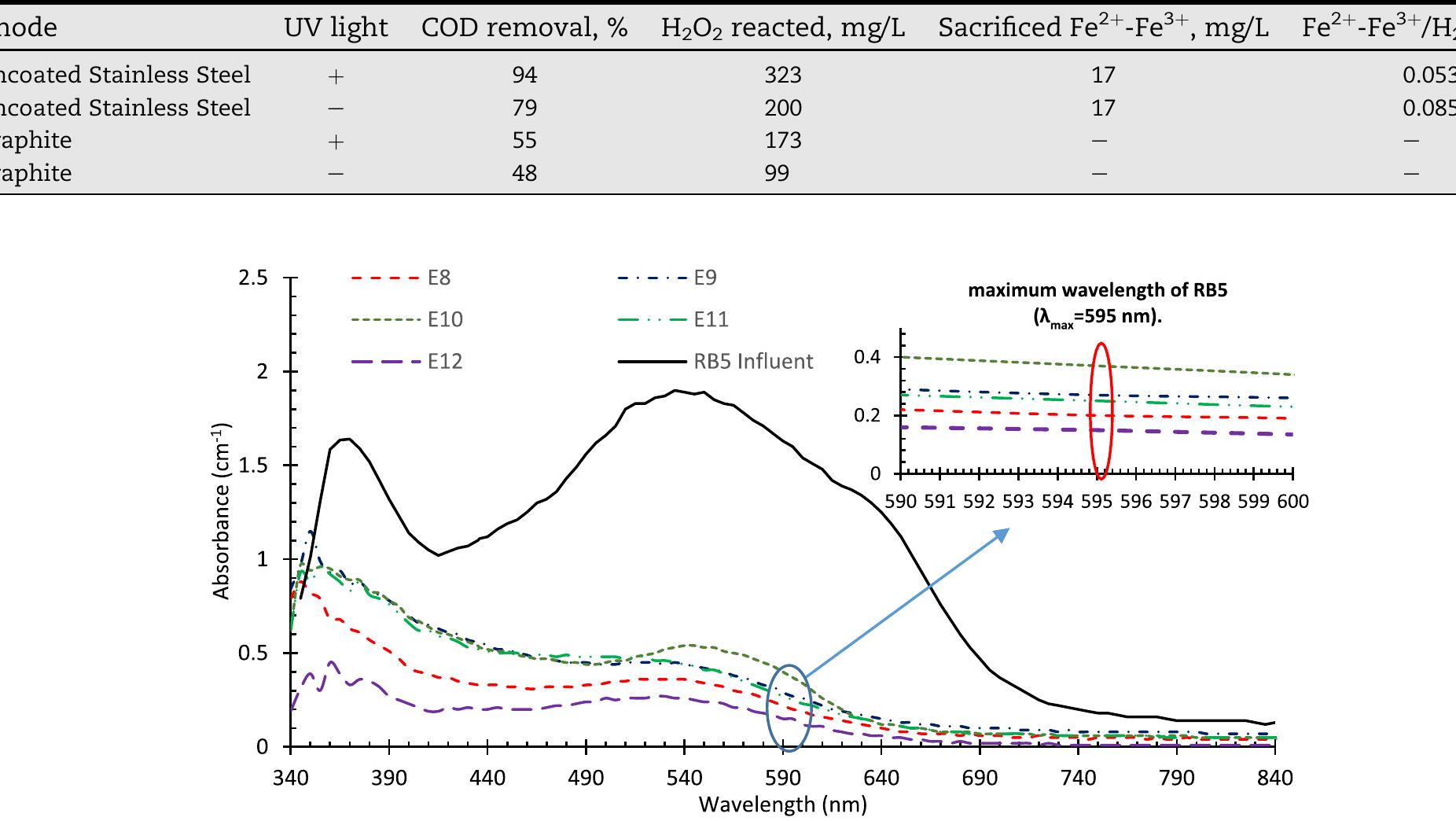
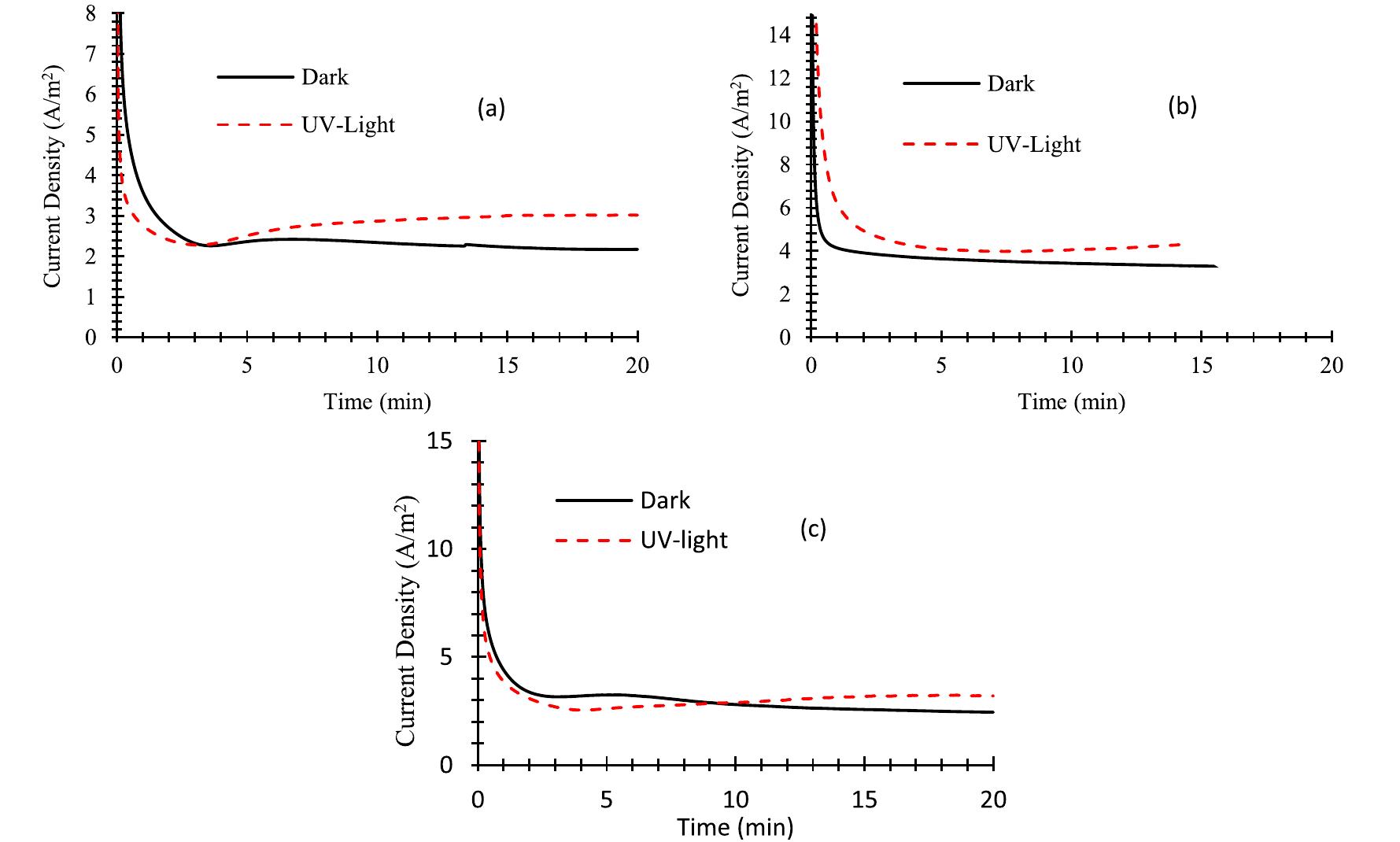
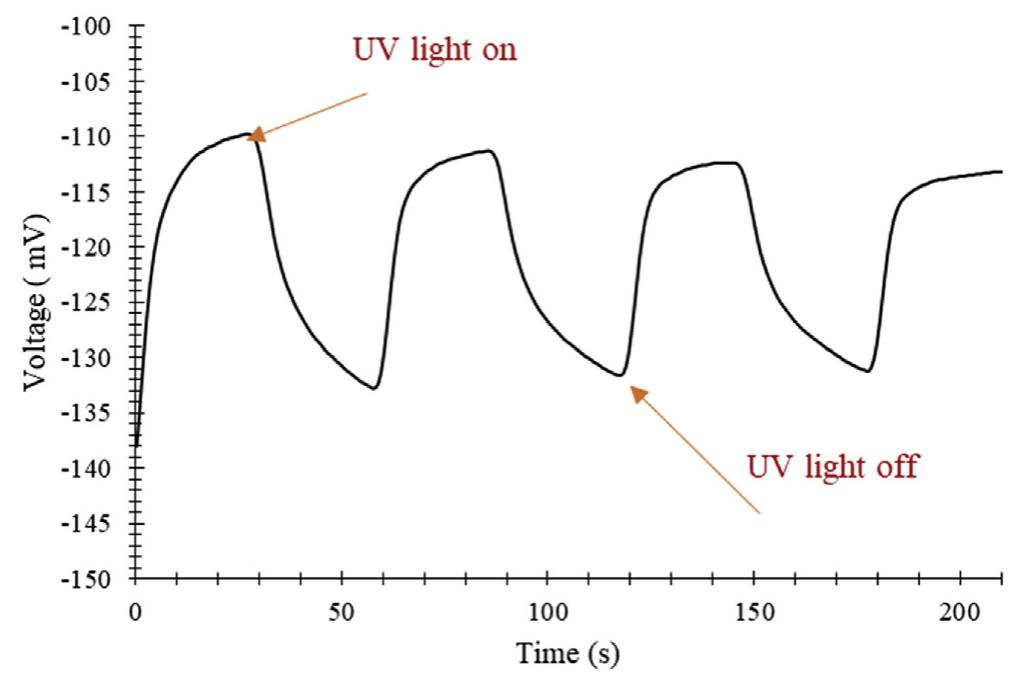
![2+ Fe.’ +H,0.—Fe;; +HO* +OH™ Fig. 4 — Hydroxyl radical formation mechanisms in the reactors used in this study. The Fenton type reactions take place in the presence of HO, and iron ions in an acidic medium. Hydrogen peroxide is splitting with the iron catalyst and generates hydroxyl radi- cals as follows [26]: H,0, "32 HO* The UV light can also be used in the process of hydrogen peroxide decomposition for hydroxyl radical production by the following reactions [31]: In this study, in order to observe the effect of UV/Ti02 system in wastewater degradation and hydrogen production. Two other experimental studies are also executed as being one without UV light, the other one without TiO, coating. Any process increases the reaction rate of reduction of ferric to ferrous can be used as a catalyst for the given reaction chain above. In electro-Fenton applications, this is achieved electrochemically. One way to accelerate this reaction rate is irradiation by UV/V light as follows [30]: Furthermore, some portion of the adsorbed compound can be oxidized directly via electron migration as follows [25]: These reactions have a vital importance in photo-oxidative degradation processes due to the high concentration of adsorbed water or hydroxide on the particle surface. The rate of reaction (5) is much higher than compared to the corresponding reaction (6), so it can be clearly stated that, Fenton reaction (5) with an excess amount of hydrogen peroxide is rate-limiting only for the initial time interval (in the order of seconds or minutes). After this short-term, the oxidization rate is controlled by the rate of oxidation is controlled by reaction (6) [26—29]:](https://figures.academia-assets.com/108733766/figure_004.jpg)
![Fig. 4. Decolorization efficiency of simulate textile wastewater (STW) by ZnO, PPy composite, precursors of composite and lamp (photolysis). Polypyrrole is commonly used as a dye adsorbent by intraparticle diffusion [43], due to the size and spherical-shape of the polymer (see](https://figures.academia-assets.com/107625837/figure_003.jpg)