Key research themes
1. How do magnetic matrices and separator configurations optimize high gradient magnetic separation (HGMS) for efficient extraction of rare earth and weakly magnetic particles?
This research area investigates the design, material properties, and geometric configurations of magnetic matrices and separators to enhance the magnetic field gradient and thus improve the capture efficiency of weakly magnetic or rare earth particles during HGMS. Understanding and optimizing these factors are critical to achieving high selectivity and throughput for fine and paramagnetic minerals, which are otherwise difficult to separate due to their low magnetic susceptibility.
2. What selective and environmentally-friendly chemical approaches enable efficient extraction and recycling of Nd-Fe-B magnet phases?
This research theme addresses selective chemical leaching and extraction techniques to separate matrix and rare earth-rich phases in Nd-Fe-B magnets, with a focus on methods that are more sustainable and environmentally benign than traditional pyrometallurgical or harsh hydrometallurgical routes. The goal is to achieve phase-specific removal—particularly enriching the magnetic Nd2Fe14B matrix—while minimizing damage and process hazards, therefore enhancing yields and recyclability of rare earth elements crucial for green technologies.
3. How can magnetic fields and magnetic nanoparticles be leveraged for enhanced solid phase extraction and biochemical separation applications?
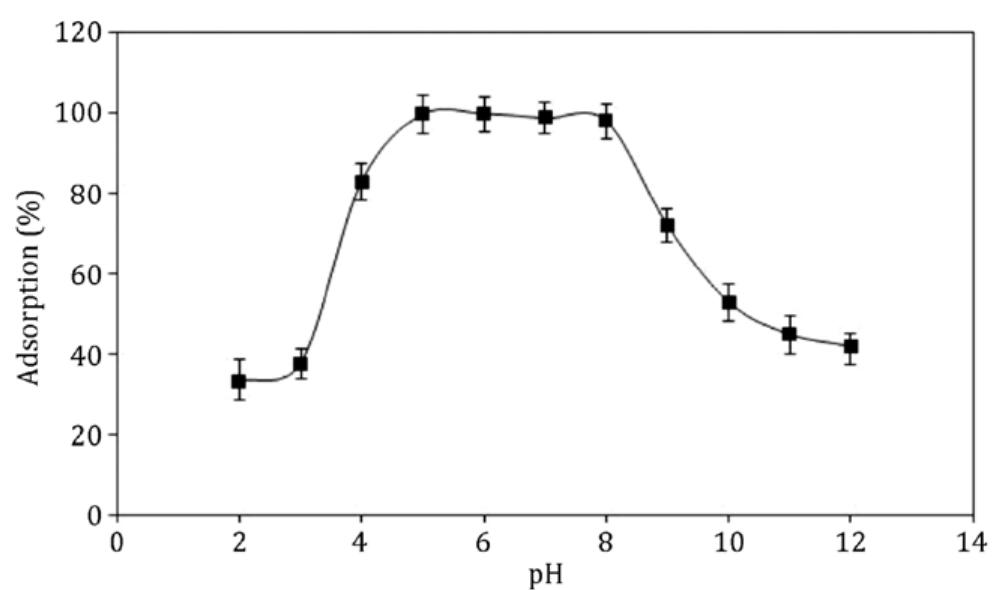
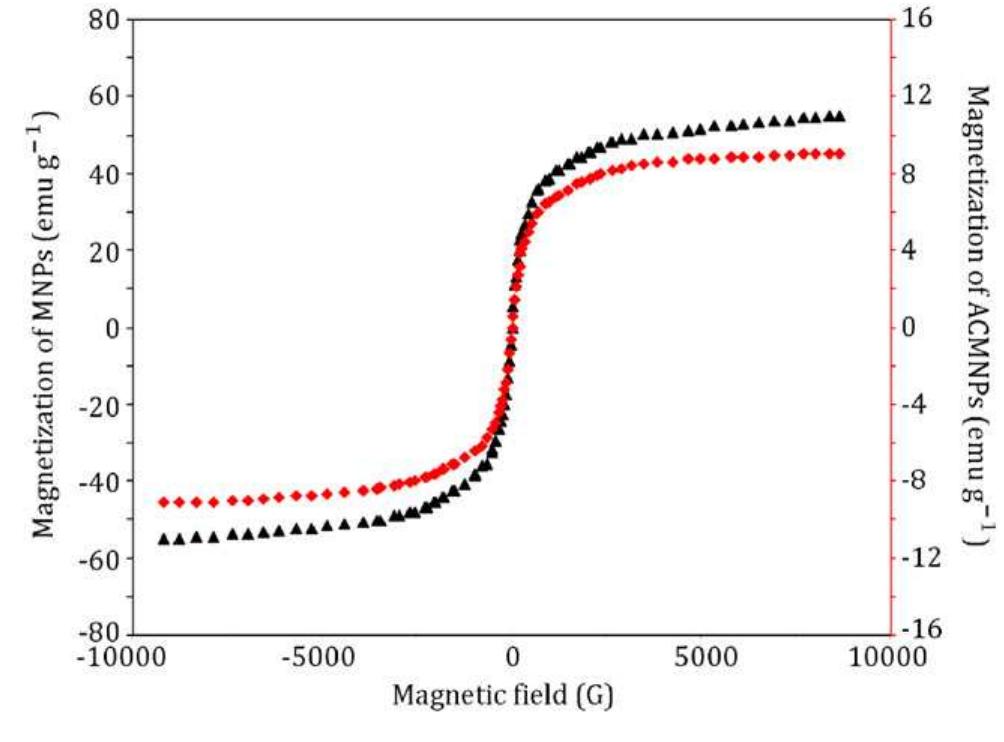
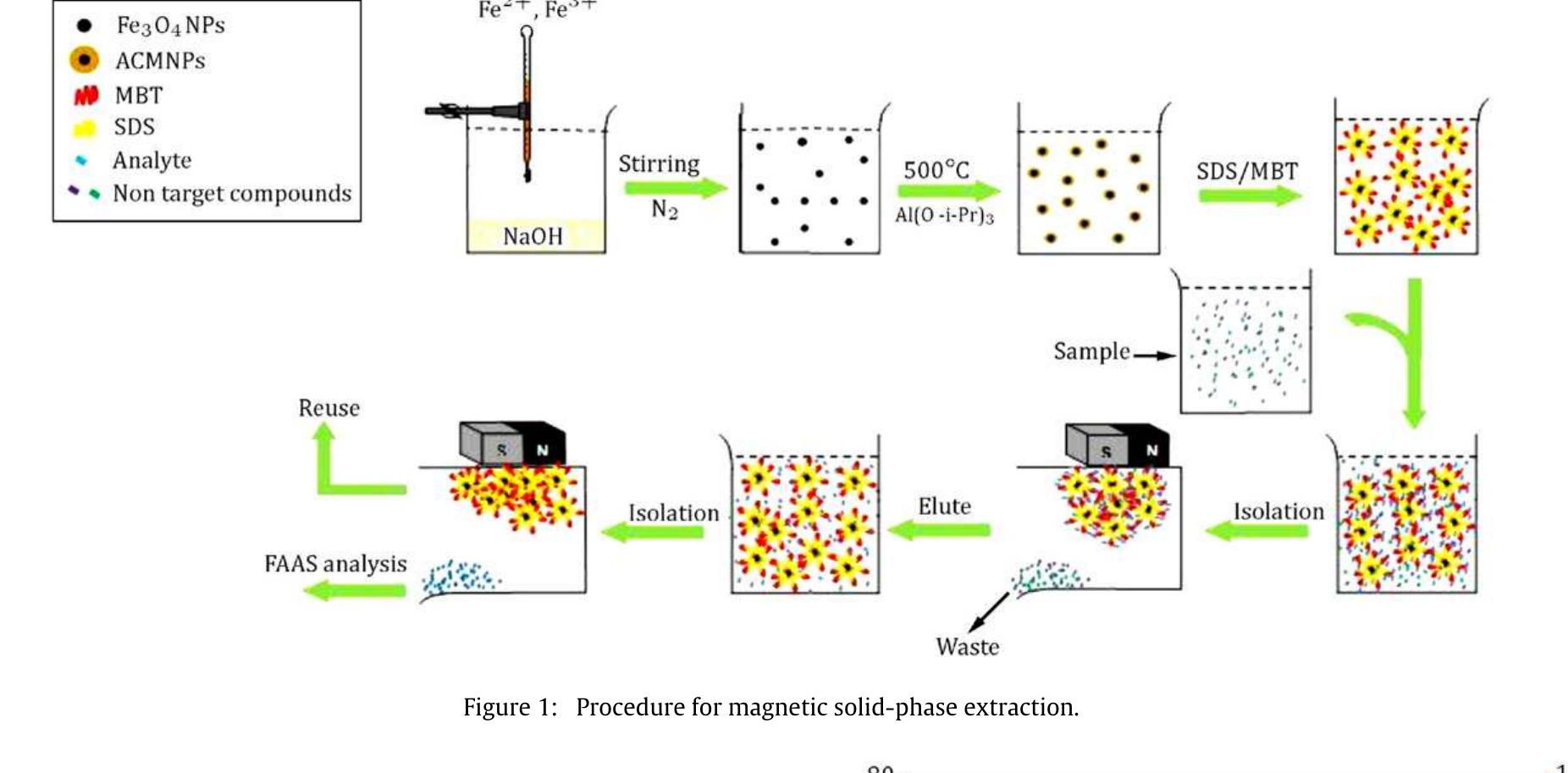
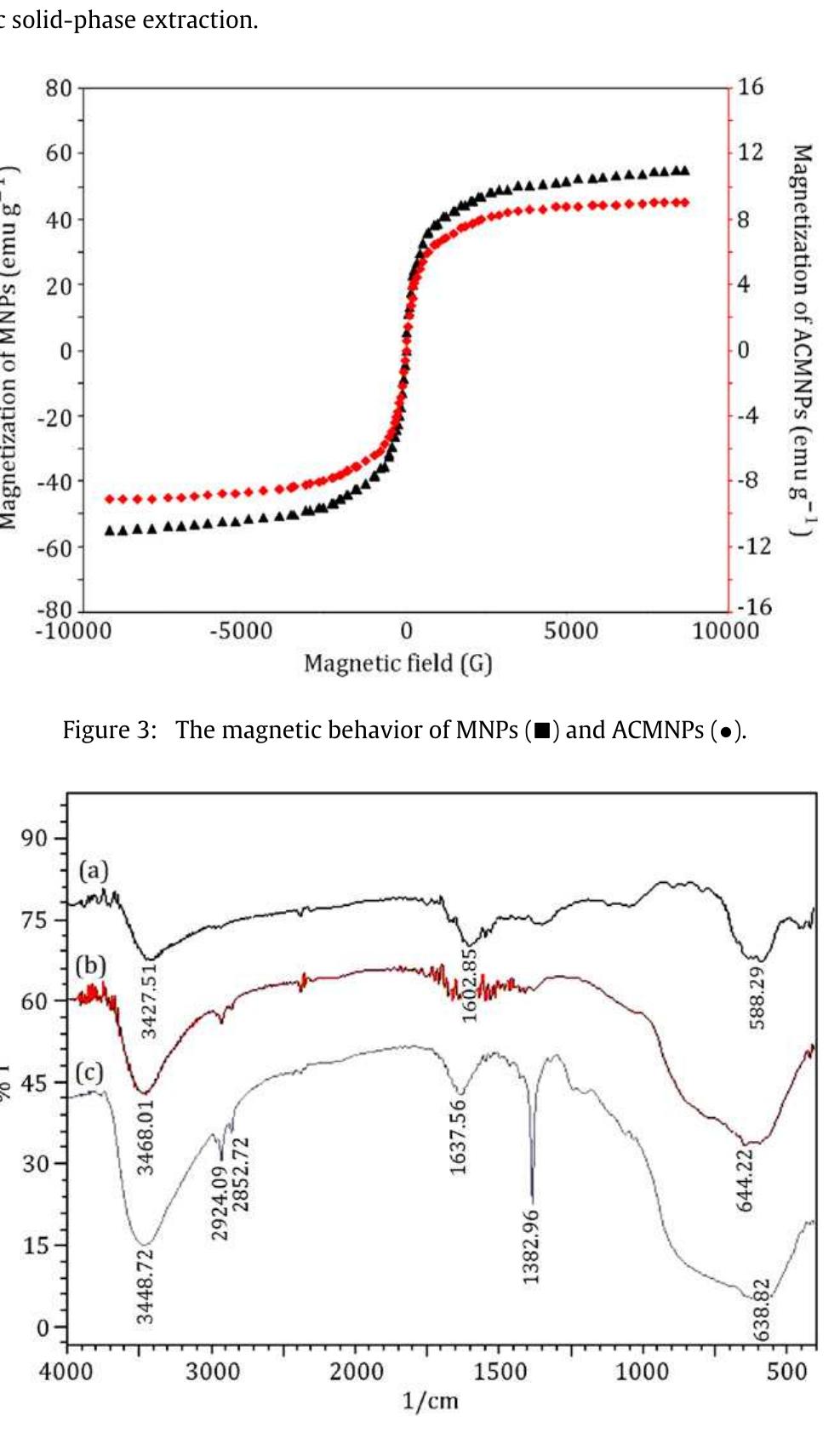
This theme explores the use of external magnetic fields and magnetically responsive materials—such as magnetic nanoparticles and ferrofluids—for improving separation efficiencies in solid phase extraction and bio-chemical contexts. Research focuses on magnetic field gradient measurement, design of magnetic matrices, and microfluidic manipulation of diamagnetic and paramagnetic particles, aiming to enable rapid, selective, and environmentally friendly extraction methods suited for biological and environmental matrices.



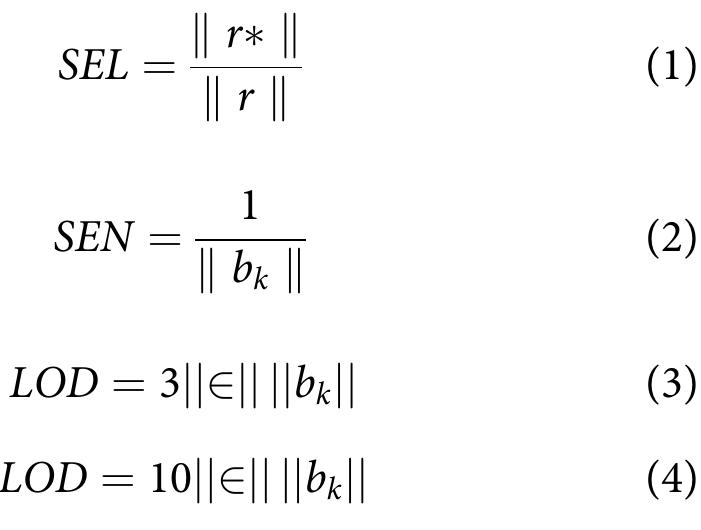
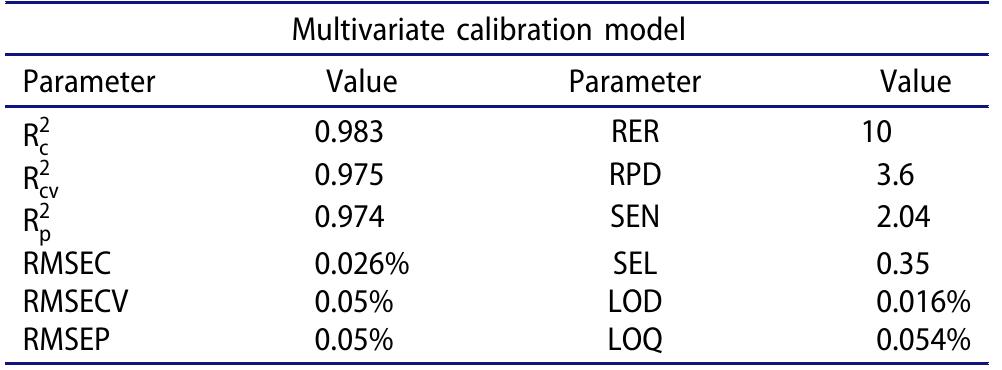
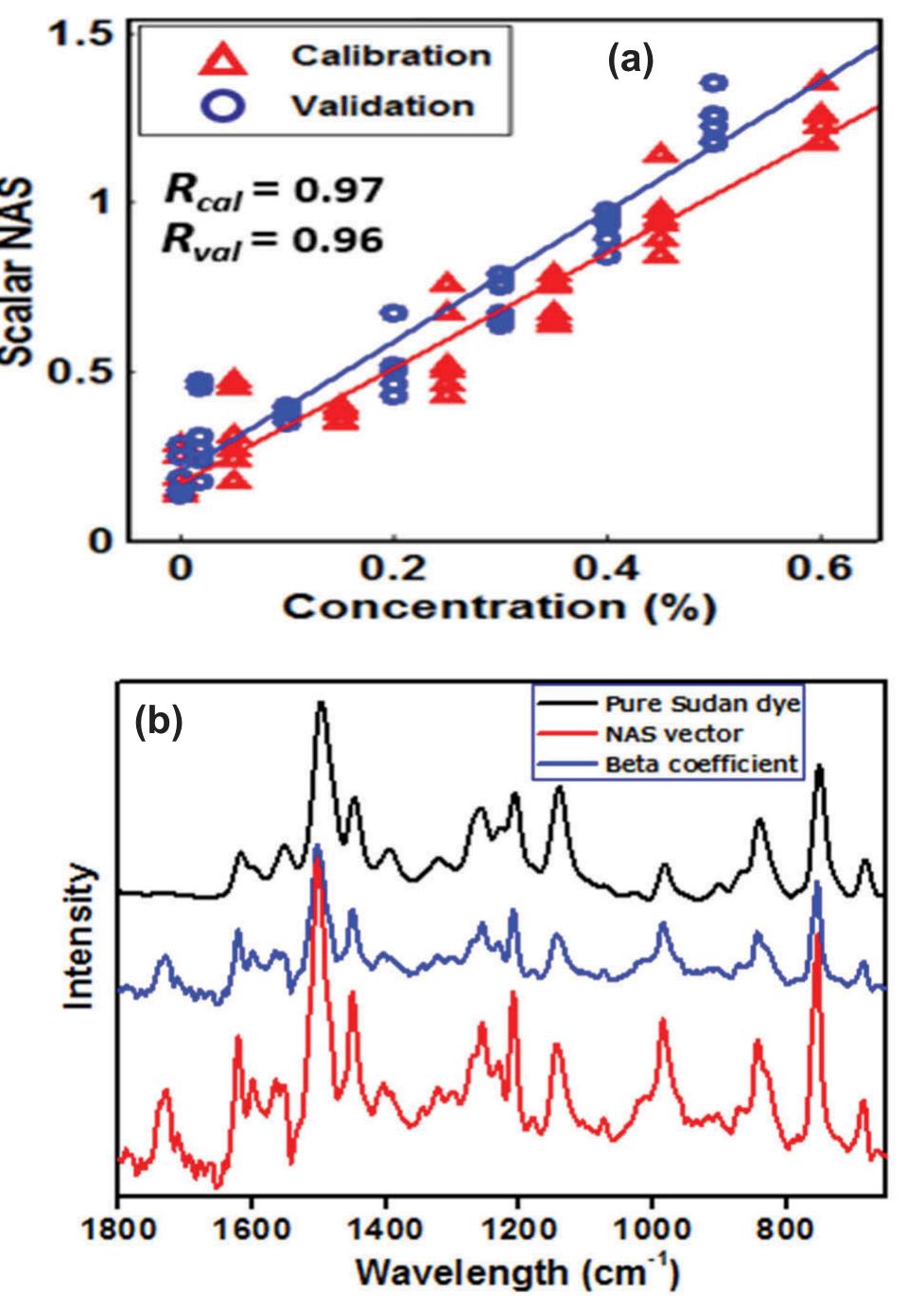
![indication and the intendment of the decision 2002/657/EC, three conditions must be satisfied: first, two SRM transitions have to be considered for both identification and quantifica- tion; the distinction of the two transitions (the most intense for quantification and the less intense for qualification) might, in We overcame the problem considering some conditions that were largely discussed elsewhere [52]. Briefly, (i) at MQL, the response must be still linear and, (11) following the](https://figures.academia-assets.com/100090839/figure_003.jpg)
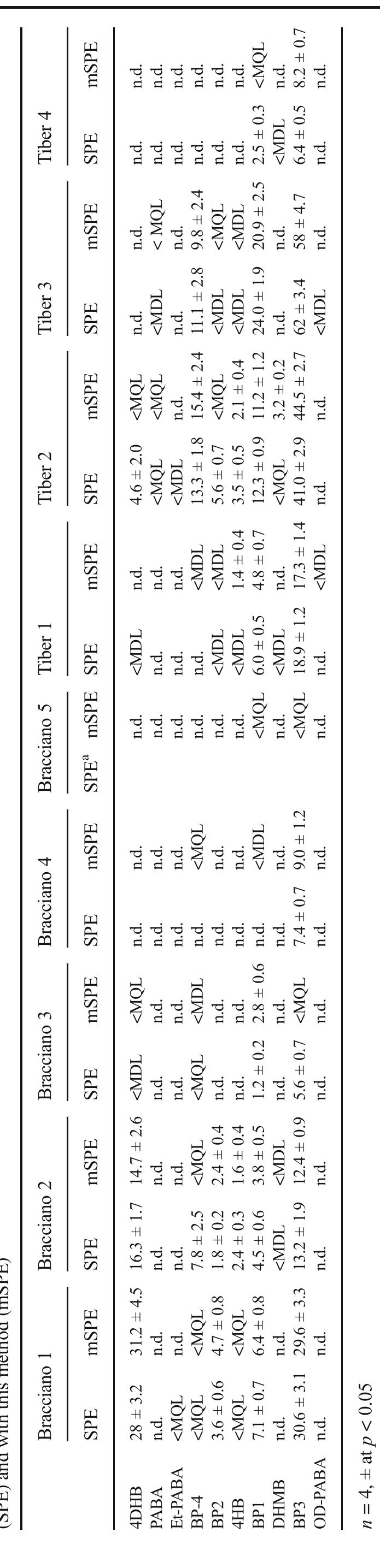
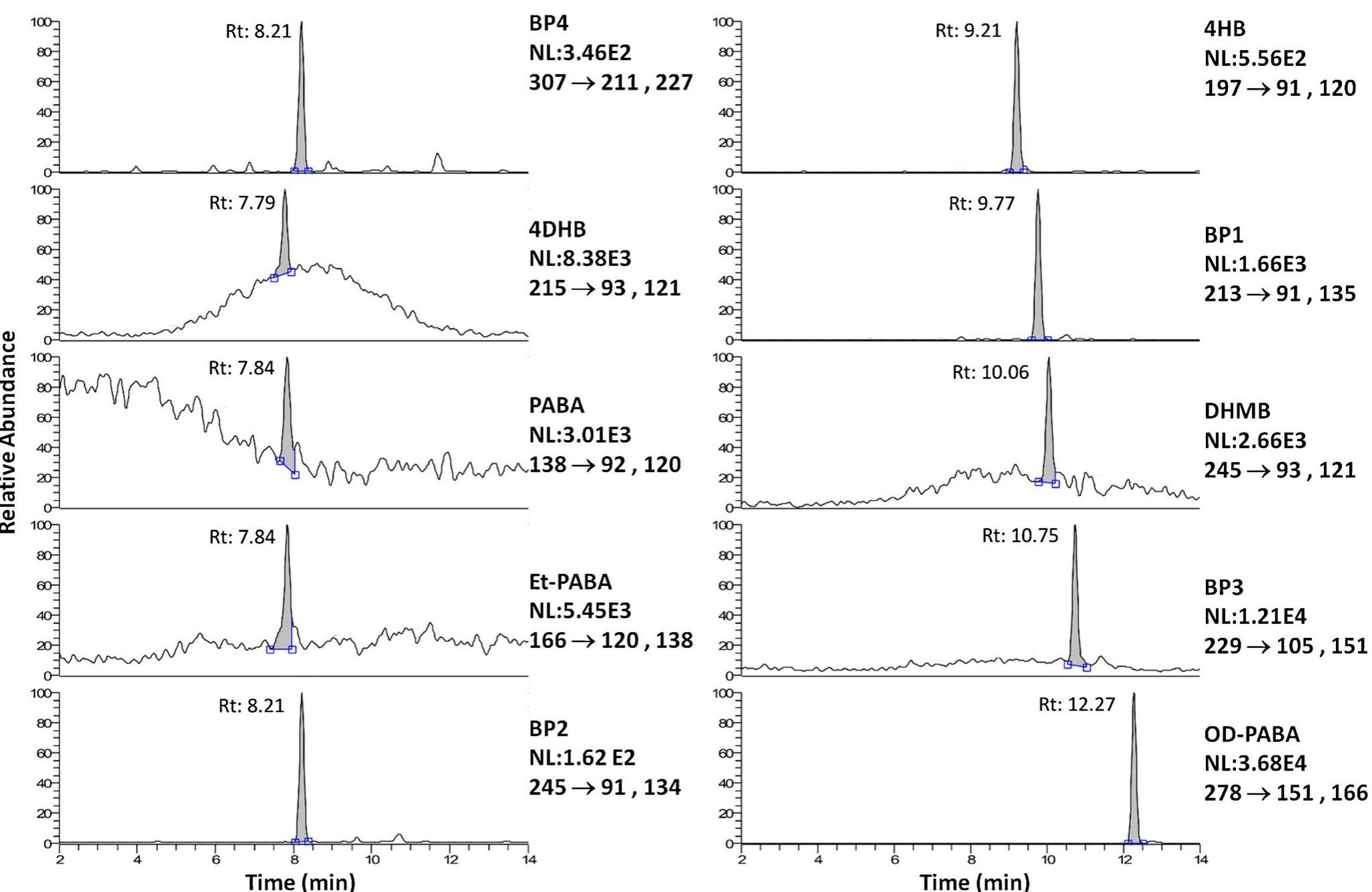
![Fig. 2 Sum of the two LC-SRM traces relative to the compounds found in the sample named “Bracciano 1.” a Method reported in [39]; b this method For concentrations, see Table 5](https://figures.academia-assets.com/100090839/figure_002.jpg)
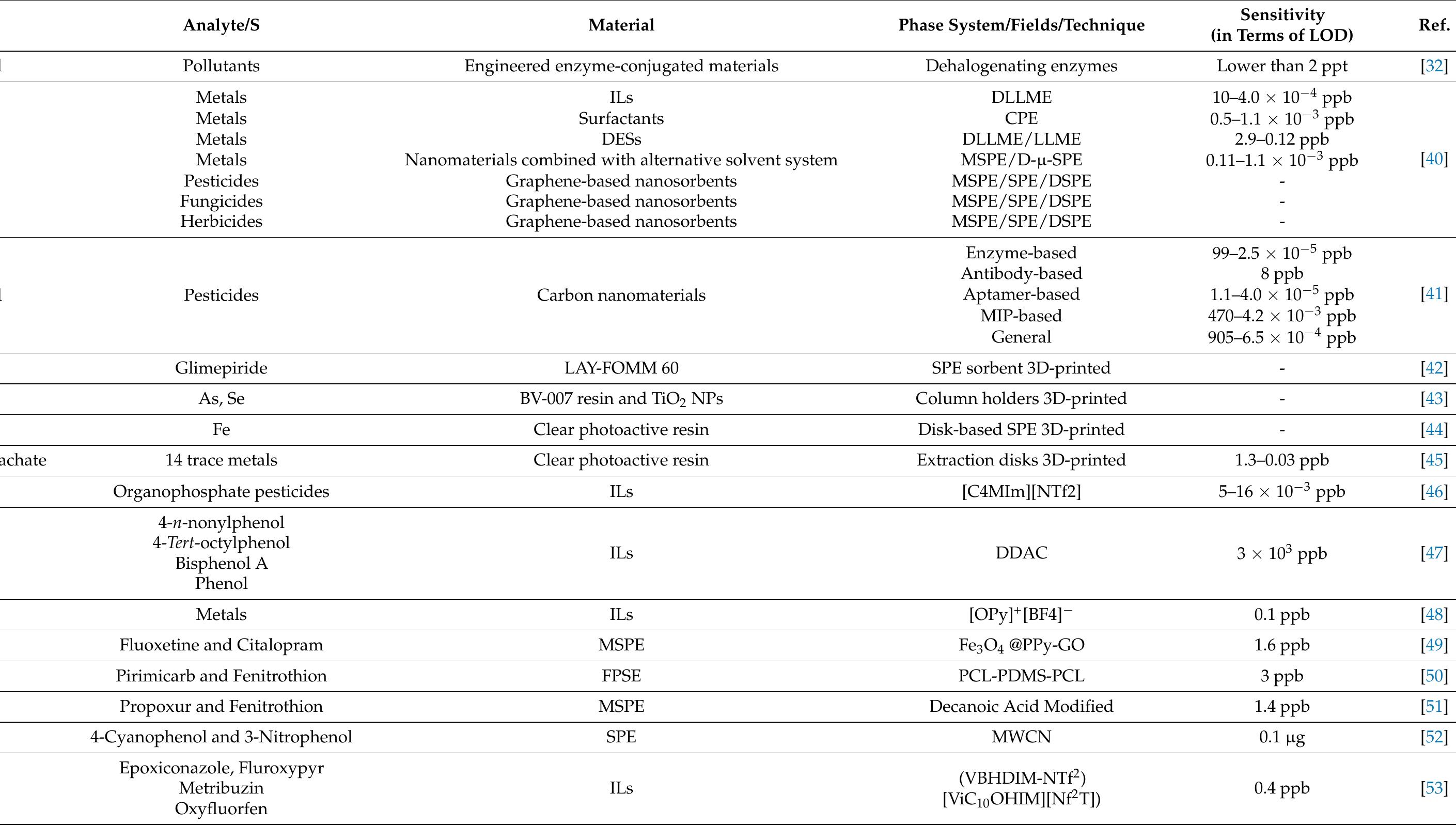
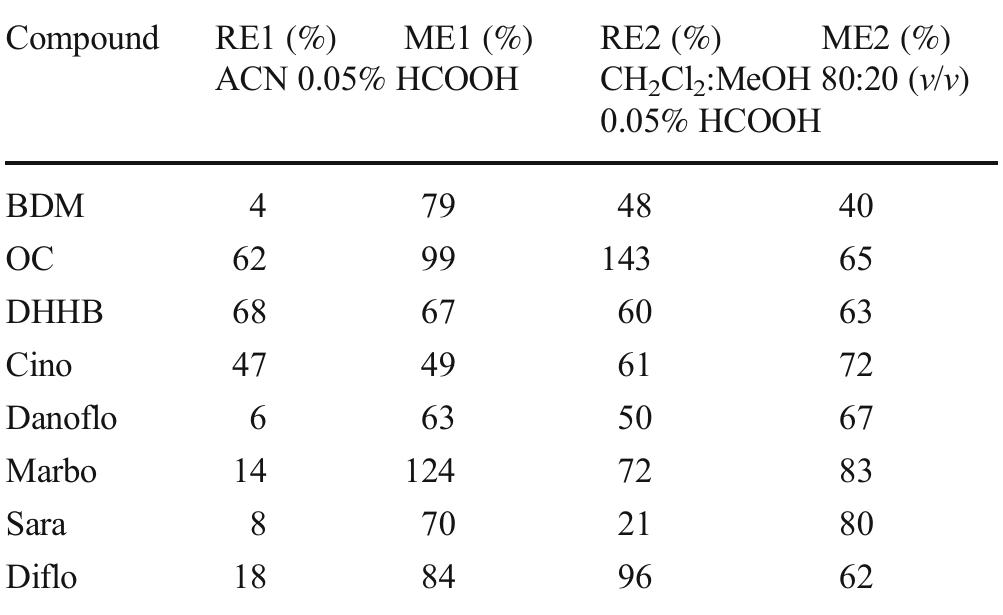
![Table 1 Recovery (RE) and matrix effect (ME) of the 50 selected com- pounds. Fifty milliliters of river water was extracted with 100 mg of mag- netic graphitized carbon black (mGCB). Both parameters are reported as percentage of the signal (see text). RE] and ME] are obtained by using 10 mL acetonitrile 0.05% formic acid as desorbing solvent; RE2 and ME2 are obtained by using 10 mL dichloromethane:methanol 80:20 (v/v) 0.05% formic acid as desorbing solvent](https://figures.academia-assets.com/100090839/table_001.jpg)
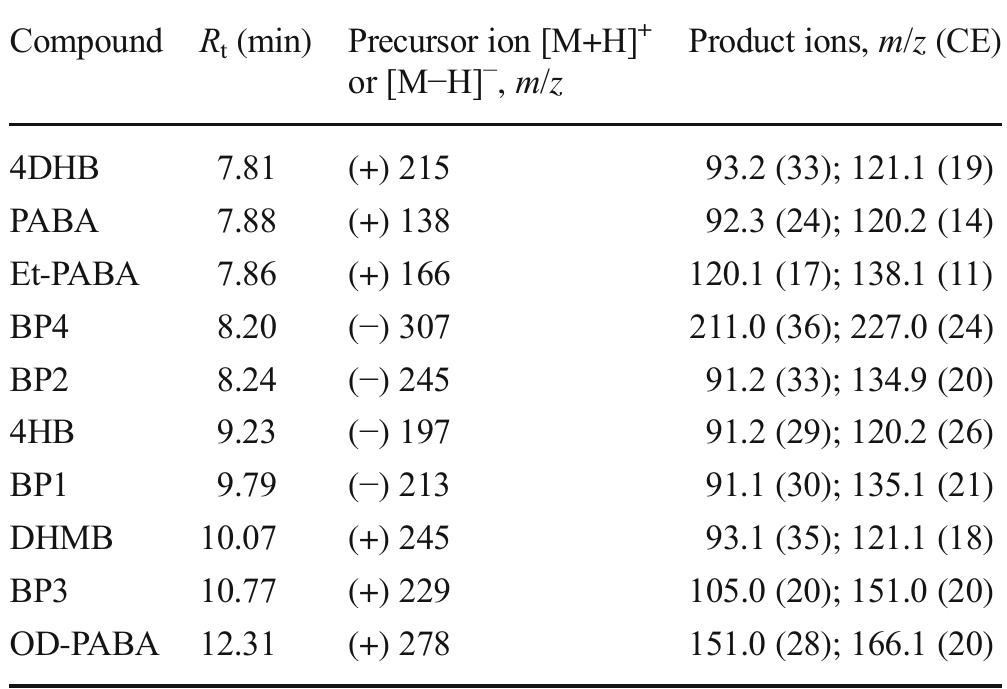
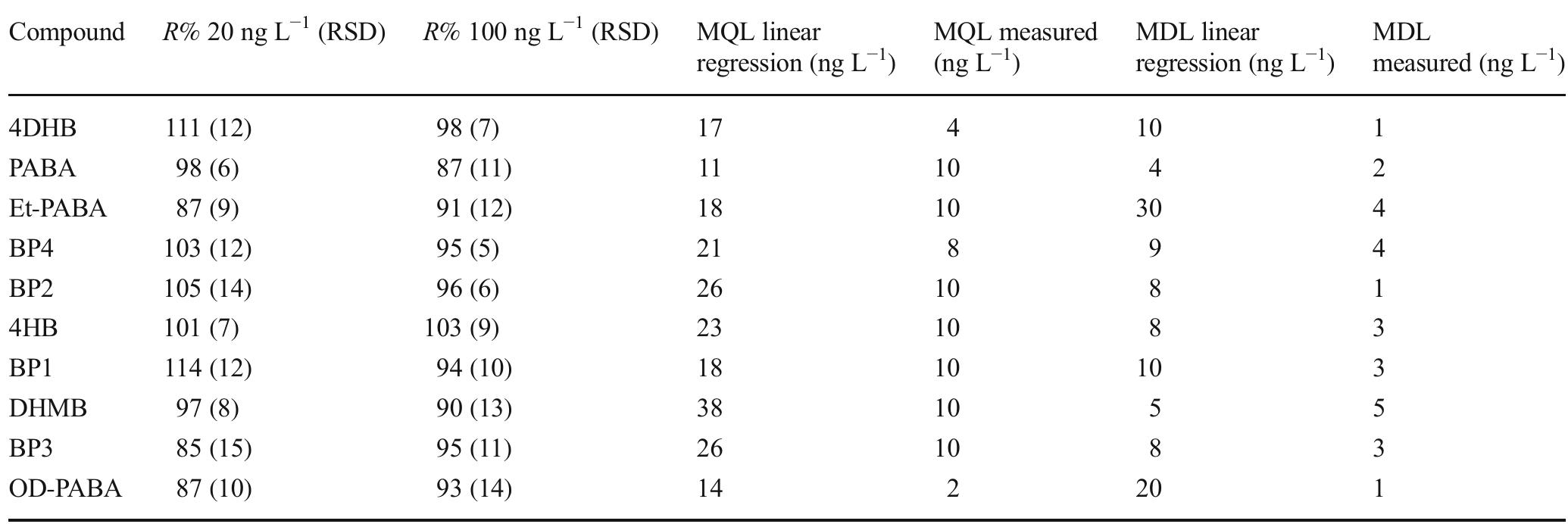
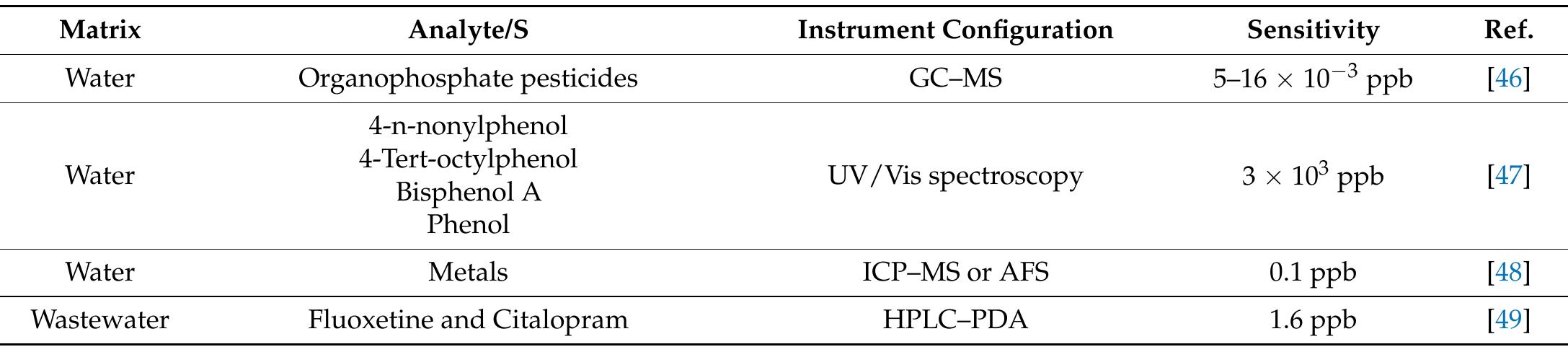
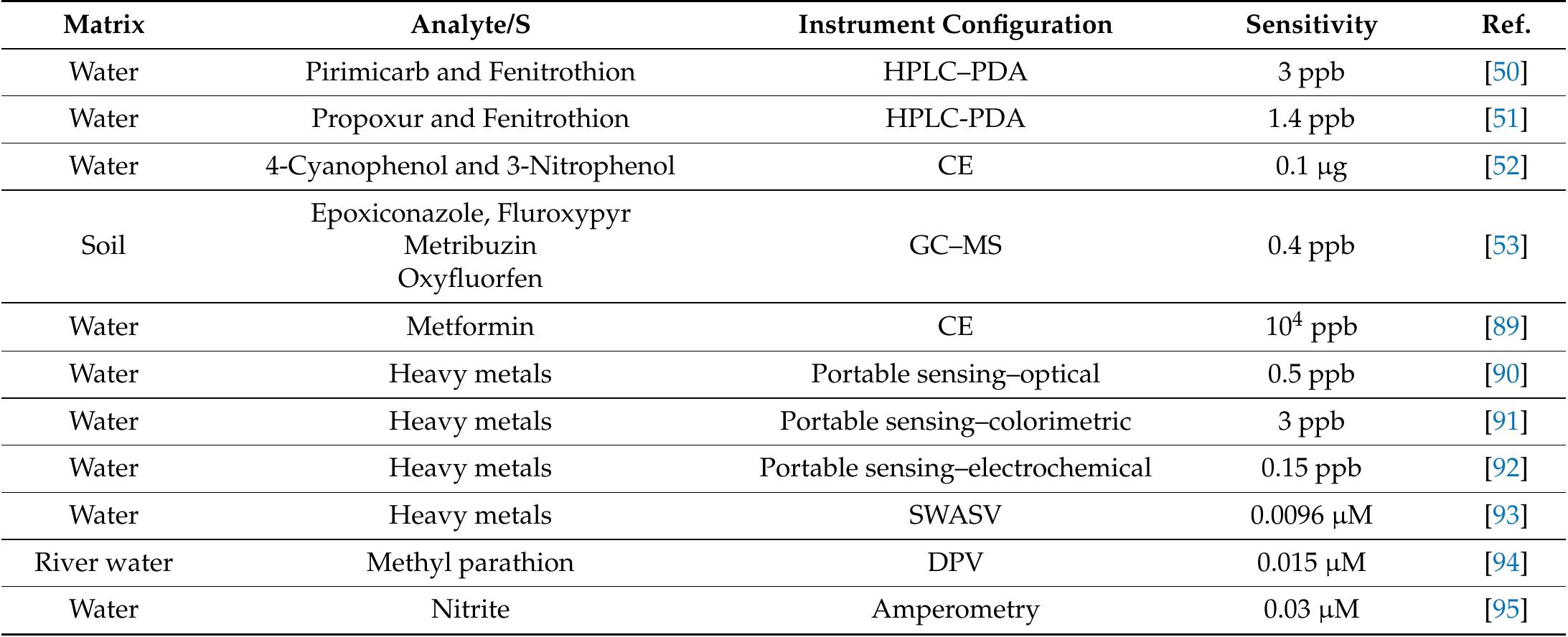
![Table 3 Extraction recovery (RE) and matrix effect (ME) assessed as reported in the text (extraction recovery, matrix effect, and process effi- ciency subsection of the “Materials and methods” section) at two con- centration levels, namely 100 and 10 ng L! attempt, we tried to use GCB not oxidized by nitric acid, as, differently from graphene and CNTs, GCB is wettable. However, in the presence of water, as reported previously [21], the chromene-like aromatic ethers react to give structures similar to benzpyrilium ion. This group strongly interacts with acidic compounds, which are recovered from water but are difficult to be desorbed from GCB, needing, for the most strongly retained sulfates, back elution from the conventional SPE cartridge with a mobile phase containing dichloromethane:methanol and a suit- able displacer, such as tetramethylammonium chloride. As it is possible to figure out from UV spectra and TGA measurements reported in the ESM, very likely the treatment with nitric acid only oxidizes the benzpyrilium function to carbonyl leaving un- changed or nearly unchanged the graphitic structure and the small amount of other oxygenated functions, such as phenol and quinones.](https://figures.academia-assets.com/100090839/table_005.jpg)