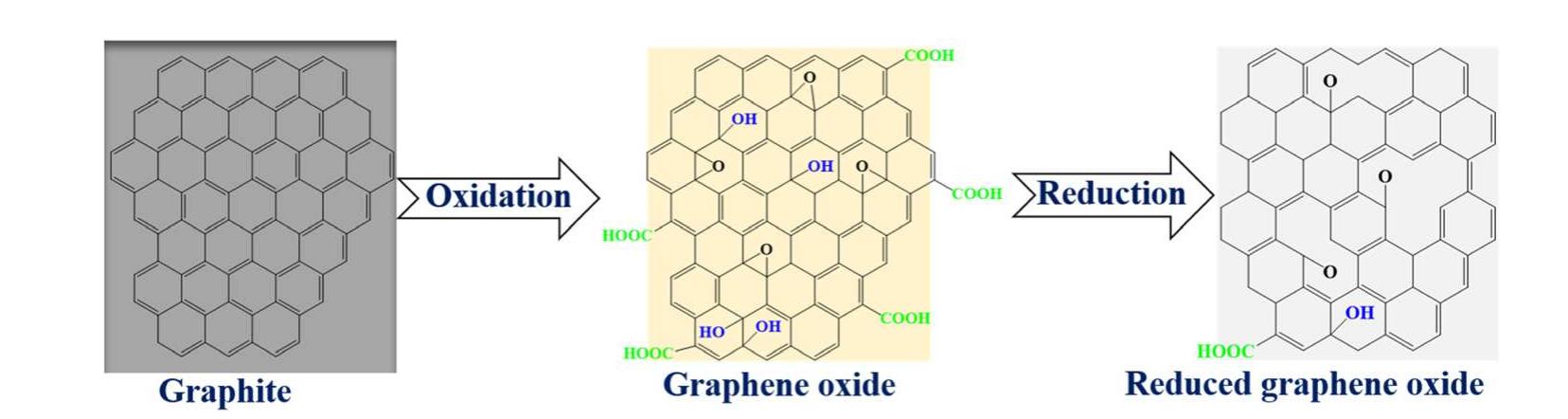
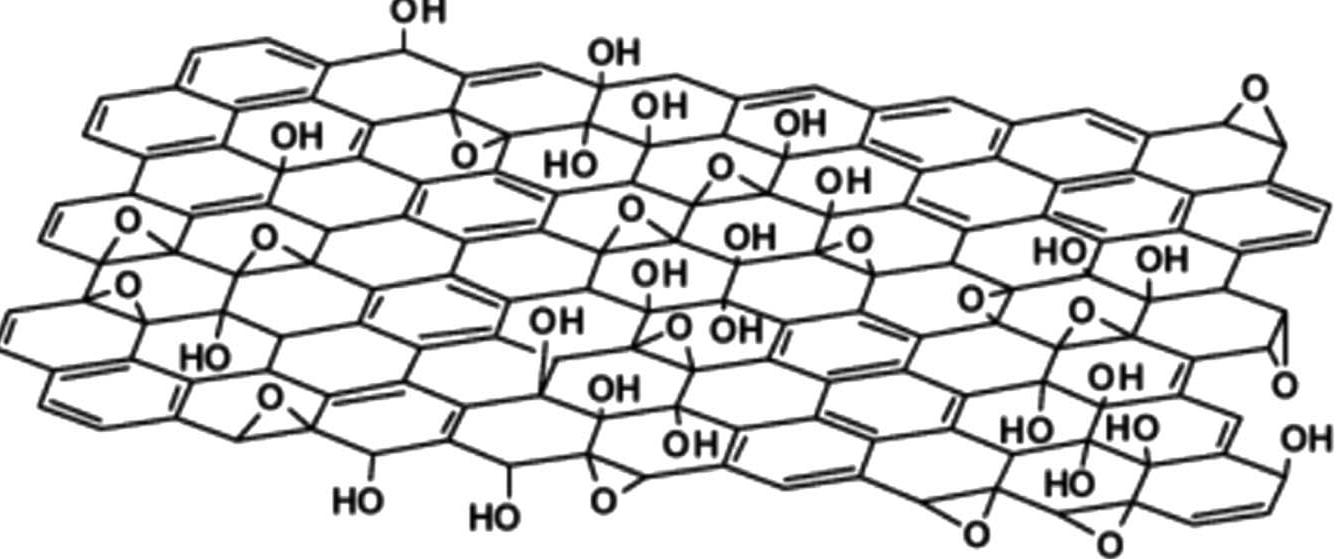
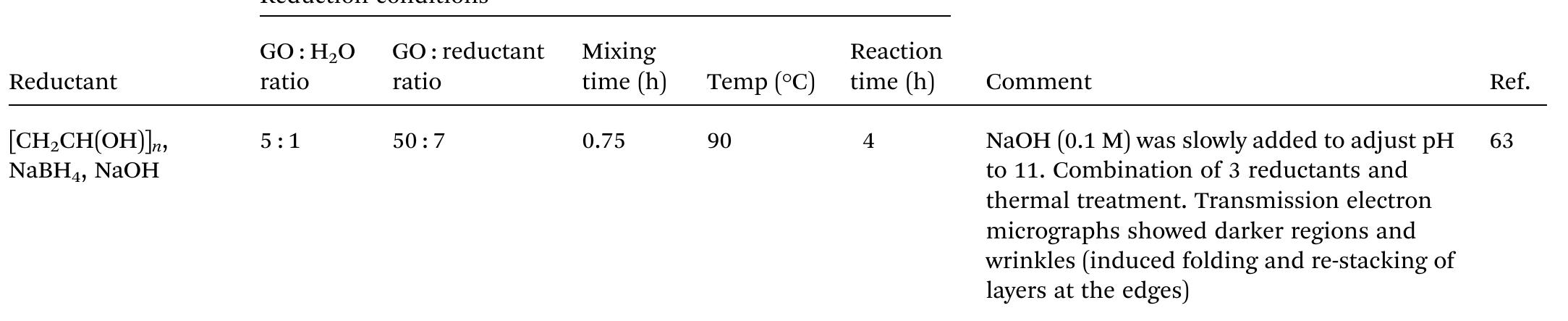
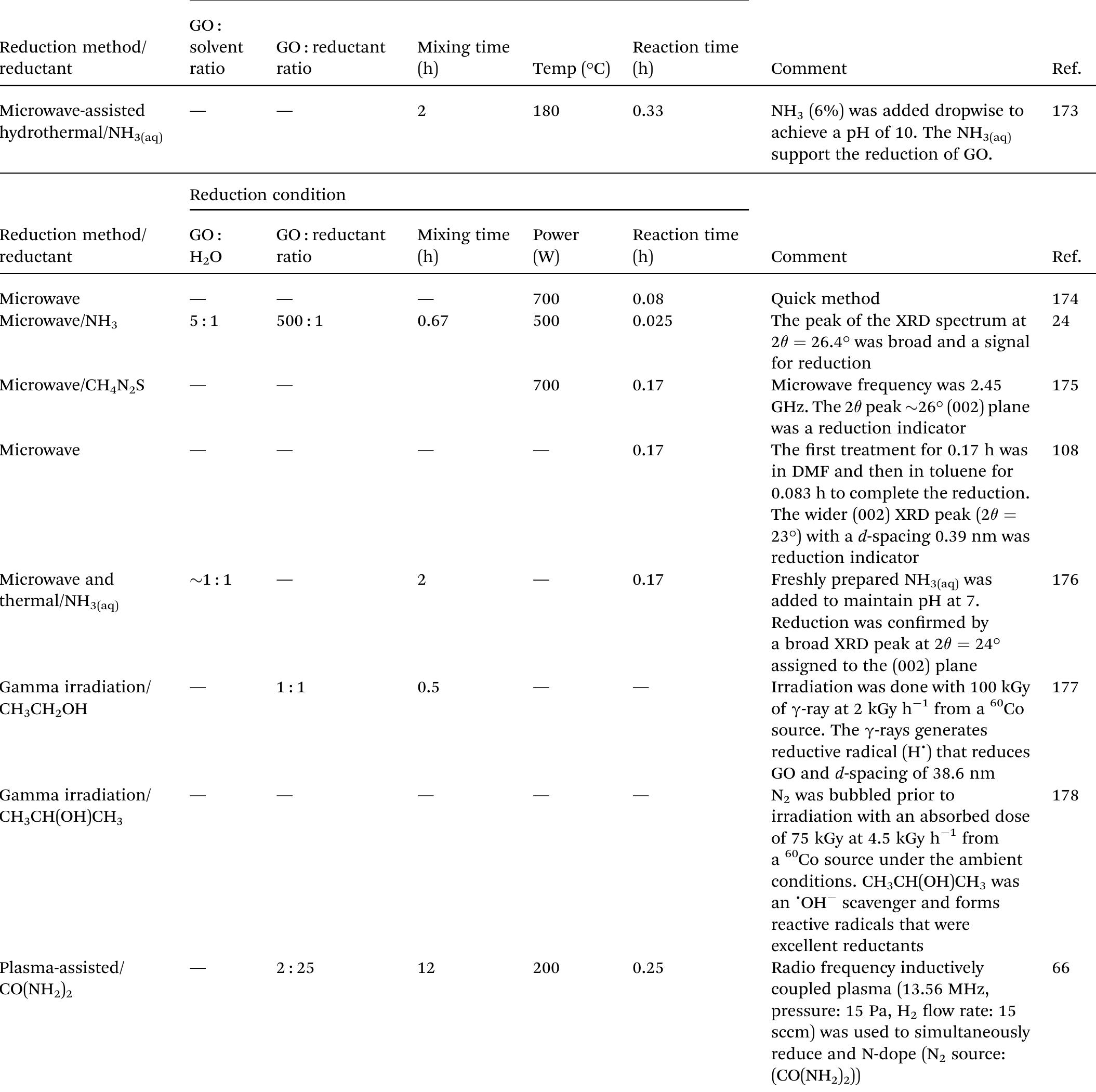
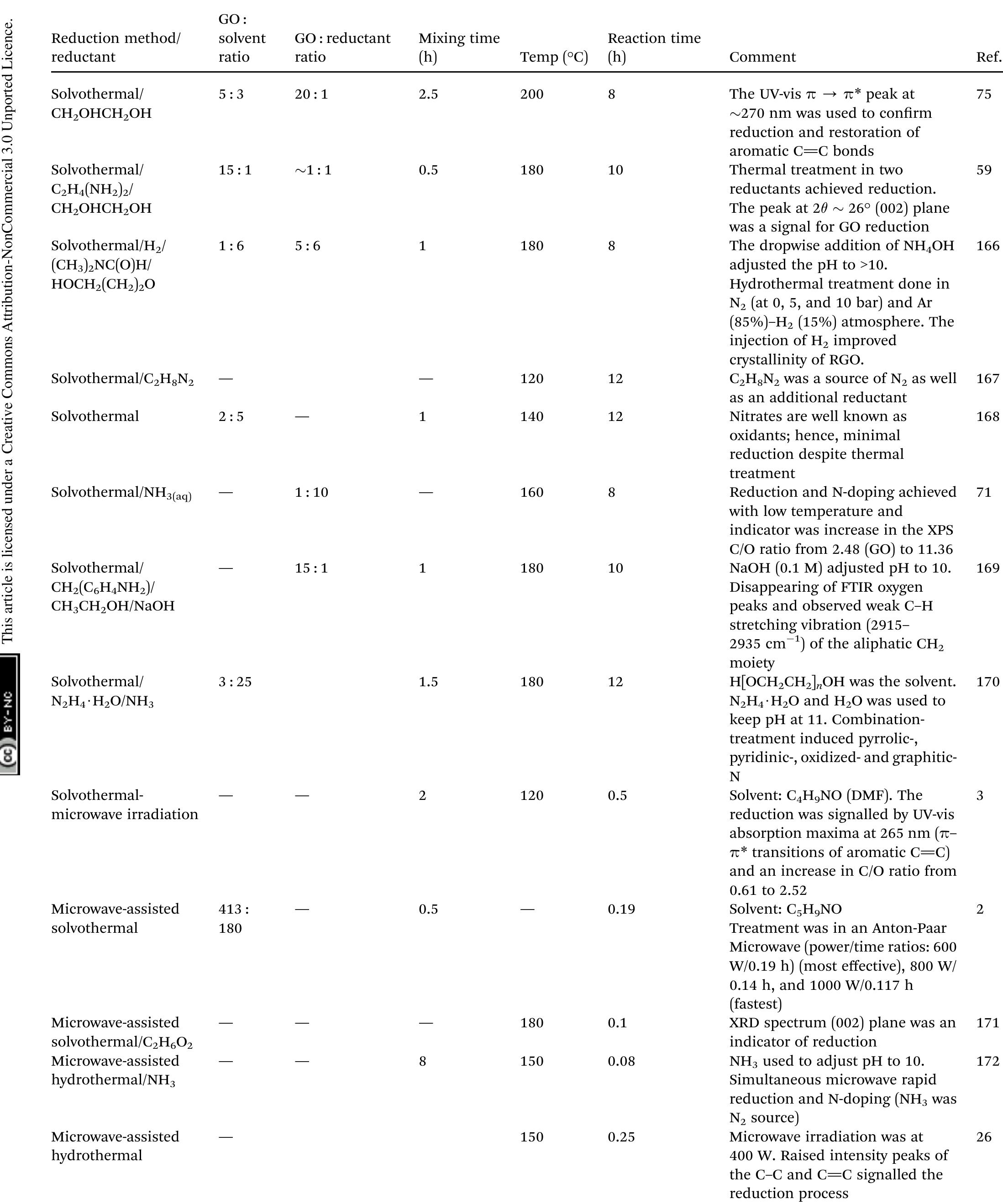
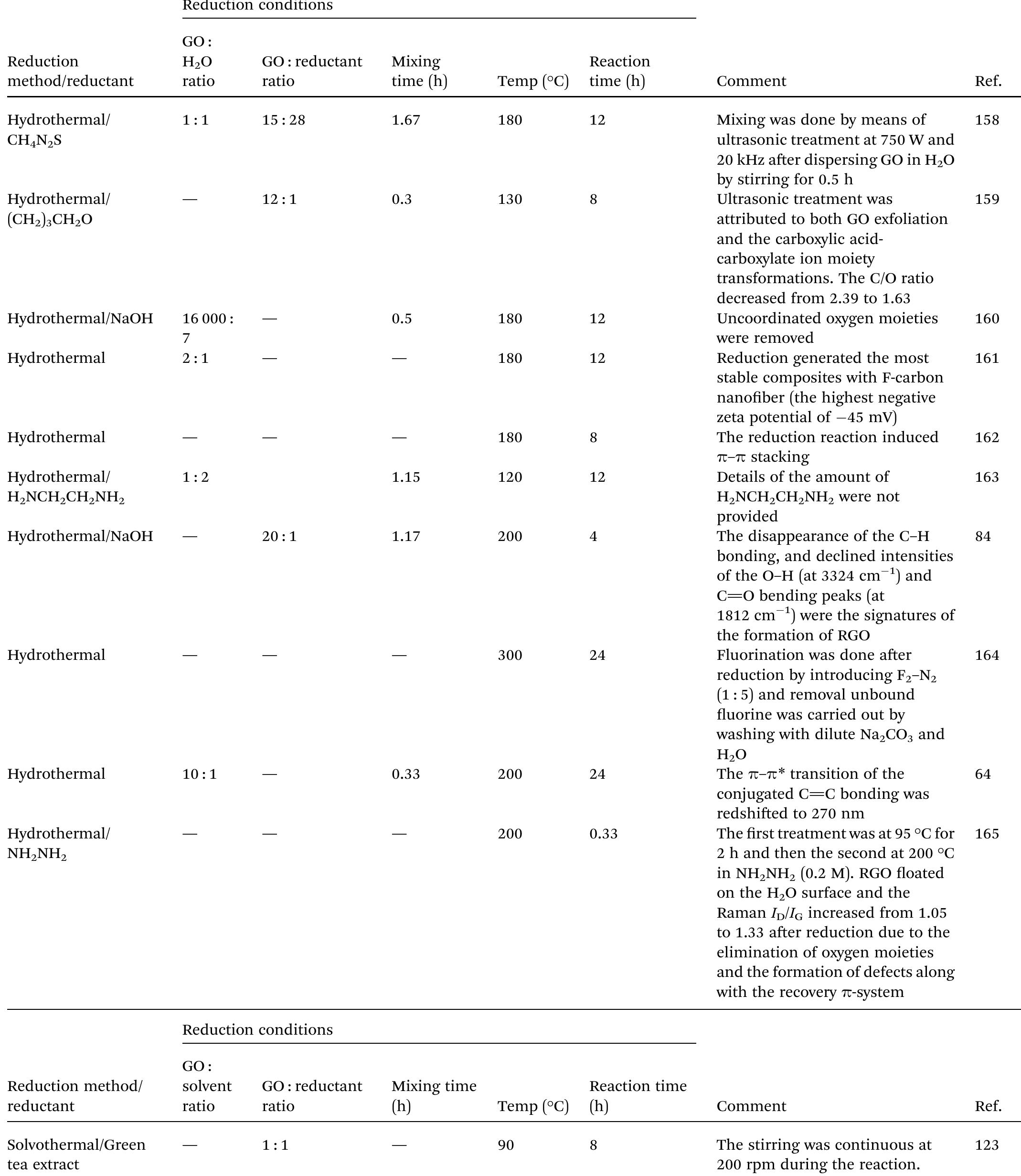
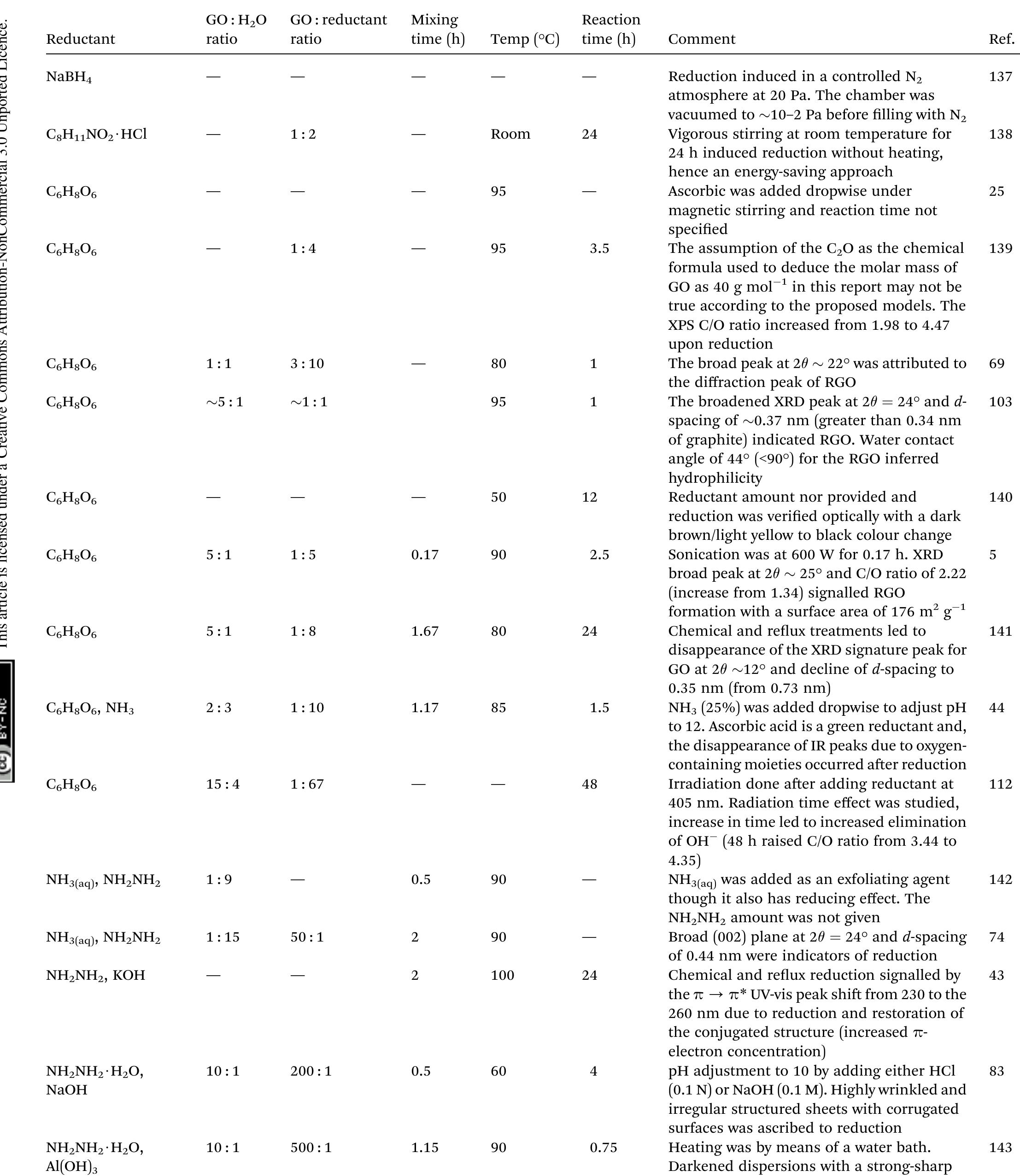
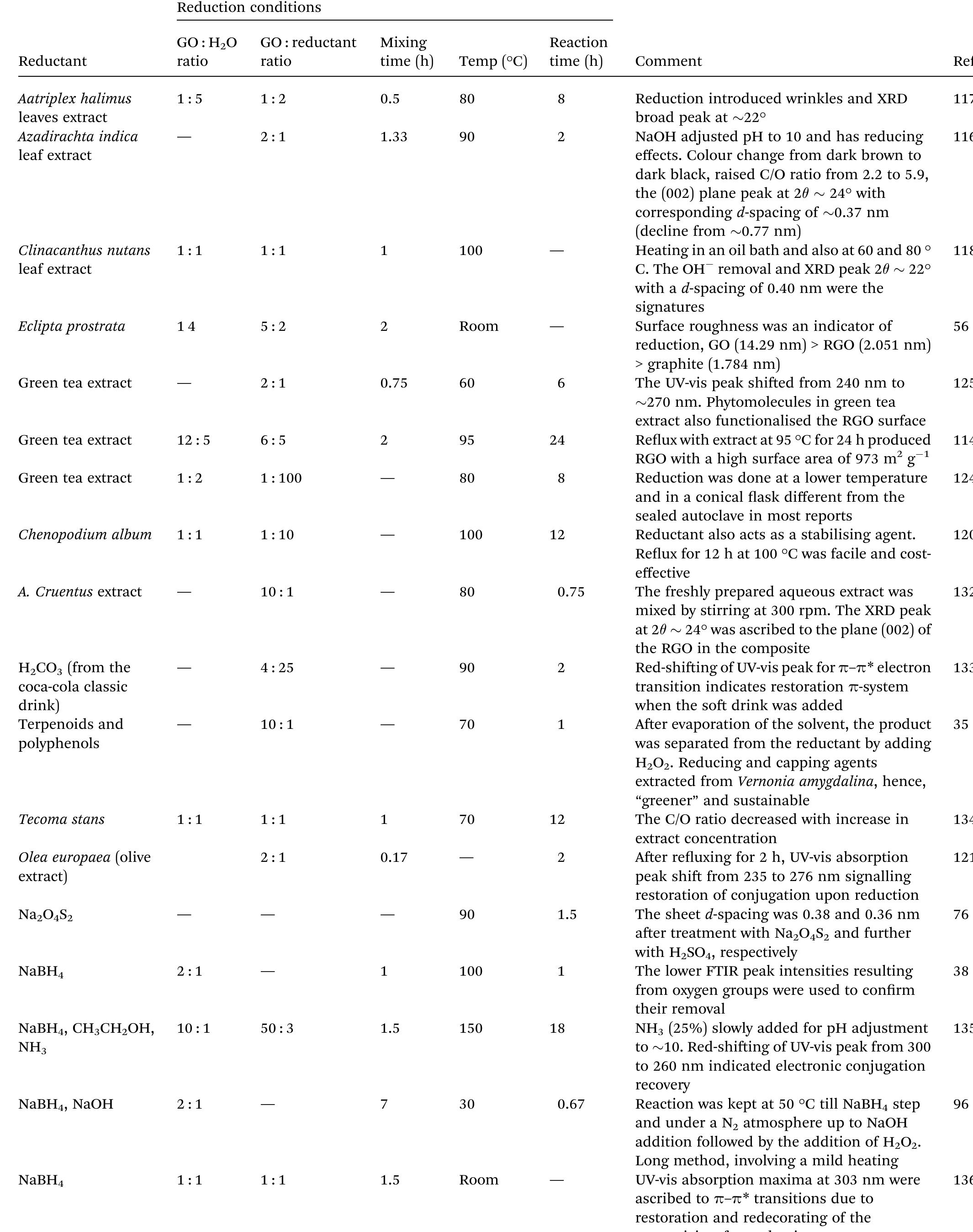
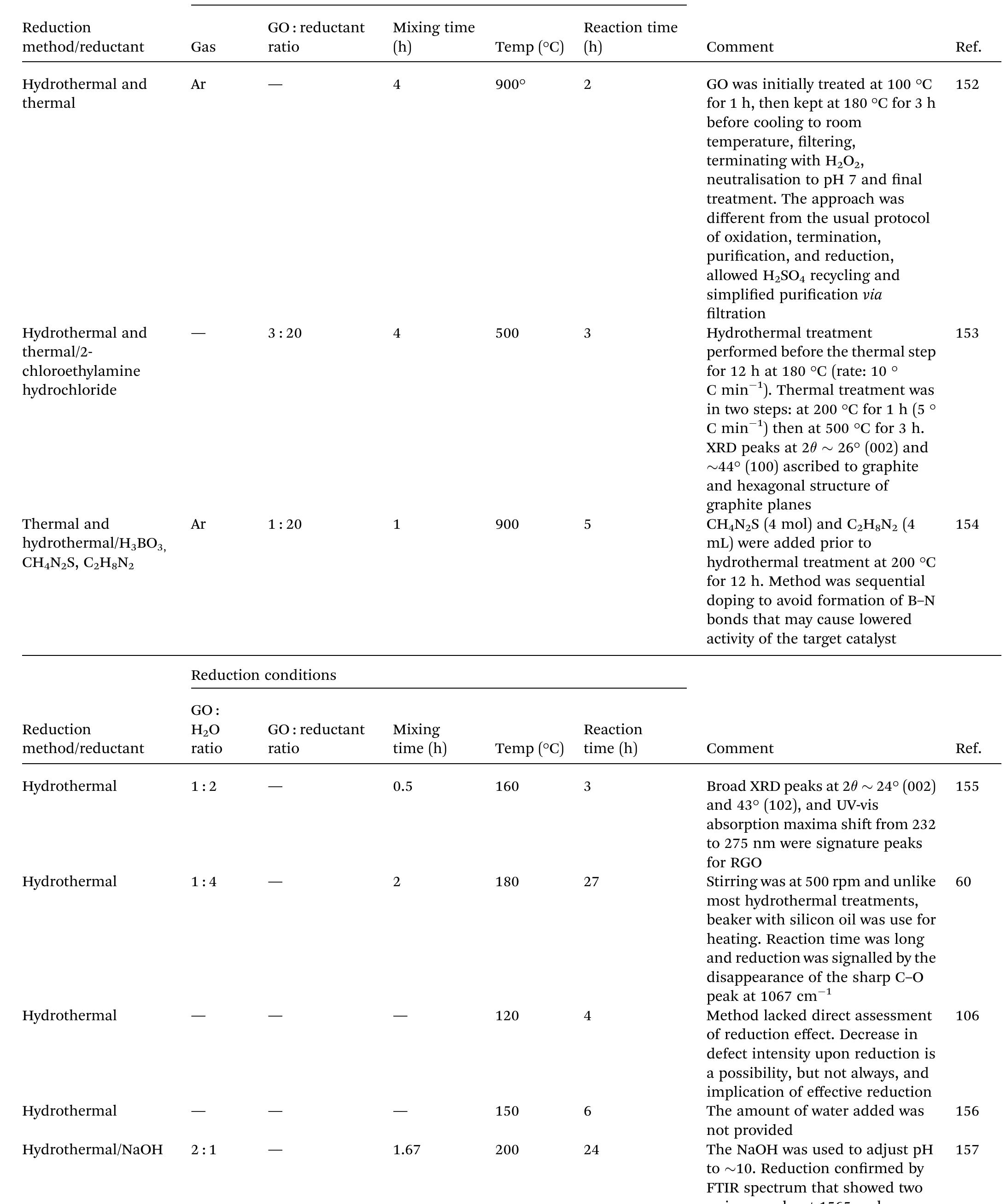
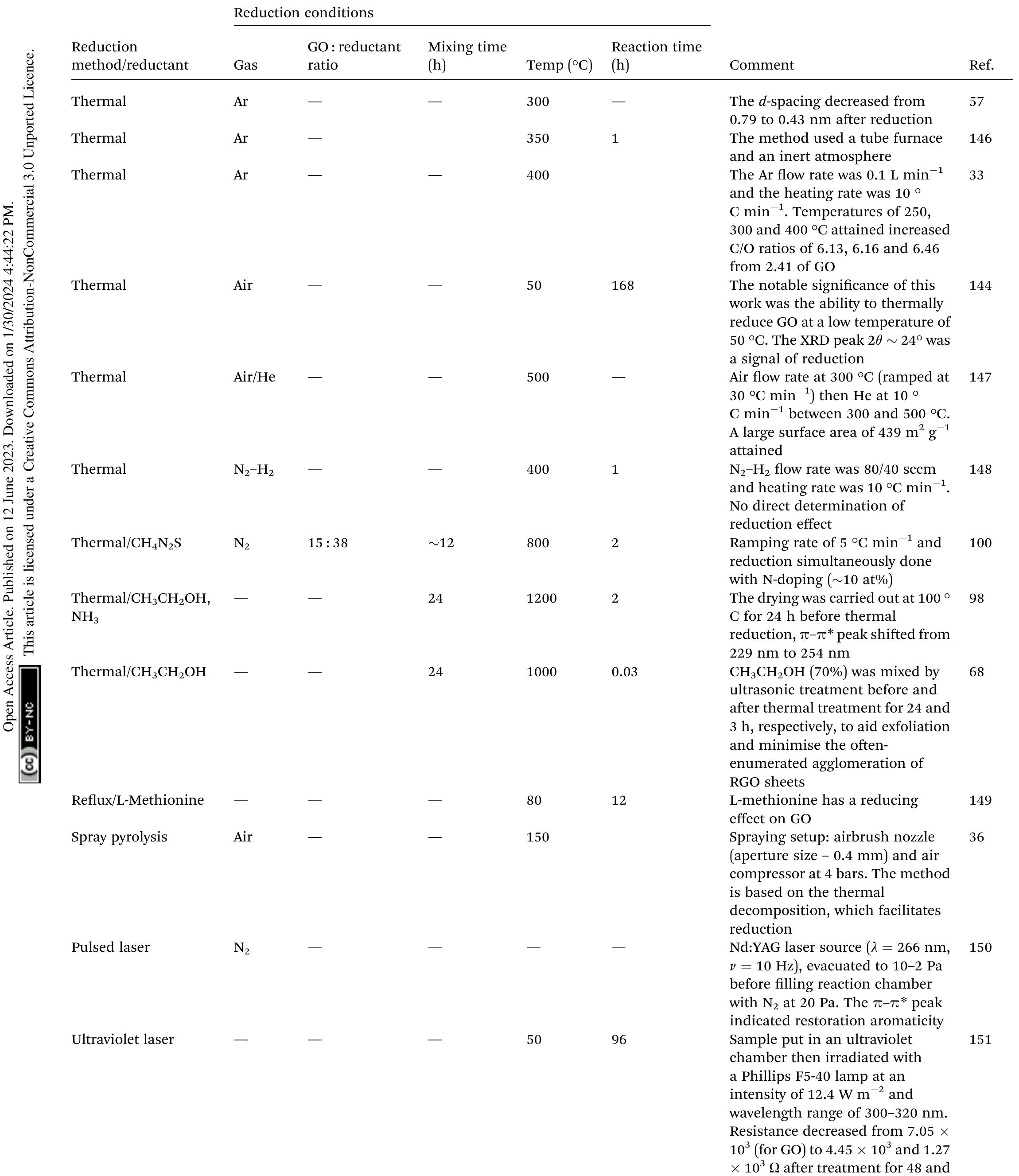
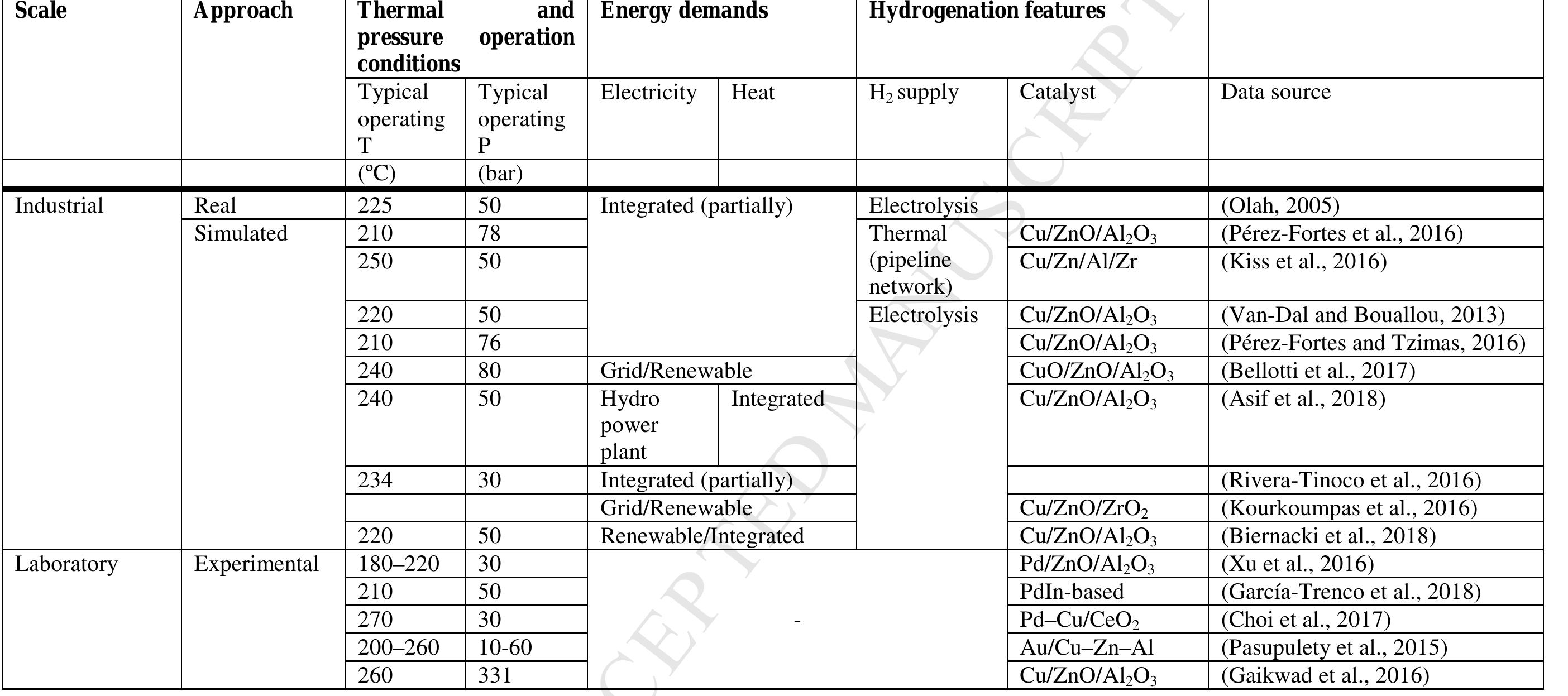
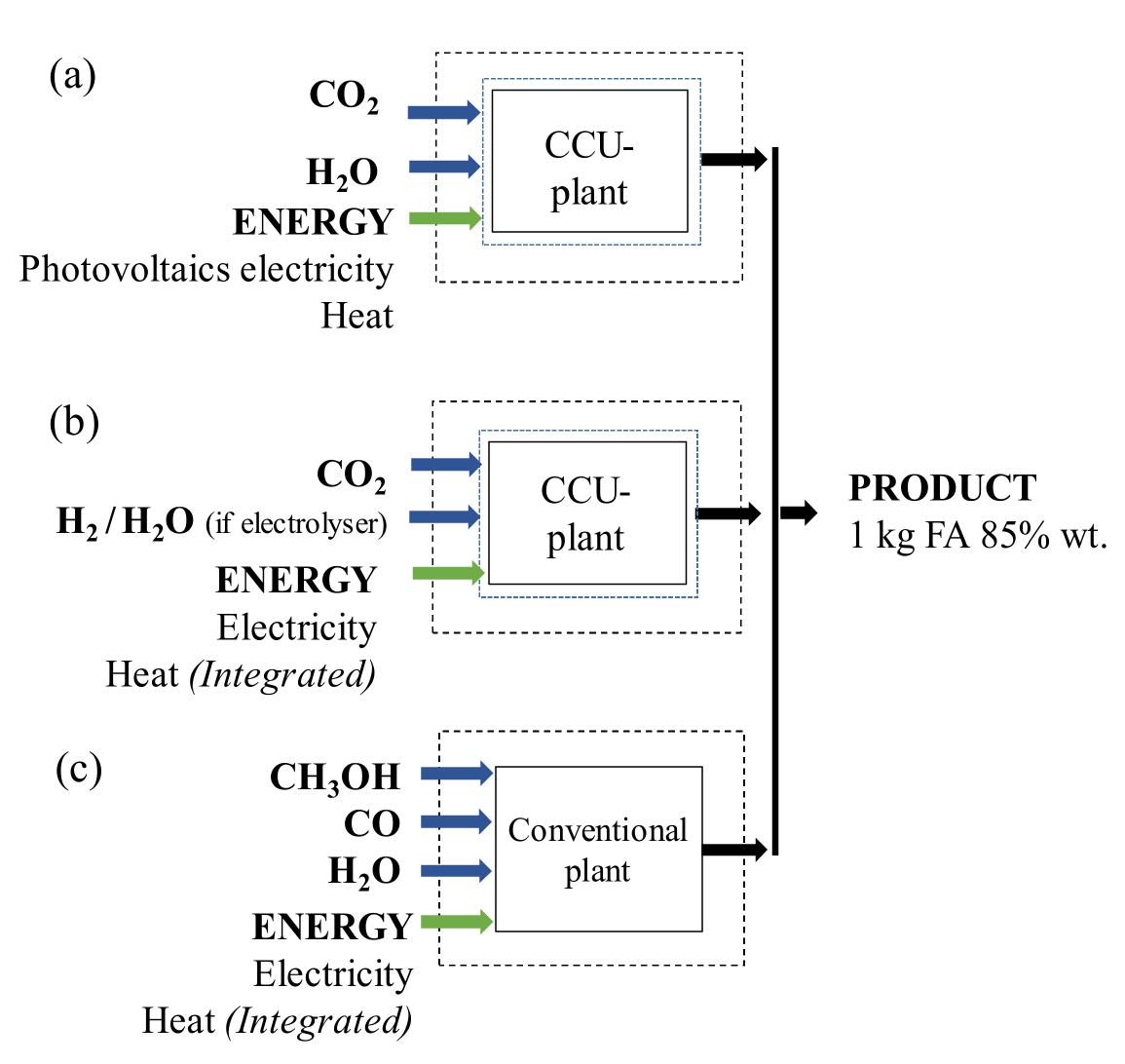
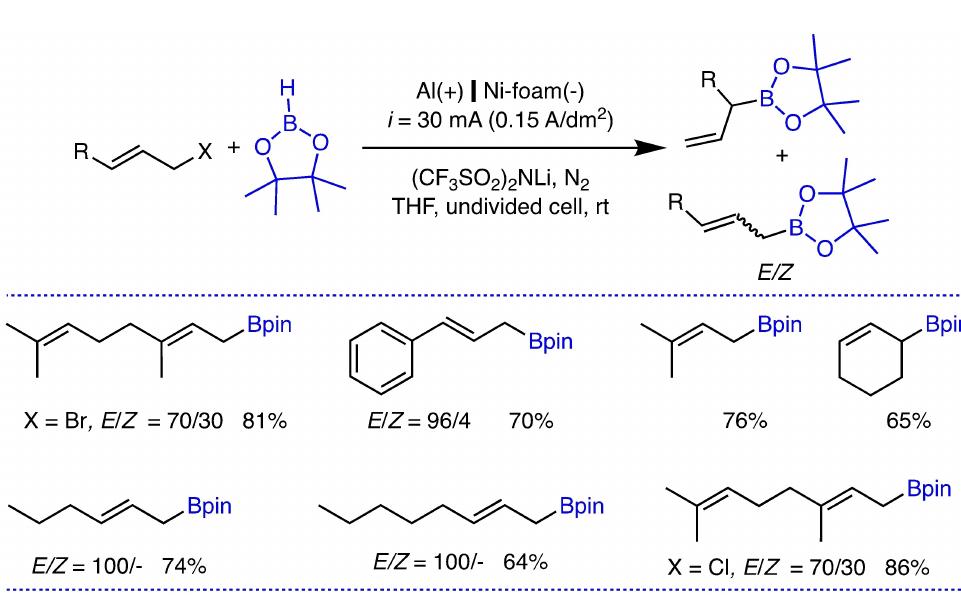
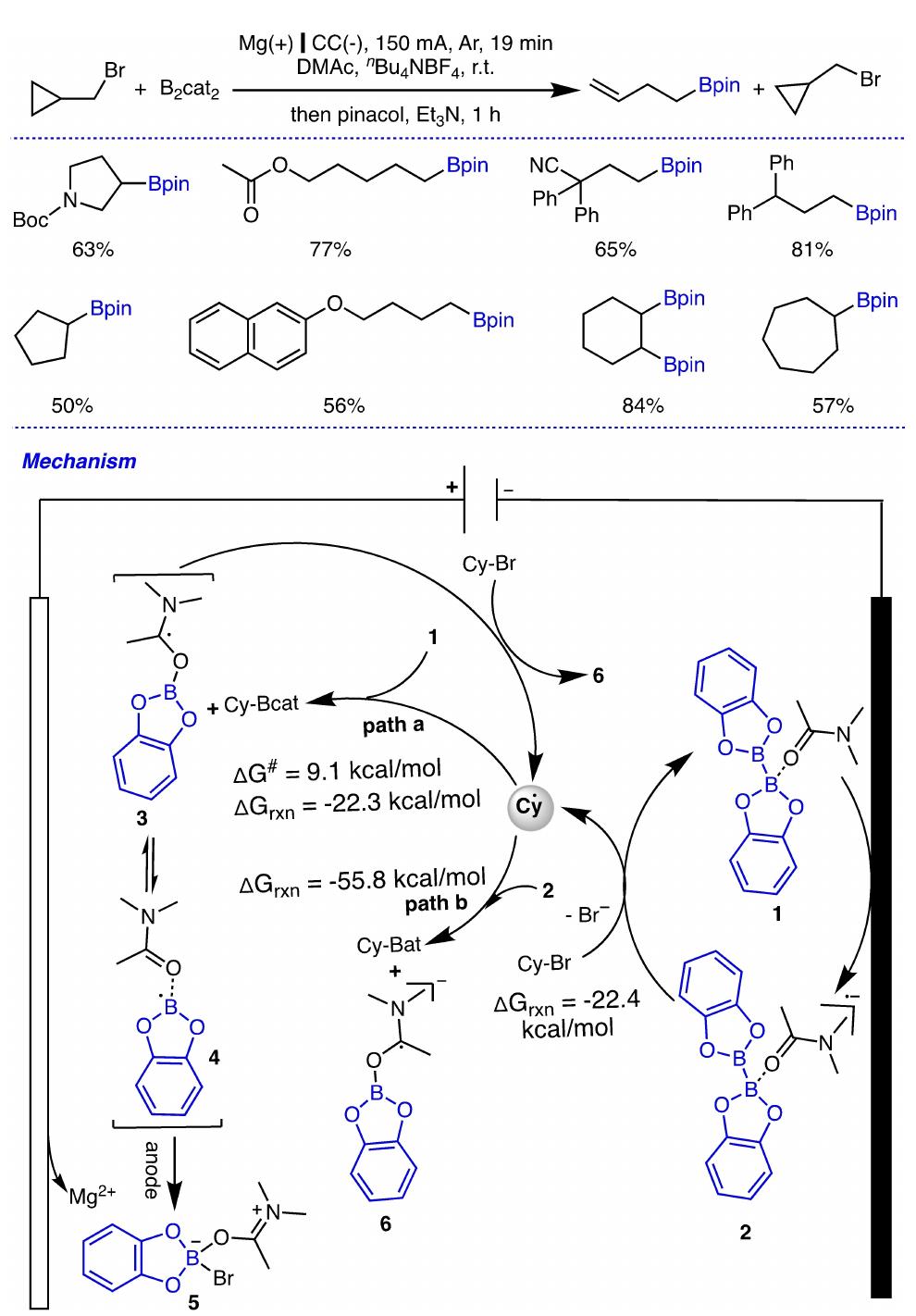
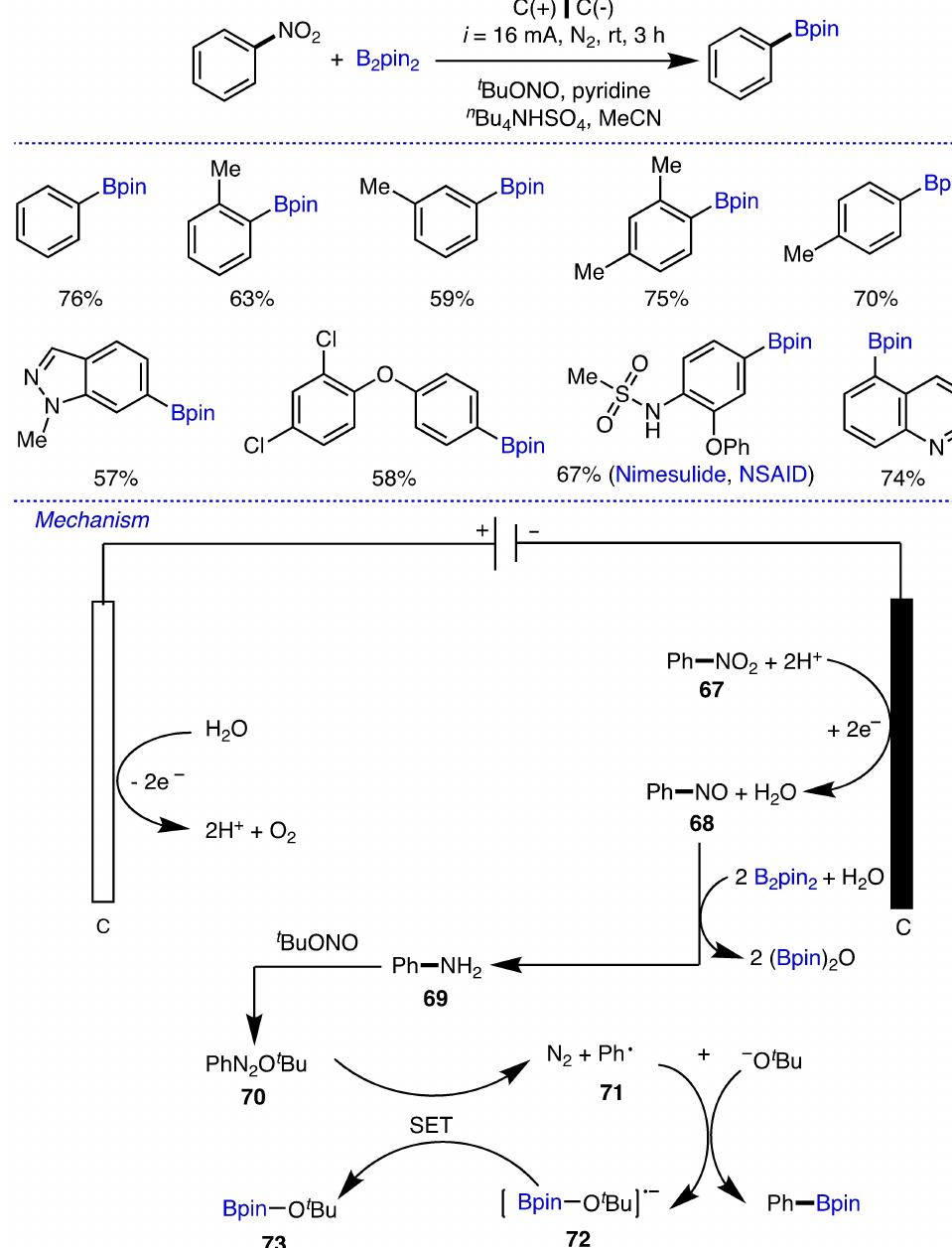
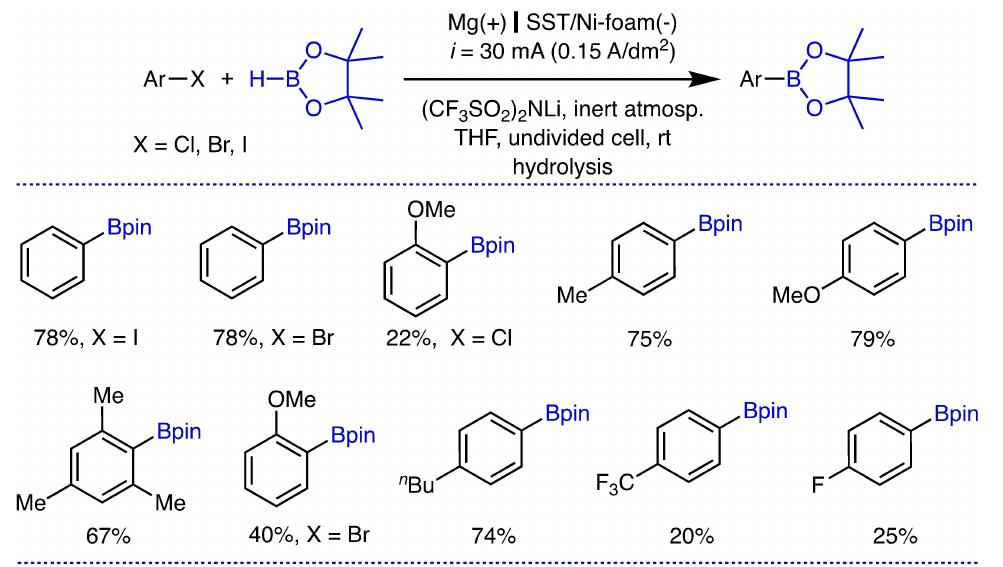
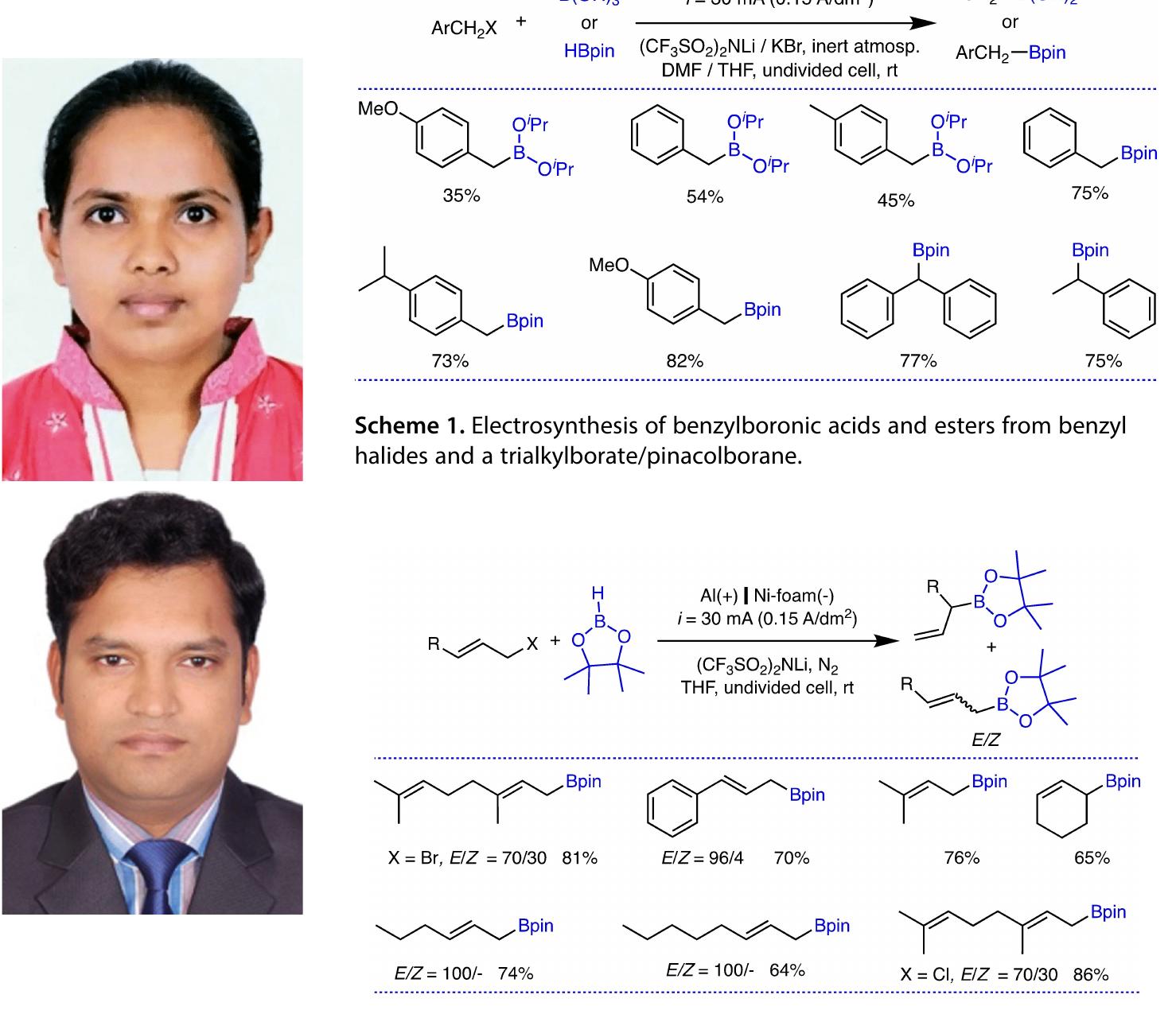
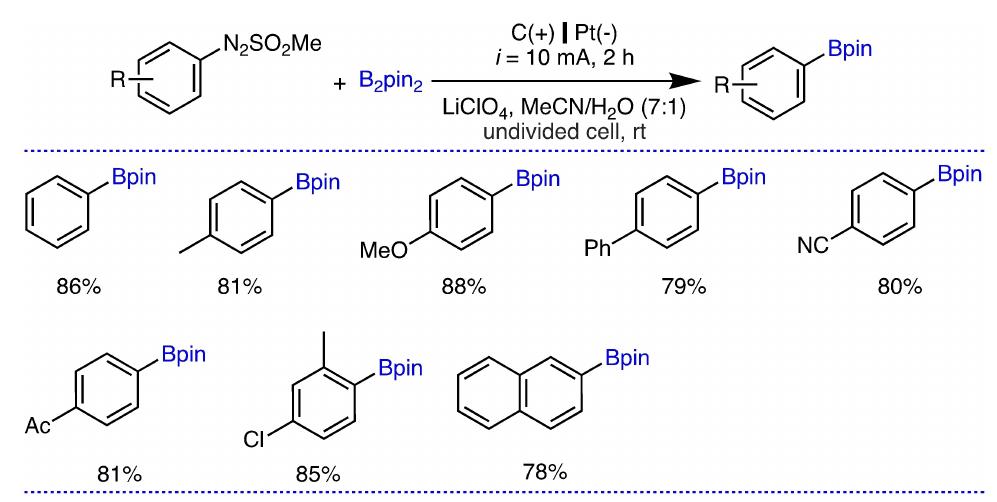
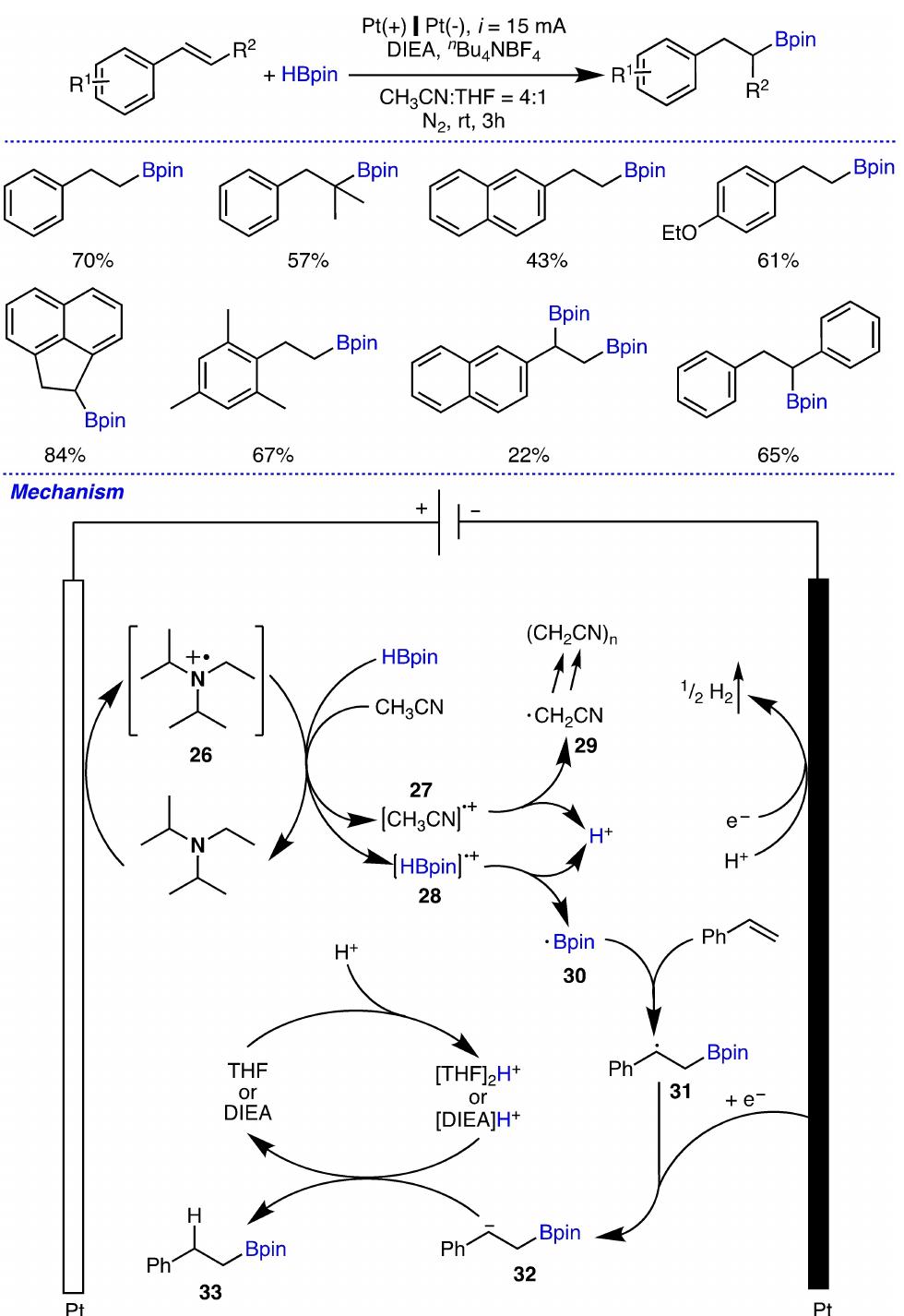
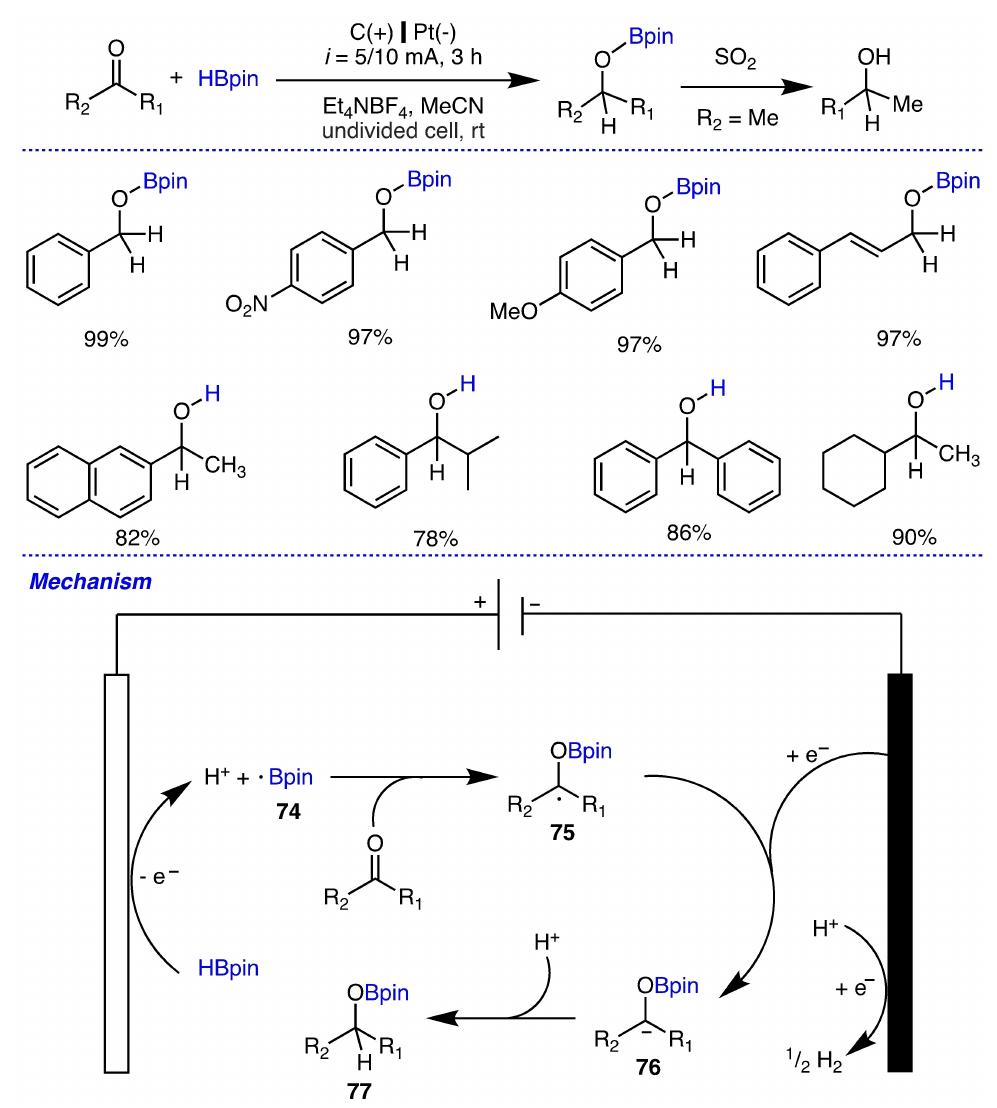
Key research themes
1. How can ionic liquids be optimized to enhance electrochemical CO2 reduction performance?
This research theme examines the role of ionic liquids (ILs) as electrolytes and co-catalysts in electrochemical CO2 reduction (CO2R). It investigates how ILs influence CO2 solubility, activation, product selectivity, current density, reaction overpotential, and inhibition of competitive reactions such as hydrogen evolution (HER). Understanding the structure-property relationships of ILs enables the tuning of electrolyte environments for improved CO2R efficiency and selectivity, which is critical for advancing sustainable carbon capture and utilization technologies.
2. What are the engineering and process challenges to scale up electrochemical CO2 reduction for industrial applications?
This theme focuses on the device design, reactor engineering, and process integration aspects critical for scaling electrochemical CO2 reduction to industrially relevant levels. It explores performance parameters such as current density, catalyst and cell stability, mass transport optimization, system configurations, and economic and durability considerations. Addressing these challenges is essential for realizing the commercial viability and sustainable implementation of CO2 electroreduction.
3. How can electrochemically derived catalysts improve selectivity and efficiency for multi-electron reduction reactions, including nitrogen and carbon dioxide reduction?
This theme encompasses the design and mechanistic understanding of catalysts derived via electrochemical methods to enhance multi-electron transfer reactions such as nitrogen reduction to ammonia and CO2 reduction to hydrocarbons and C2+ products. It explores material structures, redox behaviors, catalytic pathways, and stability improvements to overcome challenges like competing reactions and unfavorable kinetics, providing actionable insights for catalyst development.



![and they have the absence of by-products (i.e. O2 and H2) being ER the only influence in the C] considered. Agr ideal) Azr-conv-1 and Age-pr-1 are taking into account the avoided amount of CO,](https://figures.academia-assets.com/108205064/figure_004.jpg)
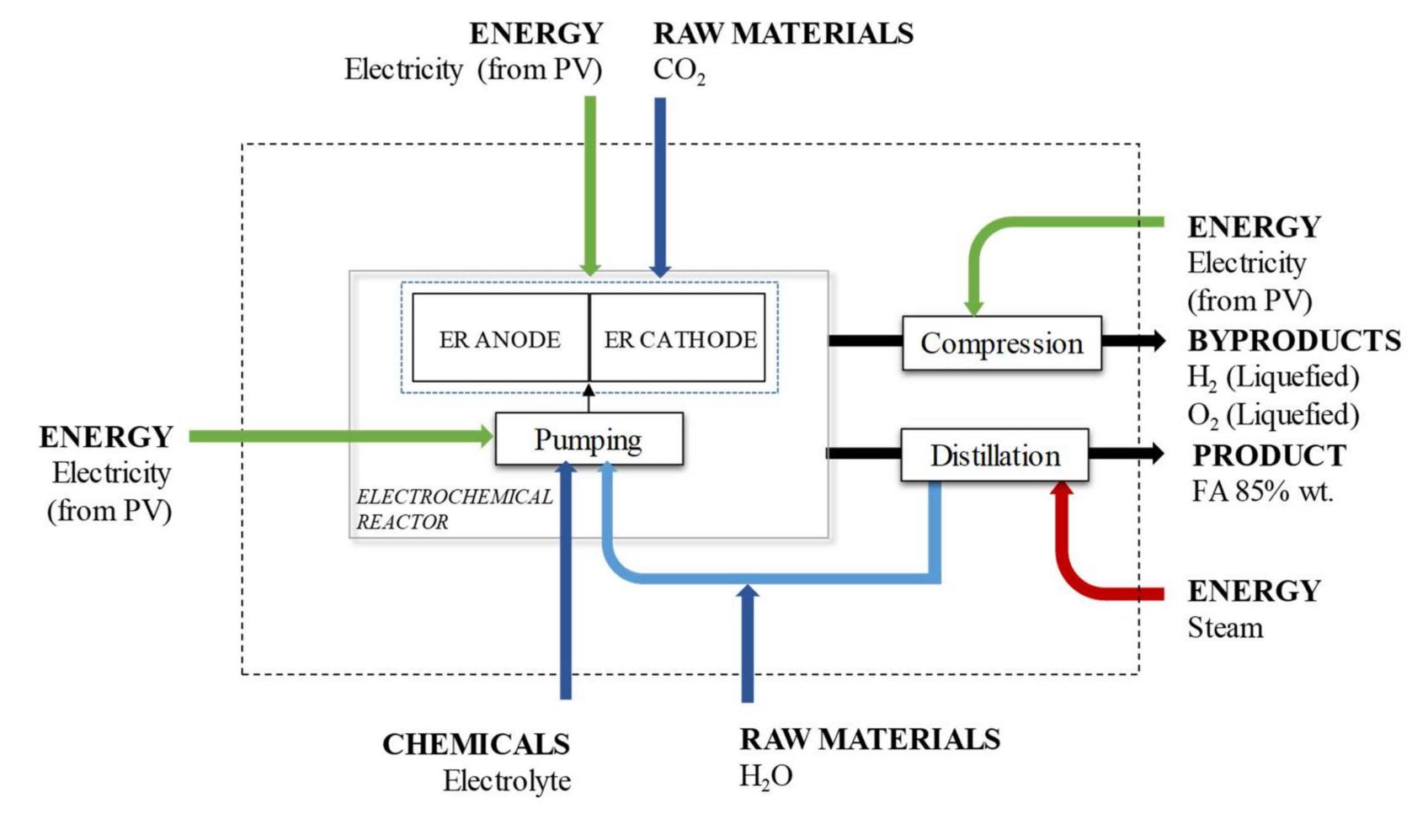
![Table 1. Main and side reactions. A purification process in the form of a distillation is required to increase the FA concentration at the outlet of ER to the accepted commercial concentration value at 85% wt. Therefore, cooling and heating are needed. The distillation process was simulated in a previous study carried out by the authors [26]. The simulation considers only presence of water (obtained as a head product) and FA obtained as the bottom product) at the required concentration. Distilled water is recirculated and reinjected to the inlet freshwater stream to the cathode. Then, net water consumption is the difference between water in the inlet stream and water that is recirculated. The pure O2 produced as a secondary product, mainly by the anodic reaction, is separated, liquefied, and recovered, as well as Hz, which is produced by the competing reaction, and it is assumed to be pure. Tian | xx 4 eens kt aed Sil peal herewd het flak wwe ballefeeeededive wrenecqeed tex Edn sores Se Hise weark:](https://figures.academia-assets.com/108205035/table_001.jpg)
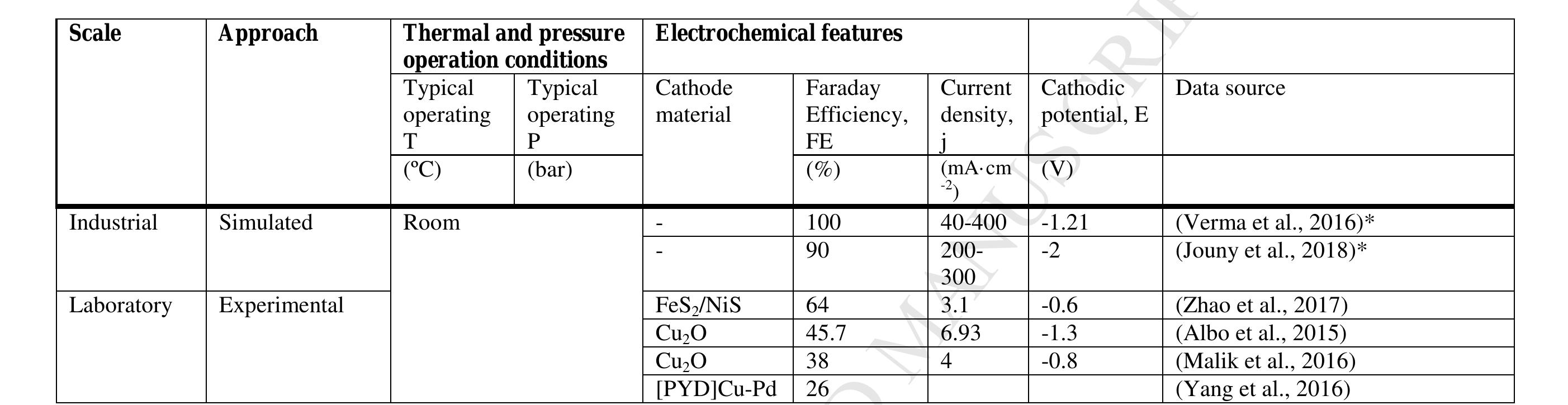
![The detailed LCIs for required materials and energy corresponding to each set of experimenta data were calculated by means of a sequence of energy and mass balances. More details can be found in the supplementary data published in a previous study [26]. Mass balances were performed in each unit, ensuring that there were no mass losses for any element. This methodology ensures that each CCU-ER alternative is treated equally; thus, the results are directly comparable. The electricity needed in the process, such as the electricity demanded in the reduction of CO> to FA, the electricity for the pumps within the reactor cell, and the electricity needed for liquefying both O2 and H2, was assumed to come from the surplus production of renewable energy in the form of photovoltaic solar energy (PV). Alternatively, wind power could be also considered. Surplus energy derives from avoiding curtailment of PV solar or wind farms due to the possibility of injecting excess power in the CCU facility. The periods in which the electricity generation exceeds the demand create a problem of “surplus production”, which can help at the ER of CO,. This selection determines the CF of each unit of electricity used for the process, which in this case was 0.03 kg-kWh~! [31]. The use of other high-carbon source for the electricity consumption will make no sense for ER purposes [26]. Pumping needs are derived from the need to overcome the pressure drop inside the reactors. Heat, as steam, is a utility used in the distillation unit. The source for this steam is natural gas and the CF per unit of steam used is 0.277 kg-kg~!, which was obtained from Ecoinvent data [27]. Table 2. Experimental conditions of the selected references/scenarios for the carbon footprint of CO to formic acid.](https://figures.academia-assets.com/108205035/table_002.jpg)
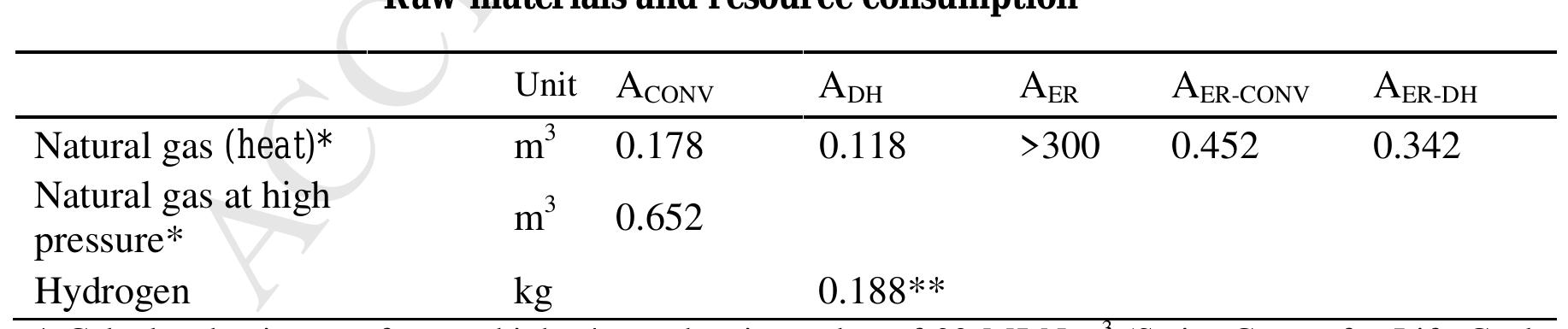
![Table 3. LCI for the ER of CO2 according to the three selected experimental data references (A1.1, A1.2, and A1.3), the conventional commercial process sourced from Ecoinvent [27] (Acony), and an alternative CCU process based on the catalytic conversion of CO2 and Hp [7] (Ajcr). The functional unit is 1 kg FA. It can be seen in Table 3 that values of ER energy and steam are expected to be the main resources ntributing to the overall CF. The electricity consumption by ER per mass unit of FA ranges between 59 kWh-kg~! and 11.93 kWh-kg~!, with A1.2 alternative having the best conditions in terms of 2ctric consumption. The differences are mainly due to the combinations of reaction rates and cell tentials at a certain current density. A1.2 has the lowest flow rate (for pumping) and the highest radaic Efficiency. These values are well over the minimum theoretical electricity consumption of "2 kWh-ke! (originally 1.803 MWh.ton~! when related to CO as reported in [17]). A1.1 alternative hibits the highest consumption of steam by unit of mass of FA 401 MJ-kg~!, as the FA concentration](https://figures.academia-assets.com/108205035/table_003.jpg)
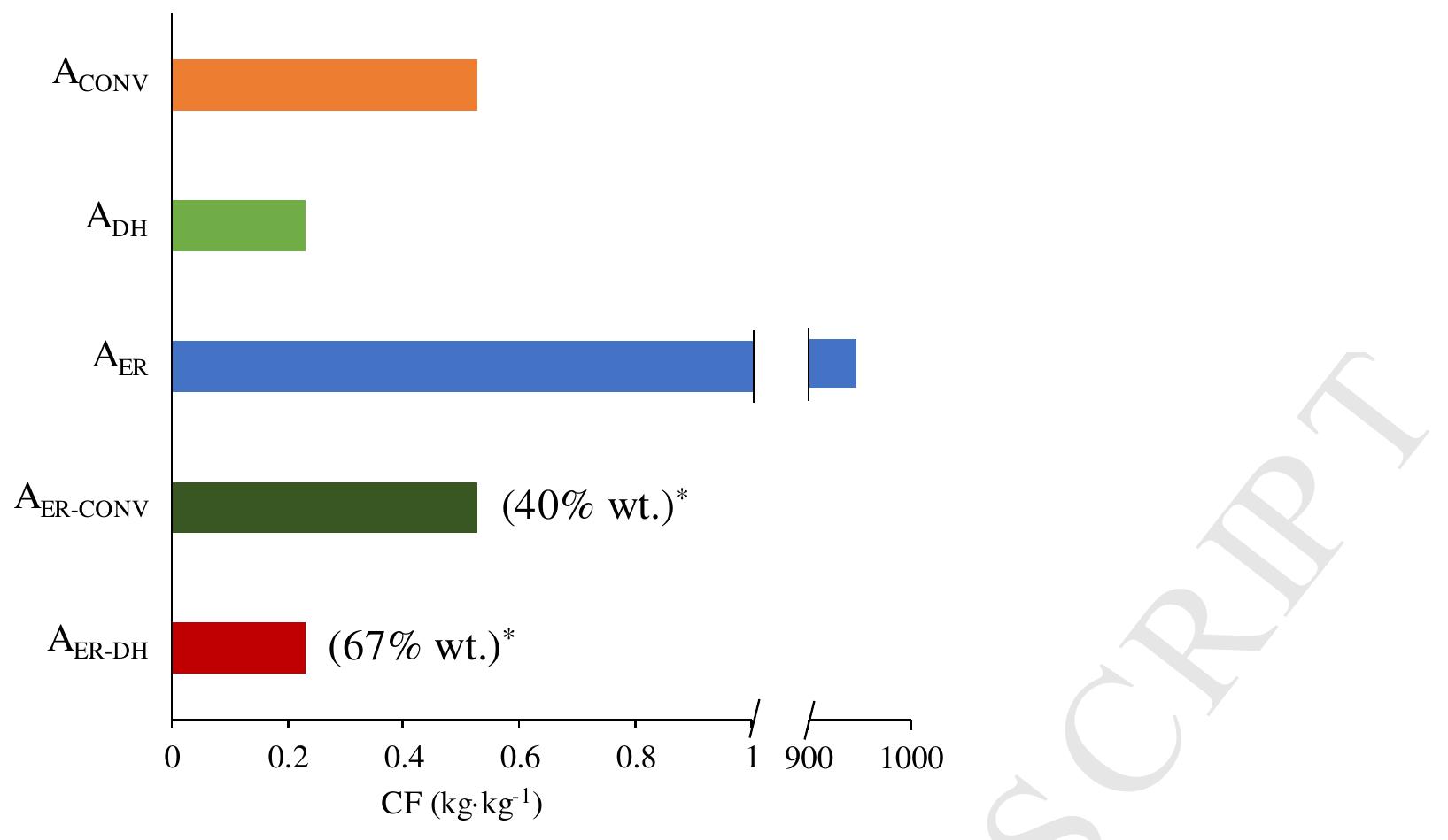
![Figure 3. CF values for the selected scenarios (functional unit is 1 kg of FA at a commercial concentration of 85% wt). After a proper LCI was available for each individual case of production of FA, the CF calculations were accomplished with the values of the resources obtained in each alternative. It is worth mentioning that no direct emissions from the ER process are considered. Also, it must be highlighted that the CF calculation was run only for the Al.1, A1.2, and A1.3 alternatives, as the CF values for Acony and Ajcr were provided by the corresponding references [6,7,27]. Ecoinvent database was used to obtain the CO2 equivalent emissions for the requested resources such as conventional FA production, H20, Hp, Oz, and steam [27], and the CO, equivalent emissions for the requested PV solar energy were based on the results of a previous study [31]. The CO used as feedstock was considered not to contribute to the CF, as all the emissions are allocated to the corresponding main product, which can be electricity in the case of a coal-fired power plant. Figure 3 displays the CF values corresponding to each alternative, considering the next 5 resources: (i) steam for purification; (ii) avoided emissions from the byproduct O; (iii) avoided emissions from the byproduct Hp; (iv) electricity as PV solar energy for the reactor, the pumping, and the compression/liquefaction of Hz /Oz; and, finally, (v) the ionized water as raw material, using a functional unit of 1 kg of FA as reference, considering that the FA is at a concentration of 85% wt. The mass of CO2 per mass unit of FA was excluded from the use of resources, but in any case, it would represent a value of —1 ke-keot. The CF of the electrode materials and catalysers were neglected, as their lifetimes were assumed to be long enough.](https://figures.academia-assets.com/108205035/figure_003.jpg)
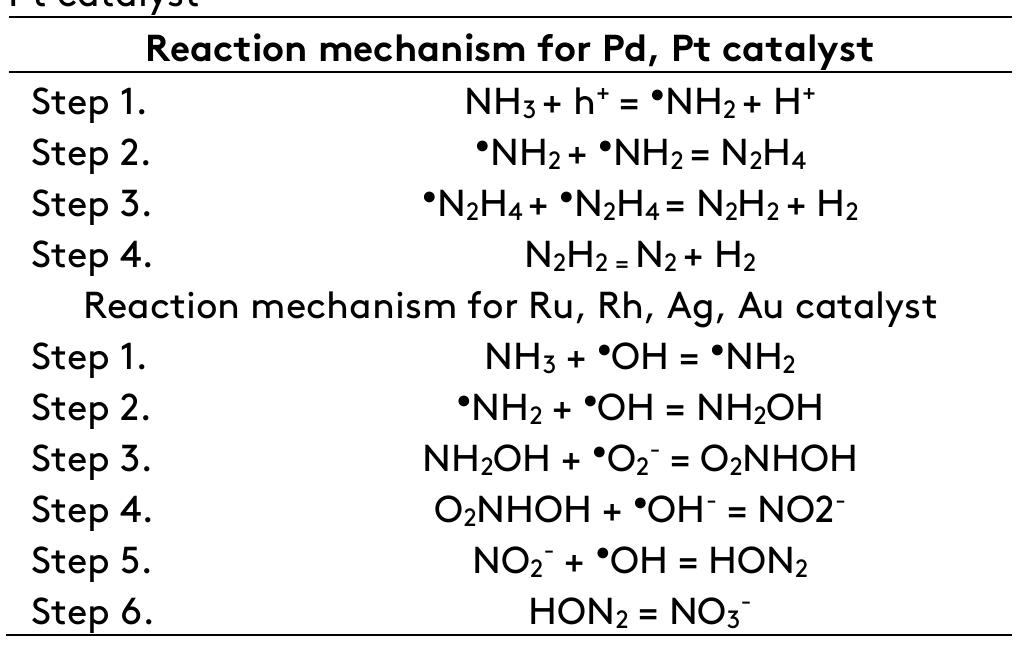
![Fig. 2. Process of electrochemical reaction on the surface of a catalyst. TiO2 occurred was observed to be more than that of Electrochemical methods constitute the use of a cathode and anode assembly immersed in an electrolyte containing the contaminant solution in an undivided or divided reactor cell. A DC supply was to be provided for the passing of current and an electrolyte for the exchange of ions/electrons [53]. Ewans et al. deciphered those reactions as electrochemical reactions in which the chemical reaction was induced due to charge transfer between the electrodes and across the interface between the electrode and electrolyte [54]. For any electrochemical reaction, the correct combination of cathode and anode affects the product selectivity in oxidation and reduction processes. A noncatalyzed EC process has similar shortcomings to that of a non-catalyzed PC process; the use of catalyzed EC methods can solve the limitations of non-catalyzed ECs. Figure 2 explains the mechanism of an_ electrochemical reaction occurring on the surface of different forms of catalystexposed. 3. Use of electrocatalysts nitrogen compounds from the wastewater. A stud) of noble metals like Ag, Au, Pd, Pt, Ir, Sn, and Fe was carried out in the EC oxidation of ammonia anc reduction of nitrate [55,56]. Further, noble metal: were loaded on the surface of catalysts forminc bimetallic catalysts systems or cocatalyst:](https://figures.academia-assets.com/105232543/figure_002.jpg)

![Fig. 1. Process of photochemical reaction on the surface of a catalyst A non-catalyzed photochemical process occurs by the irradiation of dissolved oxygen, thereby generating an oxidant species. But the subsequent decrease of dissolved oxygen discontinues the reaction after some time. This creates a need for research and development of novel catalyzed photocatalytic processes. The basic principle applicable in catalyzed PCs is the generation of reactive oxygen species by indenting light on a photocatalyst. A photocatalyst is a substance that does not directly take part in the reaction, but it actuates the reaction by absorbing light and generating the required electron/hole combinations. The light source may be natural sunlight (visible spectrum range) or an artificial UV A semiconducting material is the best-suited material for a photocatalyst due to its ability to conduct electricity when subjected to light of sufficient intensity (wavelength) at room temperature. The semiconductor absorbs the incident photon, which has higher energy than that of the bandgap of the material. Figure 1 explains the mechanism occurring in any photochemical process. These generated energy carriers (e and h*) migrate to the surface [29]. The photocatalytic reaction is dependent on the energy potential of the valence band (Ew), energy potential of the conduction band (E.b), and the required reaction potential for the oxidation and reduction reactions. Heterogeneous redox photocatalysis is a widely adopted method of contaminant removal in wastewater applications. The catalyzed PC reaction overcomes the_ limitation of the noncatalyzed process, and it is a noteworthy approach for indemnification of toxicity imparted by ammonia and_ nitrate compounds in wastewater.](https://figures.academia-assets.com/105232543/figure_001.jpg)


